Abstract
Treatment of indolent lymphoma has improved significantly in recent decades since the advent of rituximab (anti-CD20 monoclonal antibody). Although, some patients with limited disease can be cured with radiation therapy alone, most patients experience disease progression and recurrence during follow-up despite early initiation of treatment. Thus, watch-and-wait is still regarded the standard for asymptomatic patients. Patients with indolent lymphoma have a significant heterogeneity in terms of tumor burden, symptoms (according to anatomical sites) and the need for instant therapy. Therefore, the initiation of treatment and treatment option should be decided with a clear goal in each patient according to the need for therapy and clinical benefits with the chosen treatment. In this review, we cover the current treatment of follicular lymphoma and marginal zone lymphoma.
Indolent lymphoma refers to a type of slowly growing non-Hodgkin’s lymphoma (NHL) that has a long clinical course and is usually incurable. Indolent lymphomas include follicular lymphoma (FL), marginal zone lymphoma (MZL), small lymphocytic lymphoma (SLL), lymphoplasmacytic lymphoma and cutaneous T-cell lymphomas. Indolent lymphomas comprise 35–45% of NHL, with FL being the most frequent [1, 2]. In South Korea, MZL is the most common indolent lymphoma (21%), and the incidence of MZL and FL has been increasing in recent years [2, 3].
The majority of patients with indolent lymphoma can be observed provided they do not have symptoms due to lymphoma or rapid progression. For patients with indolent B cell lymphoma, current first-line treatments [radiotherapy (RT), anti-CD20 monoclonal antibodies, and chemoimmunotherapy] achieve a quite long progression-free interval and the expected overall survival (OS) is well beyond 20 years in rituximab era [4].
While indolent lymphomas are usually responsive to many treatment modalities, the protracted nature of the disease requires patients to be managed over a lifetime. Thus, careful judgement on the need of treatment and proper treatment modality should be made before the initiation of treatment considering the clinical situation and patient’s need. In this article, we overview the current treatment of the two most common indolent lymphomas; FL and MZL.
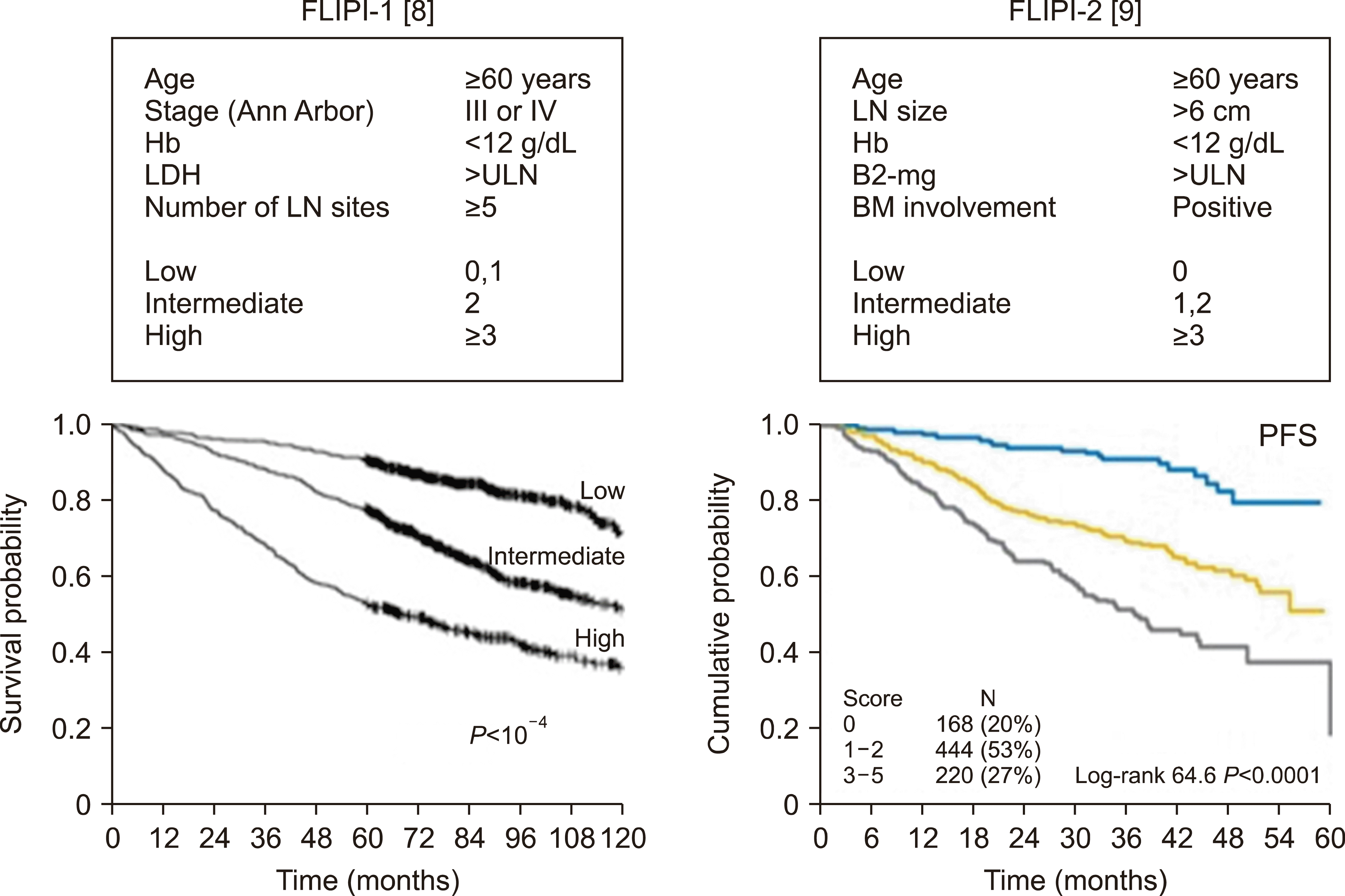
Follicular lymphoma comprises about 20% of all newly diagnosed non-Hodgkin’s lymphomas (NHLs) [5]. BCL2 is characteristically over-expressed in about 90% of FL patients by t(14;18) translocation, leading to anti-apoptotic properties [6]. Pathologic grading based on the number of centroblasts predicts clinical outcomes. FL3B with BLC-6 translocation shows more aggressive clinical course similar to diffuse large B-cell lymphoma (DLBCL) [7]. Thus, FL3B is treated as DLBCL and the others (FL1-2 and FL3A) are treated according to the guidelines for FL. The Follicular Lymphoma International Prognostic Index (FLIPI) delineates three distinct prognostic groups and FLIPI-2 is more predictive for patients who are treated in rituximab era (Fig. 1) [8, 9]. However, these prognostic models do not guide the initiation of treatment and treatment modalities. Initiation of treatment is generally guided by GELF criteria [10] or its modified version (Table 1).
Limited disease: About 10–15% of patients are diagnosed with FL are at a limited stage (stage I and II). RT is effective in the treatment of limited stage disease with long-term disease control rates of >90%. RT could be a curative treatment for limited stage FL [11]. RT dose of 24Gy is enough to control limited disease and additional dose is not beneficial [12]. Extended-field RT does not improve overall survival (OS) and also reduction of radiation field did not affect progression-free survival (PFS) [13, 14]. The addition of systemic therapy (rituximab or chemoimmunotherapy) improves PFS but not OS [15-17]. For selected patients, watch-and-wait could be a reasonable option given that there were no differences in OS outcomes between different treatment modalities [18, 19]. In a study, 67% of the patients did not require therapy at a median follow-up of 7 years [20]. Bulky and non-contiguous disease can be treated with rituximab or chemoimmunotheapy (CIT) with or without RT (Table 2).
Advanced disease: Watch-and-wait is regarded standard practice for advanced stage FL with low tumor burden even in the rituximab era, as there is no survival benefit with early treatment [21-23]. Initiation of treatment should be guided by indications for treatment (Table 1).
CIT is the most commonly used first-line therapy for patients with advanced stage FL. In a randomized phase III trial (FOLL-05), the efficacy of RCVP (rituximab, cyclophosphamide, vincristine and prednisone) and RCHOP (rituximab, cyclophosphamide, doxorubicin, vincristine and prednisone) was compared with RFM (rituximab, fludarabine, and mitoxantrone) as a first-line treatment in patients with advanced stage FL. With a median follow-up of 34 months, the 3-year progression-free survival (PFS) was 52%, 68% and 63%, respectively (P=0.011). RCHOP had less grade 3 or 4 neutropenia and secondary malignancies than RFM. RCVP was inferior to other combinations, thus RCVP should be avoided in patients who have high-risk features [24].
In a phase III trial (Stil NHL1) that compared BR (bendamustine and rituximab) and RCHOP as first-line treatment for patients with indolent lymphoma and mantle cell lymphoma (MCL), the overall response rate (ORR) was not different between BR and RCHOP (BR 93% vs. RCHOP 91%). However, with a median follow-up of 45 months, BR showed superior PFS than RCHOP (69 mo vs. 31 mo, P<0.001) without OS difference [25]. In a following phase III trial (BRIGHT), BR was confirmed to be at least not inferior in terms of complete response (CR) and PFS [26].
Obinutuzumab is a new type II anti-CD20 monoclonal antibody with enhanced antibody-dependent cellular cytotoxicity. In a phase III study (GALLIUM), obinutuzumab was compared with rituximab as first-line treatment in combination with chemotherapy (bendamustine, CVP or CHOP) in previously untreated, advanced stage FL patients [27]. Although, 3-year PFS was superior in obinutuzumab arm (80% vs. 73%, HR 0.66), serious infection, mortality and secondary malignancies were higher in Obinutuzumab arm. Thus, the benefits of obinutuzumab over rituximab as first-line therapy are not clear.
Based on promising results of a lenalidomide and rituximab combination in phase II studies [28, 29], a phase III trial (RELEVANCE) of lenalidomide and rituximab combination was conducted. Lenalidomide and rituximab combination showed similar 3-year PFS (77%) compared to rituximab-based CIT (78%) with lower grade 3 or 4 neutropenia and febrile neutropenia [30].
Rituximab monotherapy could be considered for patients who are not candidate for CIT as it showed high response rate (72–73%) with a median time to progression of 2 years for patients with low tumor burden [31, 32].
In a phase III trial (E4402 study, RESORT), rituximab maintenance and rituximab retreatment were compared in patients with low tumor burden FL who responded to rituximab induction therapy. In the study, rituximab retreatment is shown to be comparable to rituximab maintenance therapy in term of treatment failure (3.9 yr vs. 4.3 yr, P=0.54) while saving rituximab dose [37].
Rituximab maintenance following CIT (RCVP, RCHOP, or RFCM) also showed improved 3-year PFS (75% vs. 52%, P=0.001) in a phase III trial (PRIMA) [38]. Although half of the patients in maintenance group remained progression-free at 10 years, the estimated OS did not differ between the two groups (80%). Obinutuzumab maintenance following obinutuzumab-containing CIT is also available based on the GALLIUM trial [27]. For patients who achieved PR with BR treatment, rituximab maintenance improved duration of response in a retrospective analysis, but not in patients achieving CR [39].
For a relapsing disease, biopsy confirmation is strongly recommended to exclude transformation to a high-grade lymphoma. About 20% of patients with FL have transformation at disease progression [40]. FDG-PET scan is useful to guide optimal biopsy site (with highest intensity uptake).
Watch-and-wait is still valid option for relapsed FL patients with low tumor burden without transformation to high grade lymphoma. Symptomatic single lesion could be successfully relieved with low dose radiation (4Gy). For patients who need second-line therapy, there are many treatment options including rituximab monotherapy, CIT, lenalidomide-based combination, PI3K inhibitors and hematopoietic stem cell transplantation (HSCT) (Table 2). The clinical situation is different for each patient depending on disease characteristics, the first-line therapy and timing of progression. Thus, in order to select the optimal treatment, it is important to consult with the patient about the purpose of treatment before making any treatment decisions.
Rituximab monotherapy could be considered for relapsed FL patients with low tumor burden. Rituximab monotherapy induces response in half of the patients with relapsed FL [41]. For patients who had rituximab induction therapy could retreated with rituximab if the progression of disease (POD) is long enough (>24 mo).
CIT is the most favored second-line therapy. Initial treatment could be used again in late relapse (>2 yr). However, BR is not generally recommended for patients who were treated with BR as first-line therapy because of the increased risk of infections and secondary malignancies. BR showed longer PFS (34 mo) in patients with relapsed or refractory indolent lymphoma compared with fludarabine and rituximab (12 mo) [42]. The addition of rituximab improved PFS by 12 months compared with CHOP in patients with relapsed FL [43]. However, this outcome could not be reproducible in the current practice because most of FL patients treated with rituximab or anthracycline containing regimen as first-line therapy.
Obinutuzumab-based CIT was evaluated in several studies. In a randomized phase III trial (GADOLIN), bendamustine and obinutuzumab combination improve PFS compared with bendamustine monotherapy (25 mo vs. 14 mo, P<0.001) in patients with rituximab-refractory indolent lymphoma [44, 45].
Rituximab or obinutuzumab maintenance could be used after achieving response [44-47]. However, for patients who progressed early after rituximab-containing treatment, obinutuzumab is preferred for its potential to overcome rituximab-resistance [48].
Single agent lenalidomide showed ORR of 23% for patients with relapsed FL with median response duration >16 months [49]. In a randomized phase II trial (CALGB 50401), the addition of rituximab to lenalidomide improved the ORR (76% vs. 53%, P=0.029) and the median time to progression (2 yr vs. 1 yr) [50]. This result was confirmed in a randomized phase III trial (AUGMENT), in which lenalidomide and rituximab combination induced the median PFS of 39 months (14 mo for lenalidomide group, P<0.01) for patients with previously treated FL [51]. Early relapse after CIT (POD≤24) is a validated prognostic factor for survival in patients with FL [52]. The patients with POD≤24 months could be treated with chemotherapy-free combination of rituximab and lenalidomide.
Several PI3K inhibitors (idelalisib, copanlisib, duvelisib, umbralisib) were FDA-approved for relapsed/refractory FL after 2 prior therapies with response rate of 45–61% and the median duration of response of 10–12 months [53-55]. However, the use of idelalisib is restricted by its considerable toxicities such as pneumonitis, transaminitis, and opportunistic infections. In a post hoc analysis, copanlisib is as effective in FL patients with POD≤24 months as in patients with POD >24 months (ORR 60%) and has better toxicity profiles. However, hyperglyemia and hypertension as well as relative inconvenience of frequent visits for intravenous injection are problematic. Develisib and umbralisib showed relatively favorable toxicity profiles compared to idelarisib. Many clinical trials testing PI3K inhibitors in combination with other treatments are ongoing.
Among epigenetic regulators, tazemetostat showed an ORR of 69% (CR 13%) and the median PFS of 14 months for EZH-mutant FL patients [56].
Y-ibritumomab tiuxetan showed significantly higher ORR (80% vs. 56%) and CR rate (30% vs.16%) compared to rituximab monotherapy [57]. Time to progression (15 mo vs. 10 mo) and response duration (17 mo vs. 11 mo) were longer in patients treated with Y-ibritumomab [58]. However, its use is currently limited. Auto-HSCT consolidation showed improved OS and PFS in patients with refractory or relapsed disease [59-61]. For younger patients who experienced multiple recurrence, allo-HSCT could be considered as curative treatment, although no supportive data has published yet [62].
In a phase II trial (ZUMA-5), axicabtagene ciloleucel (axi-cel) achieved 94% of ORR (CR 79%) in patients with refractory/relapsed FL who had more than two lines of previous treatment including anti-CD20 monoclonal antibodies and anthracycline containing regimen [63]. With a median follow-up of 17.5 months, 62% of the FL patients had ongoing responses. In two clinical studies evaluated different CAR-T cell therapies in NHL patients, liso-cel and tisa-cel showed promising outcomes (2-year PFS 60%) even in transformed FL patients [64-66].
MZL can occur in lymph nodes (nodal MZL), spleen (splenic MZL) and extranodal sites (ENMZL). Overall, MZLs comprise about 5–15% of all NHLs in western countries. In Korea, MZL is the most common low-grade lymphoma comprising 21% of mature B cell neoplasms [2]. Chronic immune stimulation by diverse infectious agents such as Helicobacter pylori (H. pylori), chlamydia psittaci (C. psittaci), Campylobacter jejuni (C. jejuni) and Borrelia burgdorferi (B. burgdorferi), or chronic inflammation seems to play an important role in the pathogenesis of MZLs [67]. These causative agents could be targeted for the treatment lymphoma before conventional cancer treatments.
H. pylori infection plays central role in the pathogenesis of gastric ENMZL [70, 71]. For patients with H. pylori infection, eradication of H. pylori induces regression of lymphoma in most cases (70–95%) with excellent long-term survival [72, 73]. Eradication regimens generally include a proton pump inhibitor and a combination of two different antibiotics (clarithromycin plus amoxicillin or metronidazole) [74, 75]. Predictive factors of resistance to H. pylori eradication include t(11;18), H. pylori-negativity, and submucosal invasion [76]. H. pylori eradication was more effective in gastric MALT lymphoma involving distal part [77]. When H. pylori eradication is not effective, second line eradication should be tried with alternative combinations. Microscopic persistence of lymphoma is relatively common after clinical regression of lymphoma and should be followed at least 12 months before decide another treatment [78]. H. pylori eradication could be tried in H. pylori negative patients, because some patients respond to H. pylori eradication possibly due to false negativity or infection of other Helicobacter species.
After failure with H. pylori eradication, asymptomatic patients can be observed. Involved sited radiation therapy (ISRT) is very effective inducing long-term remission in patients who experienced treatment failure with H. pylori eradication [79, 80].
Rituximab monotherapy was effective in patients with relpased/refractory to H. pylori eradication. With an ORR of 77% (CR 46%) and 54% of disease-free survival at a median follow-up of 28 months [81]. In a retrospective study, rituximab resulted in an ORR of 73% with favorable long-term survival (5-year OS and PFS, 70% and 95%, respectively) [82].
Surgical resection is also effective in the treatment of gastric ENMZL [83], However, surgical treatment should be reserved for patients who have justifiable reasons for surgery (major bleeding, perforation and obstruction) given long-term consequences of gastrectomy and effectiveness of other treatment options.
For advanced disease, treatment should be initiated when the indication of treatment is present. Treatment options are similar to advanced FL (Table 3).
Non-gastric ENMZL also have antigenic stimulus from infectious agents according to the disease sites [84]. Although the pathogenesis is not fully elucidated, a critical role for an antigenic driver has been postulated. Unlike in gastric ENMZL, treatment of underlying infectious causes does not guarantee the remission of disease in non-gastric ENMZLs. However, antibiotic therapy for C.psittaci in ENMZL involving ocular adnexa and HCV treatment in HCV infected patients could be considered if urgent treatment is not needed [85, 86].
ISRT should be considered as initial treatment for patients with localized non-gastric ENMZL involving ocular adnexa, salivary gland and thyroid. Which induced favorable 10-year relapse-free survival (74%) and OS (89%) in a retrospective analysis [87]. Recently lower dose RT is favored for its excellent local control with less radiation-related side effects [88]. Especially for ocular lymphoma, low-dose RT has definite benefit [89]. For elderly patients or frail patients who are not suitable for systemic therapy low-dose RT could be a reasonable treatment option.
Surgical resection is performed usually for the diagnosis of ENMZL involving specific anatomical sites such as thyroid, breast or colon. With completely resected lymphomas, no additional therapy is needed.
For asymptomatic patients watch-and-wait is also reasonable strategy. For patients who need therapy, single agent rituximab could be an option [90, 91]. Rituximab is more effective in patients who were not exposed to chemotherapy (ORR 87% vs. 45%, P=0.03) [90].
Chemoimmunotherapy of chlorambucil and rituximab improved 5-year event-free survival (68%) compared with chlorambucil (51%) or rituximab monotherapy (50%) in patients with ENMZL [92]. Bendamustine and rituximab combination showed good response in patients with ENMZL including patients with t(11;18) [93].
Lenalidomide and rituximab combination showed good response (ORR 80%, CR 54%) and durable response (91% of patients are progression-free at 27 mo) in patient with ENMZL avoiding chemotherapy-related side effects in a phase II trial [94].
Other regimens such as rituximab and fludarabine combination or anthracycline-base combinations are also effective, especially for patients with aggressive clinical course [95].
Splenic MZL typically presents with splenomegaly with or without cytopenias. More than 80% of patients show bone marrow involvement. Thus, the diagnosis of splenic MZL could be suggested with bone marrow specimen in many cases before splenectomy [96, 97].
Patients who do not have lymphoma-related symptoms or progressive disease could be followed clinically without treatment for a quite long time, given that early treatment does not improve survival outcomes [98, 99]. In patients who have hepatitis C infection, treatment of HCV infection is considered first because HCV eradication could induce lymphoma regression in many cases (73–90%) [100-102]. Interferon-free treatment with direct acting antiviral (DAA) agents was also suggested in a recent study [101]. In the study, lymphoma response was observed in 73% (27/37) of patients with MZL. For a proper treatment of HCV infection, hepatology consultation is recommended.
Traditionally splenectomy was considered as first therapy for splenic MZL. However, single agent rituximab showed favorable treatment outcomes without splenectomy [103-107]. In a retrospective analysis (N=108), rituximab monotherapy (375 mg/m2 4–8 weekly dose) induced an ORR 92% (44% CR) with a favorable 10-year PFS (64%) without significant toxicities. Furthermore, the effect of rituximab treatment lasted long duration [107, 108].
Rituximab monotherapy is also very useful for patients who have autoimmune cytopenias.
In a retrospective analysis including 226 patients, single agent rituximab offered the most risk/benefit ratio [106].
Given that splenectomy has significant side effects such as the risk of infection by encapsulated bacteria, splenectomy should be reserved for the diagnosis of histological transformation and refractory disease to rituximab therapy.
For patients who progress after rituximab therapy could be treated with rituximab re-treatment, splenectomy, combination chemotherapy or chemoimmunotherapy.
In recent decades, treatment of indolent lymphoma has advanced significantly and now there are many more treatment options including chemotherapy-free combination, new molecular targeted agents and CAR-T cell therapy for patients with indolent lymphoma. Treatment of indolent lymphoma involves prudency and endurance in both patient and clinician, given its long-term clinical course and frequent recurrence. Treatment modality should be chosen or sequenced to maximize the treatment effect and quality of life at the same time. Finally, special attention should always be paid to each disease progression to rule out the possibility of histologic transformation.
REFERENCES
1. Al-Hamadani M, Habermann TM, Cerhan JR, Macon WR, Maurer MJ, Go RS. 2015; Non-Hodgkin lymphoma subtype distribution, geodemographic patterns, and survival in the US: a longitudinal analysis of the National Cancer Data Base from 1998 to 2011. Am J Hematol. 90:790–5. DOI: 10.1002/ajh.24086. PMID: 26096944.

2. Yoo KH, Lee H, Suh C. CISL. 2018; Lymphoma epidemiology in Korea and the real clinical field including the Consortium for Improving Survival of Lymphoma (CISL) trial. Int J Hematol. 107:395–404. DOI: 10.1007/s12185-018-2403-9. PMID: 29357080.

3. Kim JS, Liu Y, Ha KH, Qiu H, Rothwell LA, Kim HC. 2020; Increasing incidence of B-cell non-Hodgkin lymphoma and occurrence of second primary malignancies in South Korea: 10-year follow-up using the Korean National Health Information Database. Cancer Res Treat. 52:1262–72. DOI: 10.4143/crt.2020.089. PMID: 32599988. PMCID: PMC7577801.

4. Tan D, Horning SJ, Hoppe RT, et al. 2013; Improvements in observed and relative survival in follicular grade 1-2 lymphoma during 4 decades: the Stanford University experience. Blood. 122:981–7. DOI: 10.1182/blood-2013-03-491514. PMID: 23777769. PMCID: PMC3739040.

5. Swerdlow SH, Campo E, Pileri SA, et al. 2016; The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. 127:2375–90. DOI: 10.1182/blood-2016-01-643569. PMID: 26980727. PMCID: PMC4874220.

6. Ngan BY, Chen-Levy Z, Weiss LM, Warnke RA, Cleary ML. 1988; Expression in non-Hodgkin's lymphoma of the bcl-2 protein associated with the t(14;18) chromosomal translocation. N Engl J Med. 318:1638–44. DOI: 10.1056/NEJM198806233182502. PMID: 3287162.

7. Katzenberger T, Ott G, Klein T, Kalla J, Müller-Hermelink HK, Ott MM. 2004; Cytogenetic alterations affecting BCL6 are predo-minantly found in follicular lymphomas grade 3B with a diffuse large B-cell component. Am J Pathol. 165:481–90. DOI: 10.1016/S0002-9440(10)63313-5. PMID: 15277222. PMCID: PMC1618575.

8. Solal-Celigny P, Roy P, Colombat P, et al. 2004; Follicular lymphoma international prognostic index. Blood. 104:1258–65. DOI: 10.1182/blood-2003-12-4434. PMID: 15126323. PMCID: PMC6938796.

9. Federico M, Bellei M, Marcheselli L, et al. 2009; Follicular lymphoma international prognostic index 2: a new prognostic index for follicular lymphoma developed by the international follicular lymphoma prognostic factor project. J Clin Oncol. 27:4555–62. DOI: 10.1200/JCO.2008.21.3991. PMID: 19652063.

10. Brice P, Bastion Y, Lepage E, et al. 1997; Comparison in low-tumor- burden follicular lymphomas between an initial no-treatment policy, prednimustine, or interferon alfa: a randomized study from the Groupe d'Etude des Lymphomes Folliculaires. Groupe d'Etude des Lymphomes de l'Adulte. J Clin Oncol. 15:1110–7. DOI: 10.1200/JCO.1997.15.3.1110. PMID: 9060552.
11. Brady JL, Binkley MS, Hajj C, et al. 2019; Definitive radiotherapy for localized follicular lymphoma staged by (18)F-FDG PET-CT: a collaborative study by ILROG. Blood. 133:237–45. DOI: 10.1182/blood-2018-04-843540. PMID: 30446493.

12. Lowry L, Smith P, Qian W, et al. 2011; Reduced dose radiotherapy for local control in non-Hodgkin lymphoma: a randomised phase III trial. Radiother Oncol. 100:86–92. DOI: 10.1016/j.radonc.2011.05.013. PMID: 21664710.

13. Wilder RB, Jones D, Tucker SL, et al. 2001; Long-term results with radiotherapy for Stage I-II follicular lymphomas. Int J Radiat Oncol Biol Phys. 51:1219–27. DOI: 10.1016/S0360-3016(01)01747-3. PMID: 11728680.

14. Campbell BA, Voss N, Woods R, et al. 2010; Long-term outcomes for patients with limited stage follicular lymphoma: involved regional radiotherapy versus involved node radiotherapy. Cancer. 116:3797–806. DOI: 10.1002/cncr.25117. PMID: 20564082.

15. Yahalom J, Varsos G, Fuks Z, Myers J, Clarkson BD, Straus DJ. 1993; Adjuvant cyclophosphamide, doxorubicin, vincristine, and prednisone chemotherapy after radiation therapy in stage I low-grade and intermediate-grade non-Hodgkin lymphoma. Results of a prospective randomized study. Cancer. 71:2342–50. DOI: 10.1002/1097-0142(19930401)71:7<2342::AID-CNCR2820710728>3.0.CO;2-I. PMID: 8453557.

16. Ruella M, Filippi AR, Bruna R, et al. 2016; Addition of rituximab to involved-field radiation therapy prolongs progression-free survival in stage I-II follicular lymphoma: results of a multicenter study. Int J Radiat Oncol Biol Phys. 94:783–91. DOI: 10.1016/j.ijrobp.2015.12.019. PMID: 26972651.

17. MacManus M, Fisher R, Roos D, et al. 2018; Randomized trial of systemic therapy after involved-field radiotherapy in patients with early-stage follicular lymphoma: TROG 99.03. J Clin Oncol. 36:2918–25. DOI: 10.1200/JCO.2018.77.9892. PMID: 29975623.

18. Friedberg JW, Byrtek M, Link BK, et al. 2012; Effectiveness of first-line management strategies for stage I follicular lymphoma: analysis of the National LymphoCare Study. J Clin Oncol. 30:3368–75. DOI: 10.1200/JCO.2011.40.6546. PMID: 22915662. PMCID: PMC3675665.

19. Michallet AS, Lebras LL, Bauwens DD, et al. 2013; Early stage follicular lymphoma: what is the clinical impact of the first-line treatment strategy? J Hematol Oncol. 6:45. DOI: 10.1186/1756-8722-6-45. PMID: 23816219. PMCID: PMC3723590.

20. Advani R, Rosenberg SA, Horning SJ. 2004; Stage I and II follicular non-Hodgkin's lymphoma: long-term follow-up of no initial therapy. J Clin Oncol. 22:1454–9. DOI: 10.1200/JCO.2004.10.086. PMID: 15024027.

21. Nastoupil LJ, Sinha R, Byrtek M, et al. 2016; Outcomes following watchful waiting for stage II-IV follicular lymphoma patients in the modern era. Br J Haematol. 172:724–34. DOI: 10.1111/bjh.13895. PMID: 26729445.

22. Solal-Céligny P, Bellei M, Marcheselli L, et al. 2012; Watchful waiting in low-tumor burden follicular lymphoma in the rituximab era: results of an F2-study database. J Clin Oncol. 30:3848–53. DOI: 10.1200/JCO.2010.33.4474. PMID: 23008294.

23. Ardeshna KM, Qian W, Smith P, et al. 2014; Rituximab versus a watch-and-wait approach in patients with advanced-stage, asymptomatic, non-bulky follicular lymphoma: an open-label randomised phase 3 trial. Lancet Oncol. 15:424–35. DOI: 10.1016/S1470-2045(14)70027-0. PMID: 24602760.

24. Luminari S, Ferrari A, Manni M, et al. 2018; Long-term results of the FOLL05 trial comparing R-CVP versus R-CHOP versus R-FM for the initial treatment of patients with advanced-stage symptomatic follicular lymphoma. J Clin Oncol. 36:689–96. DOI: 10.1200/JCO.2017.74.1652. PMID: 29095677.

25. Rummel MJ, Niederle N, Maschmeyer G, et al. 2013; Bendamustine plus rituximab versus CHOP plus rituximab as first-line treatment for patients with indolent and mantle-cell lymphomas: an open-label, multicentre, randomised, phase 3 non-inferiority trial. Lancet. 381:1203–10. DOI: 10.1016/S0140-6736(12)61763-2. PMID: 23433739.

26. Flinn IW, van der Jagt R, Kahl BS, et al. 2014; Randomized trial of bendamustine-rituximab or R-CHOP/R-CVP in first-line treatment of indolent NHL or MCL: the BRIGHT study. Blood. 123:2944–52. DOI: 10.1182/blood-2013-11-531327. PMID: 24591201. PMCID: PMC4260975.

27. Marcus R, Davies A, Ando K, et al. 2017; Obinutuzumab for the first-line treatment of follicular lymphoma. N Engl J Med. 377:1331–44. DOI: 10.1056/NEJMoa1614598. PMID: 28976863.

28. Fowler NH, Davis RE, Rawal S, et al. 2014; Safety and activity of lenalidomide and rituximab in untreated indolent lymphoma: an open-label, phase 2 trial. Lancet Oncol. 15:1311–8. DOI: 10.1016/S1470-2045(14)70455-3. PMID: 25439689. PMCID: PMC4370362.

29. Martin P, Jung SH, Pitcher B, et al. 2017; A phase II trial of lenalidomide plus rituximab in previously untreated follicular non-Hodgkin's lymphoma (NHL): CALGB 50803 (Alliance). Ann Oncol. 28:2806–12. DOI: 10.1093/annonc/mdx496. PMID: 28945884. PMCID: PMC5789808.

30. Morschhauser F, Fowler NH, Feugier P, et al. 2018; Rituximab plus lenalidomide in advanced untreated follicular lymphoma. N Engl J Med. 379:934–47. DOI: 10.1056/NEJMoa1805104. PMID: 30184451.

31. Colombat P, Salles G, Brousse N, et al. 2001; Rituximab (anti-CD20 monoclonal antibody) as single first-line therapy for patients with follicular lymphoma with a low tumor burden: clinical and molecular evaluation. Blood. 97:101–6. DOI: 10.1182/blood.V97.1.101. PMID: 11133748.

32. Witzig TE, Vukov AM, Habermann TM, et al. 2005; Rituximab therapy for patients with newly diagnosed, advanced-stage, follicular grade I non-Hodgkin's lymphoma: a phase II trial in the North Central Cancer Treatment Group. J Clin Oncol. 23:1103–8. DOI: 10.1200/JCO.2005.12.052. PMID: 15657404.

33. Ghielmini M, Schmitz SF, Cogliatti SB, et al. 2004; Prolonged treatment with rituximab in patients with follicular lymphoma significantly increases event-free survival and response duration compared with the standard weekly x 4 schedule. Blood. 103:4416–23. DOI: 10.1182/blood-2003-10-3411. PMID: 14976046.

34. Martinelli G, Schmitz SF, Utiger U, et al. 2010; Long-term follow-up of patients with follicular lymphoma receiving single-agent rituximab at two different schedules in trial SAKK 35/98. J Clin Oncol. 28:4480–4. DOI: 10.1200/JCO.2010.28.4786. PMID: 20697092.

35. Taverna C, Martinelli G, Hitz F, et al. 2016; Rituximab maintenance for a maximum of 5 years after single-agent rituximab induction in follicular lymphoma: results of the randomized controlled phase III trial SAKK 35/03. J Clin Oncol. 34:495–500. DOI: 10.1200/JCO.2015.61.3968. PMID: 26712227. PMCID: PMC4872010.

36. Barta SK, Li H, Hochster HS, et al. 2016; Randomized phase 3 study in low-grade lymphoma comparing maintenance anti-CD20 antibody with observation after induction therapy: a trial of the ECOG-ACRIN Cancer Research Group (E1496). Cancer. 122:2996–3004. DOI: 10.1002/cncr.30137. PMID: 27351685. PMCID: PMC5030179.

37. Kahl BS, Hong F, Williams ME, et al. 2014; Rituximab extended schedule or re-treatment trial for low-tumor burden follicular lymphoma: eastern cooperative oncology group protocol e4402. J Clin Oncol. 32:3096–102. DOI: 10.1200/JCO.2014.56.5853. PMID: 25154829. PMCID: PMC4171355.

38. Salles G, Seymour JF, Offner F, et al. 2011; Rituximab maintenance for 2 years in patients with high tumour burden follicular lymphoma responding to rituximab plus chemotherapy (PRIMA): a phase 3, randomised controlled trial. Lancet. 377:42–51. DOI: 10.1016/S0140-6736(10)62175-7. PMID: 21176949.

39. Hill BT, Nastoupil L, Winter AM, et al. 2019; Maintenance rituximab or observation after frontline treatment with bendamustine- rituximab for follicular lymphoma. Br J Haematol. 184:524–35. DOI: 10.1111/bjh.15720. PMID: 30575016. PMCID: PMC6486816.

40. Sarkozy C, Trneny M, Xerri L, et al. 2016; Risk factors and outcomes for patients with follicular lymphoma who had histologic transformation after response to first-line immunochemotherapy in the PRIMA trial. J Clin Oncol. 34:2575–82. DOI: 10.1200/JCO.2015.65.7163. PMID: 27298402.

41. McLaughlin P, Grillo-López AJ, Link BK, et al. 1998; Rituximab chimeric anti-CD20 monoclonal antibody therapy for relapsed indolent lymphoma: half of patients respond to a four-dose treatment program. J Clin Oncol. 16:2825–33. DOI: 10.1200/JCO.1998.16.8.2825. PMID: 9704735.

42. Rummel M, Kaiser U, Balser C, et al. 2016; Bendamustine plus rituximab versus fludarabine plus rituximab for patients with relapsed indolent and mantle-cell lymphomas: a multicentre, randomised, open-label, non-inferiority phase 3 trial. Lancet Oncol. 17:57–66. DOI: 10.1016/S1470-2045(15)00447-7. PMID: 26655425.

43. van Oers MH, Klasa R, Marcus RE, et al. 2006; Rituximab maintenance improves clinical outcome of relapsed/resistant follicular non-Hodgkin lymphoma in patients both with and without rituximab during induction: results of a prospective randomized phase 3 intergroup trial. Blood. 108:3295–301. DOI: 10.1182/blood-2006-05-021113. PMID: 16873669.

44. Sehn LH, Chua N, Mayer J, et al. 2016; Obinutuzumab plus bendamustine versus bendamustine monotherapy in patients with rituximab-refractory indolent non-Hodgkin lymphoma (GADOLIN): a randomised, controlled, open-label, multicentre, phase 3 trial. Lancet Oncol. 17:1081–93. DOI: 10.1016/S1470-2045(16)30097-3. PMID: 27345636.

45. Cheson BD, Chua N, Mayer J, et al. 2018; Overall survival benefit in patients with rituximab-refractory indolent non-Hodgkin lymphoma who received obinutuzumab plus bendamustine induction and obinutuzumab maintenance in the GADOLIN study. J Clin Oncol. 36:2259–66. DOI: 10.1200/JCO.2017.76.3656. PMID: 29584548.

46. Forstpointner R, Unterhalt M, Dreyling M, et al. 2006; Maintenance therapy with rituximab leads to a significant prolongation of response duration after salvage therapy with a combination of rituximab, fludarabine, cyclophosphamide, and mitoxantrone (R-FCM) in patients with recurring and refractory follicular and mantle cell lymphomas: results of a prospective randomized study of the German Low Grade Lymphoma Study Group (GLSG). Blood. 108:4003–8. DOI: 10.1182/blood-2006-04-016725. PMID: 16946304.

47. van Oers MH, Van Glabbeke M, Giurgea L, et al. 2010; Rituximab maintenance treatment of relapsed/resistant follicular non- Hodgkin's lymphoma: long-term outcome of the EORTC 20981 phase III randomized intergroup study. J Clin Oncol. 28:2853–8. DOI: 10.1200/JCO.2009.26.5827. PMID: 20439641. PMCID: PMC2903319.

48. Freeman CL, Sehn LH. 2018; A tale of two antibodies: obinutuzumab versus rituximab. Br J Haematol. 182:29–45. DOI: 10.1111/bjh.15232. PMID: 29741753.

49. Witzig TE, Wiernik PH, Moore T, et al. 2009; Lenalidomide oral monotherapy produces durable responses in relapsed or refractory indolent non-Hodgkin's lymphoma. J Clin Oncol. 27:5404–9. DOI: 10.1200/JCO.2008.21.1169. PMID: 19805688.

50. Leonard JP, Jung SH, Johnson J, et al. 2015; Randomized trial of lenalidomide alone versus lenalidomide plus rituximab in patients with recurrent follicular lymphoma: CALGB 50401 (Alliance). J Clin Oncol. 33:3635–40. DOI: 10.1200/JCO.2014.59.9258. PMID: 26304886. PMCID: PMC4622102.

51. Leonard JP, Trneny M, Izutsu K, et al. 2019; AUGMENT: a phase III study of lenalidomide plus rituximab versus placebo plus rituximab in relapsed or refractory indolent lymphoma. J Clin Oncol. 37:1188–99. DOI: 10.1200/JCO.19.00010. PMID: 30897038. PMCID: PMC7035866.

52. Casulo C, Byrtek M, Dawson KL, et al. 2015; Early relapse of follicular lymphoma after rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone defines patients at high risk for death: an analysis from the national lymphocare study. J Clin Oncol. 33:2516–22. DOI: 10.1200/JCO.2014.59.7534. PMID: 26124482. PMCID: PMC4879714.

53. Flinn IW, Miller CB, Ardeshna KM, et al. 2019; DYNAMO: a phase II study of duvelisib (IPI-145) in patients with refractory indolent non-Hodgkin lymphoma. J Clin Oncol. 37:912–22. DOI: 10.1200/JCO.18.00915. PMID: 30742566.

54. Dreyling M, Santoro A, Mollica L, et al. 2017; Phosphatidylinositol 3-kinase inhibition by copanlisib in relapsed or refractory indolent lymphoma. J Clin Oncol. 35:3898–905. DOI: 10.1200/JCO.2017.75.4648. PMID: 28976790.

55. Gopal AK, Kahl BS, de Vos S, et al. 2014; PI3Kdelta inhibition by idelalisib in patients with relapsed indolent lymphoma. N Engl J Med. 370:1008–18. DOI: 10.1056/NEJMoa1314583. PMID: 24450858. PMCID: PMC4039496.

56. Morschhauser F, Tilly H, Chaidos A, et al. 2020; Tazemetostat for patients with relapsed or refractory follicular lymphoma: an open-label, single-arm, multicentre, phase 2 trial. Lancet Oncol. 21:1433–42. DOI: 10.1016/S1470-2045(20)30441-1. PMID: 33035457. PMCID: PMC8427481.

57. Witzig TE, Gordon LI, Cabanillas F, et al. 2002; Randomized controlled trial of yttrium-90-labeled ibritumomab tiuxetan radioimmuno-therapy versus rituximab immunotherapy for patients with relapsed or refractory low-grade, follicular, or transformed B-cell non-Hodgkin's lymphoma. J Clin Oncol. 20:2453–63. DOI: 10.1200/JCO.2002.11.076. PMID: 12011122.

58. Gordon LI, Witzig T, Molina A, et al. 2004; Yttrium 90-labeled ibritumomab tiuxetan radioimmunotherapy produces high response rates and durable remissions in patients with previously treated B-cell lymphoma. Clin Lymphoma. 5:98–101. DOI: 10.3816/CLM.2004.n.015. PMID: 15453924.

59. Schouten HC, Qian W, Kvaloy S, et al. 2003; High-dose therapy improves progression-free survival and survival in relapsed follicular non-Hodgkin's lymphoma: results from the randomized European CUP trial. J Clin Oncol. 21:3918–27. DOI: 10.1200/JCO.2003.10.023. PMID: 14517188.

60. Rohatiner AZ, Nadler L, Davies AJ, et al. 2007; Myeloablative therapy with autologous bone marrow transplantation for follicular lymphoma at the time of second or subsequent remission: long-term follow-up. J Clin Oncol. 25:2554–9. DOI: 10.1200/JCO.2006.09.8327. PMID: 17515573.

61. Evens AM, Vanderplas A, LaCasce AS, et al. 2013; Stem cell transplantation for follicular lymphoma relapsed/refractory after prior rituximab: a comprehensive analysis from the NCCN lymphoma outcomes project. Cancer. 119:3662–71. DOI: 10.1002/cncr.28243. PMID: 23921646.

62. Sureda A, Zhang MJ, Dreger P, et al. 2018; Allogeneic hematopoietic stem cell transplantation for relapsed follicular lymphoma: a combined analysis on behalf of the Lymphoma Working Party of the EBMT and the Lymphoma Committee of the CIBMTR. Cancer. 124:1733–42. DOI: 10.1002/cncr.31264. PMID: 29424927. PMCID: PMC5946312.

63. Jacobson CA, Chavez JC, Sehgal AR, et al. 2022; Axicabtagene ciloleucel in relapsed or refractory indolent non-Hodgkin lymphoma (ZUMA-5): a single-arm, multicentre, phase 2 trial. Lancet Oncol. 23:91–103. DOI: 10.1016/S1470-2045(21)00591-X. PMID: 34895487.

64. Schuster SJ, Svoboda J, Chong EA, et al. 2017; Chimeric antigen receptor T cells in refractory B-cell lymphomas. N Engl J Med. 377:2545–54. DOI: 10.1056/NEJMoa1708566. PMID: 29226764. PMCID: PMC5788566.

65. Abramson JS, Palomba ML, Gordon LI, et al. 2020; Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. Lancet. 396:839–52. DOI: 10.1016/S0140-6736(20)31366-0. PMID: 32888407.

66. Schuster SJ, Tam CS, Borchmann P, et al. 2021; Long-term clinical outcomes of tisagenlecleucel in patients with relapsed or refractory aggressive B-cell lymphomas (JULIET): a multicentre, open- label, single-arm, phase 2 study. Lancet Oncol. 22:1403–15. DOI: 10.1016/S1470-2045(21)00375-2. PMID: 34516954.
67. Zucca E, Bertoni F, Vannata B, Cavalli F. 2014; Emerging role of infectious etiologies in the pathogenesis of marginal zone B-cell lymphomas. Clin Cancer Res. 20:5207–16. DOI: 10.1158/1078-0432.CCR-14-0496. PMID: 25320370.

68. Khalil MO, Morton LM, Devesa SS, et al. 2014; Incidence of marginal zone lymphoma in the United States, 2001-2009 with a focus on primary anatomic site. Br J Haematol. 165:67–77. DOI: 10.1111/bjh.12730. PMID: 24417667. PMCID: PMC3967856.

69. Thieblemont C, Cascione L, Conconi A, et al. 2017; A MALT lymphoma prognostic index. Blood. 130:1409–17. DOI: 10.1182/blood-2017-03-771915. PMID: 28720586.

70. Isaacson PG, Spencer J. 1996; Gastric lymphoma and Helicobacter pylori. Important Adv Oncol. 111–21.
71. Wotherspoon AC. 1998; Gastric lymphoma of mucosa-associated lymphoid tissue and Helicobacter pylori. Annu Rev Med. 49:289–99. DOI: 10.1146/annurev.med.49.1.289. PMID: 9509264.
72. Thieblemont C, Zucca E. 2017; Clinical aspects and therapy of gastrointestinal MALT lymphoma. Best Pract Res Clin Haematol. 30:109–17. DOI: 10.1016/j.beha.2017.01.002. PMID: 28288705.

73. Ruskoné-Fourmestraux A, Fischbach W, Aleman BM, et al. 2011; EGILS consensus report. Gastric extranodal marginal zone B-cell lymphoma of MALT. Gut. 60:747–58. DOI: 10.1136/gut.2010.224949. PMID: 21317175.

74. Fuccio L, Laterza L, Zagari RM, Cennamo V, Grilli D, Bazzoli F. 2008; Treatment of Helicobacter pylori infection. BMJ. 337:a1454. DOI: 10.1136/bmj.a1454. PMID: 18794181.

75. Zullo A, Hassan C, Cristofari F, et al. 2010; Effects of Helicobacter pylori eradication on early stage gastric mucosa-associated lymphoid tissue lymphoma. Clin Gastroenterol Hepatol. 8:105–10. DOI: 10.1016/j.cgh.2009.07.017. PMID: 19631287.

76. Nakamura S, Sugiyama T, Matsumoto T, et al. 2012; Long-term clinical outcome of gastric MALT lymphoma after eradication of Helicobacter pylori: a multicentre cohort follow-up study of 420 patients in Japan. Gut. 61:507–13. DOI: 10.1136/gutjnl-2011-300495. PMID: 21890816.

77. Kim JS, Chung SJ, Choi YS, et al. 2007; Helicobacter pylori eradication for low-grade gastric mucosa-associated lymphoid tissue lymphoma is more successful in inducing remission in distal compared to proximal disease. Br J Cancer. 96:1324–8. DOI: 10.1038/sj.bjc.6603708. PMID: 17406363. PMCID: PMC2360178.

78. Zucca E, Bertoni F. 2016; The spectrum of MALT lymphoma at different sites: biological and therapeutic relevance. Blood. 127:2082–92. DOI: 10.1182/blood-2015-12-624304. PMID: 26989205.

79. Wirth A, Gospodarowicz M, Aleman BM, et al. 2013; Long-term outcome for gastric marginal zone lymphoma treated with radiotherapy: a retrospective, multi-centre, International Extranodal Lymphoma Study Group study. Ann Oncol. 24:1344–51. DOI: 10.1093/annonc/mds623. PMID: 23293112.

80. Pinnix CC, Gunther JR, Milgrom SA, et al. 2019; Outcomes after reduced-dose intensity modulated radiation therapy for gastric mucosa-associated lymphoid tissue (MALT) lymphoma. Int J Radiat Oncol Biol Phys. 104:447–55. DOI: 10.1016/j.ijrobp.2019.02.002. PMID: 30769175. PMCID: PMC6839887.

81. Martinelli G, Laszlo D, Ferreri AJ, et al. 2005; Clinical activity of rituximab in gastric marginal zone non-Hodgkin's lymphoma resistant to or not eligible for anti-Helicobacter pylori therapy. J Clin Oncol. 23:1979–83. DOI: 10.1200/JCO.2005.08.128. PMID: 15668468.
82. Amiot A, Lévy M, Copie-Bergman C, et al. 2014; Rituximab, alkylating agents or combination therapy for gastric mucosa-associated lymphoid tissue lymphoma: a monocentric non-randomised observational study. Aliment Pharmacol Ther. 39:619–28. DOI: 10.1111/apt.12635. PMID: 24467480.

83. Avilés A, Nambo MJ, Neri N, Talavera A, Cleto S. 2005; Mucosa- associated lymphoid tissue (MALT) lymphoma of the stomach: results of a controlled clinical trial. Med Oncol. 22:57–62. DOI: 10.1385/MO:22:1:057. PMID: 12648190.
84. Zucca E, Arcaini L, Buske C, et al. 2020; Marginal zone lymphomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 31:17–29. DOI: 10.1016/j.annonc.2019.10.010. PMID: 31912792.

85. Kiesewetter B, Raderer M. 2013; Antibiotic therapy in nongastro-intestinal MALT lymphoma: a review of the literature. Blood. 122:1350–7. DOI: 10.1182/blood-2013-02-486522. PMID: 23770778.

86. Arcaini L, Vallisa D, Rattotti S, et al. 2014; Antiviral treatment in patients with indolent B-cell lymphomas associated with HCV infection: a study of the Fondazione Italiana Linfomi. Ann Oncol. 25:1404–10. DOI: 10.1093/annonc/mdu166. PMID: 24799461.

87. Goda JS, Gospodarowicz M, Pintilie M, et al. 2010; Long-term outcome in localized extranodal mucosa-associated lymphoid tissue lymphomas treated with radiotherapy. Cancer. 116:3815–24. DOI: 10.1002/cncr.25226. PMID: 20564130.

88. Hoskin PJ, Kirkwood AA, Popova B, et al. 2014; 4 Gy versus 24 Gy radiotherapy for patients with indolent lymphoma (FORT): a randomised phase 3 non-inferiority trial. Lancet Oncol. 15:457–63. DOI: 10.1016/S1470-2045(14)70036-1. PMID: 24572077.

89. Fasola CE, Jones JC, Huang DD, Le QT, Hoppe RT, Donaldson SS. 2013; Low-dose radiation therapy (2 Gy x 2) in the treatment of orbital lymphoma. Int J Radiat Oncol Biol Phys. 86:930–5. DOI: 10.1016/j.ijrobp.2013.04.035. PMID: 23726002. PMCID: PMC7560946.
90. Conconi A, Martinelli G, Thiéblemont C, et al. 2003; Clinical activity of rituximab in extranodal marginal zone B-cell lymphoma of MALT type. Blood. 102:2741–5. DOI: 10.1182/blood-2002-11-3496. PMID: 12842999.

91. Lossos IS, Morgensztern D, Blaya M, Alencar A, Pereira D, Rosenblatt J. 2007; Rituximab for treatment of chemoimmunotherapy naive marginal zone lymphoma. Leuk Lymphoma. 48:1630–2. DOI: 10.1080/10428190701457949. PMID: 17701596.

92. Zucca E, Conconi A, Martinelli G, et al. 2017; Final results of the IELSG-19 randomized trial of mucosa-associated lymphoid tissue lymphoma: improved event-free and progression-free survival with rituximab plus chlorambucil versus either chlorambucil or rituximab monotherapy. J Clin Oncol. 35:1905–12. DOI: 10.1200/JCO.2016.70.6994. PMID: 28355112.

93. Salar A, Domingo-Domenech E, Panizo C, et al. 2017; Long-term results of a phase 2 study of rituximab and bendamustine for mucosa-associated lymphoid tissue lymphoma. Blood. 130:1772–4. DOI: 10.1182/blood-2017-07-795302. PMID: 28801448.

94. Kiesewetter B, Willenbacher E, Willenbacher W, et al. 2017; A phase 2 study of rituximab plus lenalidomide for mucosa-associated lymphoid tissue lymphoma. Blood. 129:383–5. DOI: 10.1182/blood-2016-06-720599. PMID: 27879257.

95. Salar A, Domingo-Domenech E, Estany C, et al. 2009; Combination therapy with rituximab and intravenous or oral fludarabine in the first-line, systemic treatment of patients with extranodal marginal zone B-cell lymphoma of the mucosa-associated lymphoid tissue type. Cancer. 115:5210–7. DOI: 10.1002/cncr.24605. PMID: 19672998.

96. Franco V, Florena AM, Iannitto E. 2003; Splenic marginal zone lymphoma. Blood. 101:2464–72. DOI: 10.1182/blood-2002-07-2216. PMID: 12446449. PMCID: PMC9028718.

97. Matutes E, Oscier D, Montalban C, et al. 2008; Splenic marginal zone lymphoma proposals for a revision of diagnostic, staging and therapeutic criteria. Leukemia. 22:487–95. DOI: 10.1038/sj.leu.2405068. PMID: 18094718.

98. Thieblemont C, Felman P, Berger F, et al. 2002; Treatment of splenic marginal zone B-cell lymphoma: an analysis of 81 patients. Clin Lymphoma. 3:41–7. DOI: 10.3816/CLM.2002.n.010. PMID: 12141954.

99. Iannitto E, Ambrosetti A, Ammatuna E, et al. 2004; Splenic marginal zone lymphoma with or without villous lymphocytes. Hematologic findings and outcomes in a series of 57 patients. Cancer. 101:2050–7. DOI: 10.1002/cncr.20596. PMID: 15389479.

100. Rattotti S, Ferretti VV, Rusconi C, et al. 2019; Lymphomas associated with chronic hepatitis C virus infection: a prospective multicenter cohort study from the Rete Ematologica Lombarda (REL) clinical network. Hematol Oncol. 37:160–7. DOI: 10.1002/hon.2575. PMID: 30726562.

101. Arcaini L, Besson C, Frigeni M, et al. 2016; Interferon-free antiviral treatment in B-cell lymphoproliferative disorders associated with hepatitis C virus infection. Blood. 128:2527–32. DOI: 10.1182/blood-2016-05-714667. PMID: 27605512.

102. Hermine O, Lefrere F, Bronowicki JP, et al. 2002; Regression of splenic lymphoma with villous lymphocytes after treatment of hepatitis C virus infection. N Engl J Med. 347:89–94. DOI: 10.1056/NEJMoa013376. PMID: 12110736.

103. Bennett M, Sharma K, Yegena S, Gavish I, Dave HP, Schechter GP. 2005; Rituximab monotherapy for splenic marginal zone lymphoma. Haematologica. 90:856–8. DOI: 10.1590/s1516-31802010000600012. PMID: 15951303.
104. Tsimberidou AM, Catovsky D, Schlette E, et al. 2006; Outcomes in patients with splenic marginal zone lymphoma and marginal zone lymphoma treated with rituximab with or without chemotherapy or chemotherapy alone. Cancer. 107:125–35. DOI: 10.1002/cncr.21931. PMID: 16700034.

105. Else M, Marin-Niebla A, de la Cruz F, et al. 2012; Rituximab, used alone or in combination, is superior to other treatment modalities in splenic marginal zone lymphoma. Br J Haematol. 159:322–8. DOI: 10.1111/bjh.12036. PMID: 23016878.

106. Olszewski AJ, Ali S. 2014; Comparative outcomes of rituximab-based systemic therapy and splenectomy in splenic marginal zone lymphoma. Ann Hematol. 93:449–58. DOI: 10.1007/s00277-013-1900-4. PMID: 24057925.

107. Kalpadakis C, Pangalis GA, Sachanas S, et al. 2018; Rituximab monotherapy in splenic marginal zone lymphoma: prolonged responses and potential benefit from maintenance. Blood. 132:666–70. DOI: 10.1182/blood-2018-02-833608. PMID: 29914978.

108. Kalpadakis C, Pangalis GA, Dimopoulou MN, et al. 2007; Rituximab monotherapy is highly effective in splenic marginal zone lymphoma. Hematol Oncol. 25:127–31. DOI: 10.1002/hon.820. PMID: 17514771.

Table 1
Indications for treatment in low grade lymphoma.
| Indication | Detail |
|---|---|
| High tumor burden [10] | Any site >7 cm |
| Three or more sites >3 cm | |
| Splenomegaly (>16 cm) | |
| Pleural or peritoneal effusion | |
| Circulating tumor cells >5,000/mL | |
| Cytopenia secondary to lymphoma | |
| - Absolute neutrophil count <1,000/mL | |
| - Platelet count <100,000/mL | |
| Disease-related symptoms | Fever |
| Night sweats | |
| Weight loss | |
| Compression | |
| Other lymphoma-related symptoms | |
| Steady progression | Over at least 6 months |
Table 2
Treatment of follicular lymphoma.
| Disease status | Treatment | Comment |
|---|---|---|
| Localized disease | RT | - Potentially curative (ISRT 24–30Gy) |
| - The addition of systemic therapy to RT improves PFS but not OS | ||
| Rituximab | - Radiotherapy ineligible patients | |
| CIT | - Non-contiguous, bulky disease | |
| Watch and wait | - Stable, asymptomatic patients | |
| Advanced disease | Watch and wait | - Without treatment indications (Table 1) |
| CIT± antibody maintenance | - Rituximab or obinutuzumab+(CHOP, CVP, Bebdamustine) | |
| - Maintenance improves PFS but not OS | ||
|
Rituximab Lenalidomide+rituximab |
- For low tumor burden | |
| - As effective as chemoimmunotherapy | ||
| Relapsed disease | Watch and wait | - Stable, asymptomatic patients |
| Palliative RT | - 2×2Gy | |
| CIT± antibody maintenance | - Long previous remission with CIT | |
| - Non-resistant regimen | ||
| Rituximab | - For low tumor burden | |
| Lenalidomide+rituximab | - POD≤24 months after CIT | |
| PI3K inhibitors | - Double refractory disease | |
| EZH2 inhibitor (tazemetostat) | - EZH2 mutation-positive disease | |
| Radioimmunotherapy | - Not widely used | |
| Auto/allo-HSCT | - In selected patients | |
| CAR-T cell therapy | - After ≥2 lines of systemic therapy [63] |
Abbreviations: CAR-T, chimeric antigen receptor T-cell; CIT, chemoimmunotherapy; CR, complete response; EZH2, enhancer of zeste homolog 2; HSCT, hematopoietic stem cell transplantation; ISRT, involved site RT; ORR, overall response rate; OS, overall survival; PFS, progression-free survival; PI3K, phosphatidylinositol 3-kinase; POD, progression of disease; RT, radiotherapy.
Table 3
Treatment of marginal zone lymphoma.
| Disease | Treatment | Comment |
|---|---|---|
| Gastric ENMZL | H. pylori eradication | - PPI+clarithromycin+(amoxicillin or metronidazole) |
| RT | - H.pylori(-), or eradication failure | |
| Rituximab | - For radiotherapy ineligible patients | |
| Gastrectomy | - Major gastric bleeding | |
| Non-gastric ENMZL | Watch and wait | - Stable asymptomatic disease |
| Targeting infectious agents | - HCV treatment for HCV(+) disease | |
| - Doxycycline for ocular adnexal ENMZL | ||
| RT | - Definitive or palliative | |
| Rituximab | - Higher response in CTx-naïve patients | |
| CIT | - R-chlorambucil, R-bendamustine | |
| Lenalidomide+Rituximab | - To avoid chemotherapy | |
| Surgery | - Mostly for diagnosis (thyroid, breast, intestine, etc.) | |
| Splenic MZL | Watch and wait | - Stable asymptomatic disease |
| HCV eradication | - For HCV(+) disease | |
| Rituximab | - Offer the most risk/benefit ratio [106] | |
| Splenectomy | - After rituximab failure | |
| CIT | - For symptomatic disseminated disease after rituximab or splenectomy failure | |
| Nodal MZL | Treated as guidelines for FL | - Studies enrolled solely MZL are rare |




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download