Abstract
Background
Although decades have passed since the introduction of pharmacologically induced hypertensive therapy (PIHT) against early neurologic deterioration (END) in acute ischemic stroke, the optimal duration of PIHT remains elusive.
Case Report
A 70-year-old man developed right hemiplegia and aphasia 25 hours before arrival. Computed tomography angiography (CTA) revealed acute infarction in the left middle cerebral artery (MCA) territory and occlusion of the left internal carotid artery. He experienced END 36 hours after admission, and CTA revealed a newly developed proximal MCA occlusion. PIHT was initiated to augment cerebral perfusion. As his neurologic symptoms were highly dependent on blood pressure, PIHT was inevitably sustained for over 3 weeks. Follow-up CTA revealed recanalization of the MCA.
Early neurologic deterioration (END) is a common complication occurring in up to one-third of patients with acute ischemic stroke [1]. Proximal arterial occlusion is a risk factor for predicting END [2]. Pharmacologically induced hypertensive therapy (PIHT) is a therapeutic option for END, which acts by increasing collateral flow, thereby preventing the expansion of the infarct core [3,4]. Phenylephrine is mainly used for PIHT because it selectively binds to alpha-1 receptors and causes peripheral vasoconstriction without substantial direct cerebral vasoconstriction to increase blood pressure (BP) [5]. However, PIHT is not always safe, as it may lead to adverse effects such as hemorrhagic transformation, cerebral edema, and ischemic heart diseases [4]. For this reason, PIHT is generally performed within 3–5 days [4,6]; however, such recommendations are merely based on theoretical arguments and case reports. Herein, we report a patient with acute internal carotid artery (ICA) terminus occlusion who was successfully treated with prolonged PIHT over 3 weeks, which far exceeds the conventional safe window for PIHT.
A 70-year-old man with a history of hypertension and dyslipidemia presented with aphasia and weakness of the right arm (11 hours from the last known normal) was referred to our hospital 14 hours after the detection of symptoms by a regional hospital. Upon neurological examination, he showed motor aphasia, dysarthria, and right arm weakness (initial National Institutes of Health Stroke Scale [NIHSS] score, 7). Brain computed tomography (CT) revealed a hypodense lesion in the superior division of the left middle cerebral artery (MCA) territory, and CT angiography (CTA) revealed occlusion of the left ICA with well-developed collateral circulation from the anterior communicating artery (Fig. 1A and B). Although perfusion mismatch was observed on perfusion CT processed by the RAPID software (iSchemaView, Menlo Park, CA, USA) (cerebral blood flow [CBF] <30%, volume: 23 mL; Tmax >6.0 seconds, volume: 101 mL) (Fig. 1C), endovascular therapy (EVT) was not performed as 25 hours had passed since the last known normal beyond the extended time frame for EVT. Brain magnetic resonance imaging (MRI) demonstrated acute lesions in the MCA superior division territory on diffusion-weighted images (DWI) with hyperintensity on fluid-attenuated inversion recovery (FLAIR) images (Fig. 1D and E). He was prescribed dual antiplatelet (aspirin and clopidogrel) and high-dose statin therapy, and his neurological symptoms improved thereafter (NIHSS score, 5).
However, the patient developed abrupt neurologic deterioration 33 hours after admission (BP, 117/60 mmHg) with dense right hemiplegia (motor grade 2) and global aphasia (NIHSS score, 17). Follow-up CTA revealed a newly developed left proximal MCA occlusion (Fig. 2A). Despite the definite perfusion mismatch (CBF <30%, volume: 26 mL; Tmax >6.0 seconds, volume: 175 mL) (Fig. 2C), EVT was not considered because of concerns about the possibility that the device could not access the target vessel due to left proximal ICA occlusion and the risk of hemorrhagic transformation after recanalization. Instead, triplet antiplatelet therapy (aspirin, clopidogrel, and cilostazol) and PIHT were initiated to raise the systolic BP above 140 mmHg, and he showed significant improvement (NIHSS score, 11) 10 hours after administering phenylephrine (BP, 145/78 mmHg). Initially, phenylephrine (0.1 mg/mL) was intravenously infused at a rate of 10 mL/hr (1 mg/hr, 0.28 μg/kg/min) and the dose was increased up to a rate of 35 mL/hr (3.5 mg/hr, 0.97 μg/kg/min). Brain MRI demonstrated a newly developed acute lesion in the basal ganglia and corona radiata (Fig. 2D). As the symptoms plateaued for 10 days after PIHT, the dose of phenylephrine was gradually tapered. However, phenylephrine was not discontinued because his neurologic symptoms were highly dependent on BP and showed abrupt deterioration (aphasia and hemiplegia) when phenylephrine was tapered even at a minimal rate (5 mL/hr). Thereafter, phenylephrine was administered at 35 mL/hr (3.5 mg/hr, 0.97 μg/kg/min) for 4 more days, following which the infusion rate was lowered by 5 mL/hr per day for 7 days, and phenylephrine was discontinued 21 days after the start of PIHT. The patient did not show neurologic deterioration upon dose reduction, and the therapy was discontinued 24 days after the onset of END. No abnormalities in cardiac enzymes, electrocardiogram, or chest radiography were noted during this period. Unexpectedly, follow-up perfusion CTA revealed recanalization of the MCA occlusion and improved perfusion status (Tmax >6.0 seconds, volume: 0 mL) (Fig. 3A-C). There were no further ischemic lesions on follow-up DWI or FLAIR images (Fig. 3D and E). The patient was discharged 31 days after symptom onset, with significantly improved neurological symptoms (NIHSS score, 6). He did not experience any recurrent stroke symptoms until 18 months after discharge.
Although the definition of END varies, most studies have been based on a worsening NIHSS score of 4 or more within 24–72 hours after the onset of acute ischemic stroke [2]. Symptomatic intracranial hemorrhage and malignant vasogenic edema are known to be the main causes of END; however, the exact mechanism of END is unknown in approximately half of the cases. If a clear mechanism cannot be identified, it is possible that the patient’s prognosis may deteriorate because appropriate treatment cannot be provided. It is hypothesized that the extension of symptomatic ischemic tissue is the mechanism by which END occurs without a clear reason [7]. In a study of minor stroke patients with proximal occlusion, it was determined that END may occur [8] because there is a high risk of ischemic penumbra enlargement in the presence of a proximal occlusion, followed by an increase in infarction volume. Since our patient had an NIHSS score of 7 with ICA occlusion, it would have been an indication for EVT if the patient had visited within the EVT time frame. However, our patient was transferred from another hospital, thereby going beyond the time frame for EVT. The best medical management was started because EVT was not performed; however, the patient’s neurological deficit was presumed to have worsened due to proximal occlusion. PIHT was performed as a treatment for END.
Managing patients with END is challenging in daily clinical practice. The clinical situation is complicated; therefore, a substantial proportion of patients are not subjected to treatment supported by evidence derived from randomized clinical trials, as in our case. The authors admit that our aggressive therapeutic approaches (i.e., triple antiplatelet therapy and extended use of phenylephrine) are not supported by current stroke treatment guidelines. Given that the cerebral perfusion of the patient in the left MCA territory was mainly supplied via collateral circulation from the opposite side through the anterior communicating artery, newly developed proximal MCA occlusion could have led to extensive infarction in the left MCA territory. To prevent the ischemic penumbra from expanding, the authors believed that PIHT would serve as a viable option for augmenting cerebral perfusion to the ischemic penumbra. Whether sustaining PIHT for an extended period that far exceeds the previously recommended safe window is indeed an optimal decision may be controversial. However, prolonged PIHT beyond the conventional safe window was inevitable as his symptoms were highly dependent on the BP; therefore, PIHT was sustained until he became neurologically stable regardless of the BP.
It has been recently reported that rescue mechanical thrombectomy improves the prognosis of acute neurological deterioration [9]. However, in that study, only three patients had neurological deterioration 24 hours after admission. As such, rescue thrombectomy could have been considered in our case; however, this was not supported by the treatment guidelines.
The recanalization of the MCA occlusion in our case was unexpected. Spontaneous recanalization of the occluded MCA has been reported to occur infrequently [10]. It is known to occur mainly in MCA occlusions due to cardiogenic embolism or dissection. Since digital subtraction angiography was not performed in our case, there is a limitation that we were unable to accurately determine the location of the initially occluded blood vessel. When neurologic deterioration occurs, the mechanism of MCA occlusion can be estimated as thrombus propagation or artery-to-artery embolism. According to a previous study [11], the location of the occluded blood vessel is likely to be the cavernous segment of the ICA, considering that the cervical occlusion was initially shaped like a spearhead and some flow of the cavernous portion was observed (Fig. 1). Although the mechanisms of thrombus propagation are not well known, it is known that stenosis or irregular vessel geometries affect blood flow velocity, which influences the function of the von Willebrand factor to cause thrombus formation and propagation [12].
In our case, as the BP dropped and collateral flow slowed, it was assumed that the thrombus, which was initially located in the cavernous segment, was propagated to induce occlusion of the proximal MCA. Considering that the shape of the cervical occlusion was transformed into a stump on CTA performed after neurological deterioration (Fig. 2), it is possible that a new thrombus was formed in the proximal ICA at this point, which flowed forward, thereby occluding the proximal MCA. Both mechanisms were based on atherosclerosis, and no cardioembolic source was identified in our patient. The mechanism of spontaneous recanalization of atherosclerotic occlusion is not clearly understood. Various mechanisms have been proposed to explain this mechanism [13]. Collateral circulation may facilitate fibrinolysis while compensating for the blood supply. Antiplatelet agents and statins inhibit platelet aggregation, reverse the formation of atherosclerotic plaques, and inhibit the focal inflammatory response, which may help establish spontaneous recanalization. Therefore, the authors’ use of the three antiplatelet agents was possibly helpful. We surmise that the occluded vessel was recanalized spontaneously, and at the same time, prolonged PIHT played a role by sustaining the collateral flow, thereby preventing the penumbra from converting into the infarction core, stalling until delayed recanalization occurred.
In our case, if the patient's symptoms worsened despite PIHT, additional perfusion CT should be performed to evaluate the change in mismatch volume. If the worsening of cerebral perfusion was confirmed as a mismatch volume, bypass surgery would be the only treatment option since it was too late to perform an intra-arterial procedure. Fortunately, PIHT prevented further neurological deterioration, and this case highlights that prolonged PIHT may serve as a viable therapeutic option for managing END in patients ineligible for EVT. This report provides evidence that prolonged PIHT may be beneficial in certain cases and that the duration may be modified according to the neurological status unless the patient shows adverse effects on PIHT. Although our findings may not be completely generalizable to standard clinical practice, this case has clinical implications for managing patients who experience END without alternative treatment options.
Notes
REFERENCES
1. Siegler JE, Martin-Schild S. Early neurological deterioration (END) after stroke: the END depends on the definition. Int J Stroke. 2011; 6:211–2.

2. Seners P, Turc G, Oppenheim C, Baron JC. Incidence, causes and predictors of neurological deterioration occurring within 24 h following acute ischaemic stroke: a systematic review with pathophysiological implications. J Neurol Neurosurg Psychiatry. 2015; 86:87–94.

3. Bang OY, Chung JW, Kim SK, Kim SJ, Lee MJ, Hwang J, et al. Therapeutic-induced hypertension in patients with noncardioembolic acute stroke. Neurology. 2019; 93:e1955–63.

4. Koenig MA, Geocadin RG, de Grouchy M, Glasgow J, Vimal S, Restrepo L, et al. Safety of induced hypertension therapy in patients with acute ischemic stroke. Neurocrit Care. 2006; 4:3–7.

5. Bevan JA, Duckworth J, Laher I, Oriowo MA, McPherson GA, Bevan RD. Sympathetic control of cerebral arteries: specialization in receptor type, reserve, affinity, and distribution. FASEB J. 1987; 1:193–8.

6. Lee MH, Kim JG, Jeon SB, Kang DW, Kwon SU, Kim JS. Pharmacologically induced hypertension therapy for acute stroke patients. J Stroke. 2019; 21:228–30.

7. Alawneh JA, Moustafa RR, Baron JC. Hemodynamic factors and perfusion abnormalities in early neurological deterioration. Stroke. 2009; 40:e443–50.

8. Kim JT, Park MS, Chang J, Lee JS, Choi KH, Cho KH. Proximal arterial occlusion in acute ischemic stroke with low NIHSS scores should not be considered as mild stroke. PLoS One. 2013; 8:e70996.

9. Saleem Y, Nogueira RG, Rodrigues GM, Kim S, Sharashidze V, Frankel M, et al. Acute neurological deterioration in large vessel occlusions and mild symptoms managed medically. Stroke. 2020; 51:1428–34.

10. Rha JH, Saver JL. The impact of recanalization on ischemic stroke outcome: a meta-analysis. Stroke. 2007; 38:967–73.

11. Hong JM, Lee SE, Lee SJ, Lee JS, Demchuk AM. Distinctive patterns on CT angiography characterize acute internal carotid artery occlusion subtypes. Medicine (Baltimore). 2017; 96:e5722.

12. Asada Y, Yamashita A, Sato Y, Hatakeyama K. Thrombus formation and propagation in the onset of cardiovascular events. J Atheroscler Thromb. 2018; 25:653–64.

13. Mao Y, Huang Y, Zhang L, Nan G. Spontaneous recanalization of atherosclerotic middle cerebral artery occlusion: case report. Medicine (Baltimore). 2017; 96:e7372.
Fig. 1.
Brain perfusion computed tomography angiography (CTA) and magnetic resonance imaging at admission. (A) Brain CTA revealed collateral flow via the anterior communicating artery to the middle cerebral artery (MCA) and (B) left proximal internal carotid artery occlusion with calcification (arrow, spearhead shape). (C) Perfusion mismatch was confirmed by the RAPID software (iSchemaView, Menlo Park, CA, USA) (cerebral blood flow [CBF] <30%, volume: 23 mL; Tmax >6.0 seconds, volume: 101 mL). (D) Brain MRI revealed increased signals on the diffusion-weighted imaging sequence in the left MCA superior division territory (red circles) and (E) on the fluid-attenuated inversion recovery sequence at the same lesion.
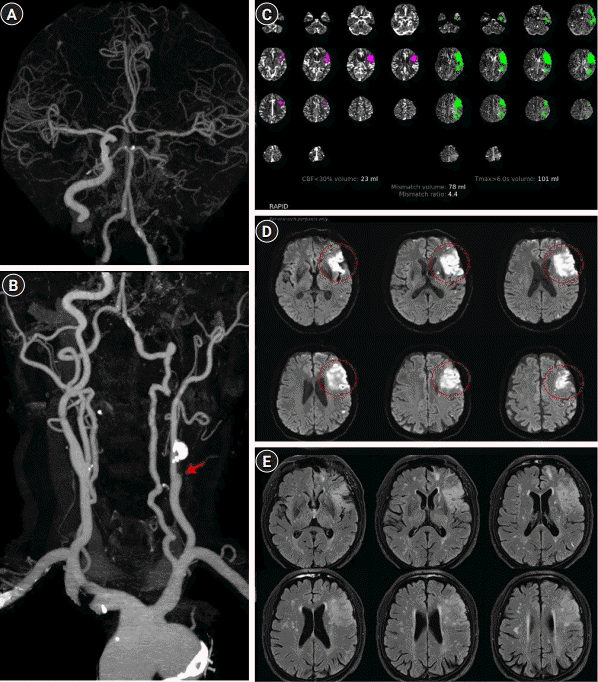
Fig. 2.
Brain perfusion computed tomography angiography (CTA) and magnetic resonance imaging after early neurologic deterioration. (A) Brain CTA revealed left proximal middle cerebral artery occlusion and (B) changes in the shape of the internal carotid artery occlusion (arrow, stump shape). (C) Perfusion mismatch identified using the RAPID software (iSchemaView, Menlo Park, CA, USA) was further expanded (cerebral blood flow [CBF] <30%, volume: 26 mL; Tmax >6.0 seconds, volume: 145 mL). (D) Diffusion-weighted imaging showed a newly developed acute lesion in the basal ganglia and corona radiata.
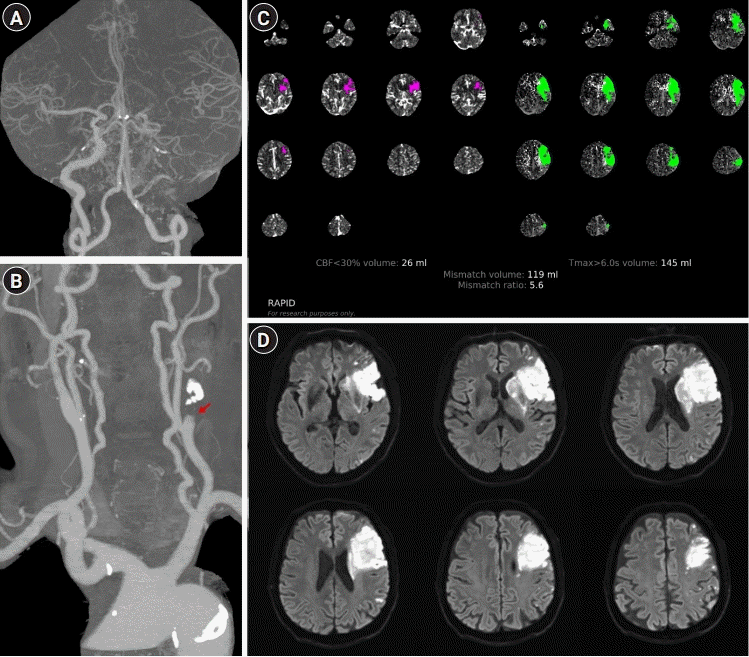
Fig. 3.
Follow-up brain perfusion computed tomography angiography (CTA) and magnetic resonance imaging (MRI) were performed on day 24 after early neurologic deterioration. (A) Brain CTA showed spontaneous recanalization of the left middle cerebral artery and (B) no interval change of the internal carotid artery occlusion. (C) Perfusion mismatch was no longer observed. (D, E) No new lesions were identified on diffusion-weighted imaging or fluid-attenuated inversion recovery. CBF, cerebral blood flow.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download