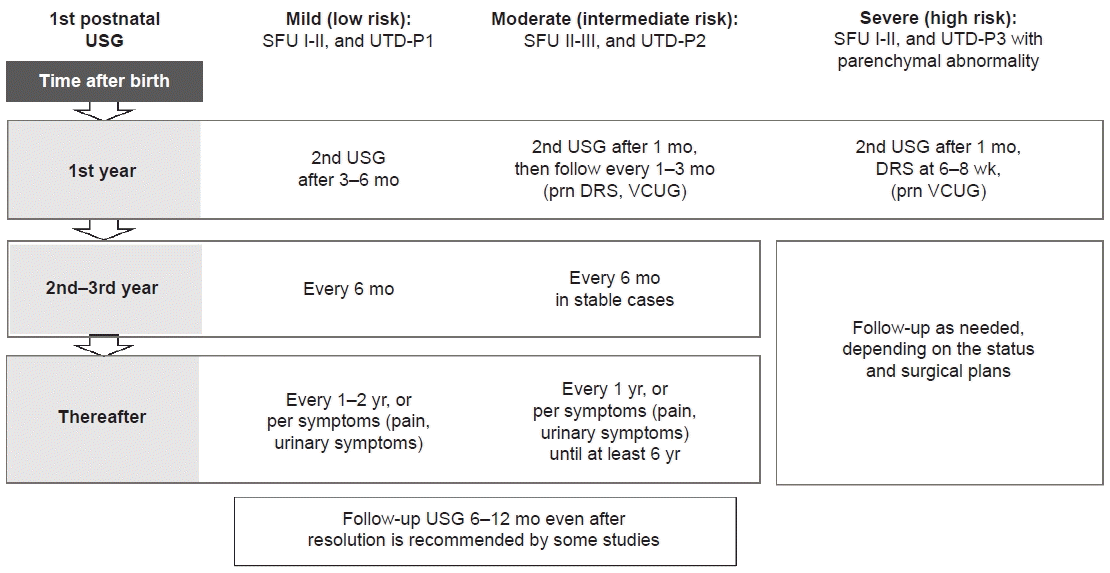
1. Nguyen HT, Herndon CD, Cooper C, Gatti J, Kirsch A, Kokorowski P, et al. The Society for Fetal Urology consensus statement on the evaluation and management of antenatal hydronephrosis. J Pediatr Urol. 2010; 6:212–31.

2. Choi HA, Lee DJ, Shin SM, Lee YK, Ko SY, Park SW. The prenatal and postnatal incidence of congenital anomalies of the kidneys and urinary tract (CAKUT) detected by ultrasound. Child Kidney Dis. 2016; 20:29–32.

3. Corbett HJ, McCarthy L. Hydronephrosis in children: pelviureteric junction dysfunction. Surgery (Oxford). 2013; 31:135–9.

4. Kim MY, Im YJ, Hyun HS, Kang HG, Ha IS, Cheong HI, et al. A case of an ureteropelvic junction obstruction caused by a crossing vessel. Child Kidney Dis. 2018; 22:28–31.

5. Davenport MT, Merguerian PA, Koyle M. Antenatally diagnosed hydronephrosis: current postnatal management. Pediatr Surg Int. 2013; 29:207–14.

6. Sidhu G, Beyene J, Rosenblum ND. Outcome of isolated antenatal hydronephrosis: a systematic review and meta-analysis. Pediatr Nephrol. 2006; 21:218–24.

7. Sarhan OM, El Helaly A, Al Otay AH, Al Ghanbar M, Nakshabandi Z. Prenatally detected, unilateral, high-grade hydronephrosis: can we predict the natural history? Can Urol Assoc J. 2018; 12:E137–41.

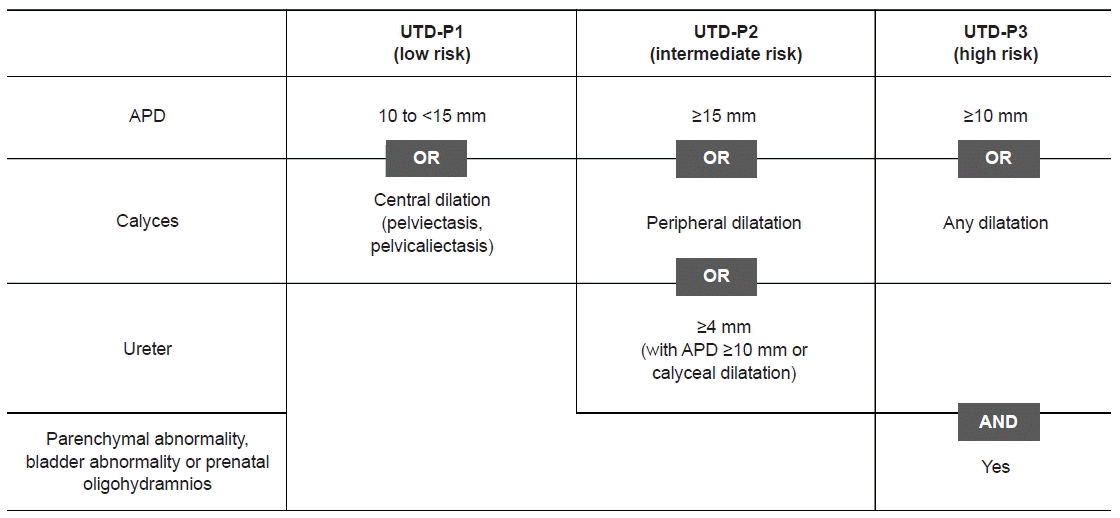
8. Nguyen HT, Benson CB, Bromley B, Campbell JB, Chow J, Coleman B, et al. Multidisciplinary consensus on the classification of prenatal and postnatal urinary tract dilation (UTD classification system). J Pediatr Urol. 2014; 10:982–98.

9. Chalmers DJ, Meyers ML, Brodie KE, Palmer C, Campbell JB. Inter-rater reliability of the APD, SFU and UTD grading systems in fetal sonography and MRI. J Pediatr Urol. 2016; 12:305.

10. Zanetta VC, Rosman BM, Bromley B, Shipp TD, Chow JS, Campbell JB, et al. Variations in management of mild prenatal hydronephrosis among maternal-fetal medicine obstetricians, and pediatric urologists and radiologists. J Urol. 2012; 188:1935–9.

11. Onen A. Grading of hydronephrosis: an ongoing challenge. Front Pediatr. 2020; 8:458.

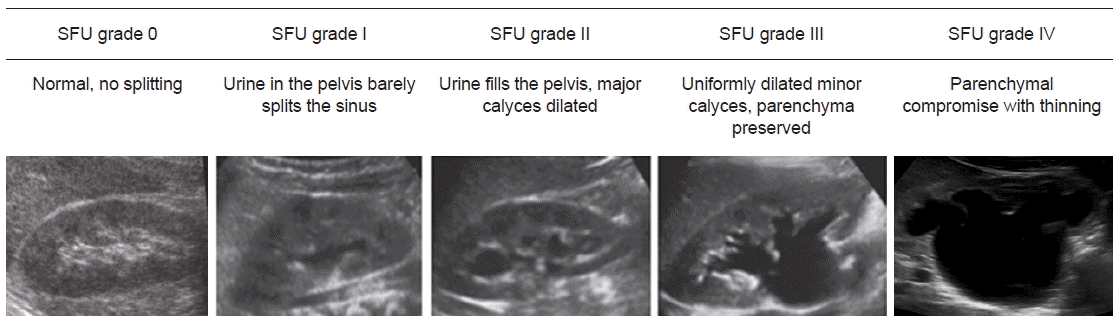
12. Fernbach SK, Maizels M, Conway JJ. Ultrasound grading of hydronephrosis: introduction to the system used by the Society for Fetal Urology. Pediatr Radiol. 1993; 23:478–80.

13. Elmaci AM, Donmez MI. Time to resolution of isolated antenatal hydronephrosis with anteroposterior diameter ≤ 20 mm. Eur J Pediatr. 2019; 178:823–8.

14. Timberlake MD, Herndon CD. Mild to moderate postnatal hydronephrosis: grading systems and management. Nat Rev Urol. 2013; 10:649–56.

15. Jung J, Lee JH, Kim KS, Park YS. Utility of Society for Fetal Urology and anteroposterior pelvic diameter grading systems for estimating time to resolution of isolated hydronephrosis: a single center study. J Urol. 2020; 204:1048–53.

16. Nguyen HT, Phelps A, Coley B, Darge K, Rhee A, Chow JS. 2021 update on the urinary tract dilation (UTD) classification system: clarifications, review of the literature, and practical suggestions. Pediatr Radiol. 2022; 52:740–51.

17. Gray MC, Zillioux JM, Varda B, Herndon CD, Kurtz MP, Chow JS, et al. Assessment of urinary tract dilation grading amongst pediatric urologists. J Pediatr Urol. 2020; 16:457.

18. Braga LH, McGrath M, Farrokhyar F, Jegatheeswaran K, Lorenzo AJ. Associations of initial Society for Fetal Urology grades and urinary tract dilatation risk groups with clinical outcomes in patients with isolated prenatal hydronephrosis. J Urol. 2017; 197(3 Pt 2):831–7.

19. Braga LH, McGrath M, Farrokhyar F, Jegatheeswaran K, Lorenzo AJ. Society for Fetal Urology classification vs urinary tract dilation grading system for prognostication in prenatal hydronephrosis: a time to resolution analysis. J Urol. 2018; 199:1615–21.

20. Cakici EK, Aydog O, Eroglu FK, Yazilitas F, Ozlu SG, Uner C, et al. Value of renal pelvic diameter and urinary tract dilation classification in the prediction of urinary tract anomaly. Pediatr Int. 2019; 61:271–7.

21. Hodhod A, Capolicchio JP, Jednak R, El-Sherif E, El-Doray Ael-A, El-Sherbiny M. Evaluation of urinary tract dilation classification system for grading postnatal hydronephrosis. J Urol. 2016; 195:725–30.

22. Han M, Kim HG, Lee JD, Park SY, Sur YK. Conversion and reliability of two urological grading systems in infants: the Society for Fetal Urology and the urinary tract dilatation classifications system. Pediatr Radiol. 2017; 47:65–73.

23. Rickard M, Easterbrook B, Kim S, Farrokhyar F, Stein N, Arora S, et al. Six of one, half a dozen of the other: a measure of multidisciplinary inter/intra-rater reliability of the Society for Fetal Urology and urinary tract dilation grading systems for hydronephrosis. J Pediatr Urol. 2017; 13:80.
24. Kaspar CD, Lo M, Bunchman TE, Xiao N. The antenatal urinary tract dilation classification system accurately predicts severity of kidney and urinary tract abnormalities. J Pediatr Urol. 2017; 13:485.

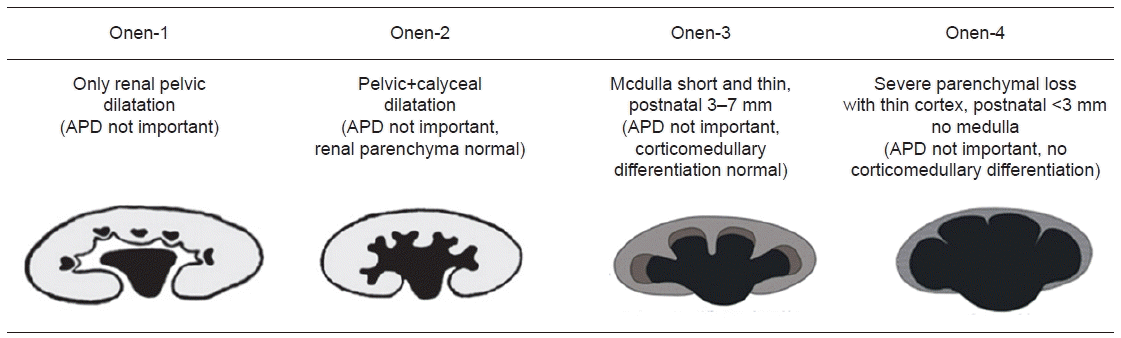
25. Onen A. An alternative grading system to refine the criteria for severity of hydronephrosis and optimal treatment guidelines in neonates with primary UPJ-type hydronephrosis. J Pediatr Urol. 2007; 3:200–5.

26. Cho HY, Jung I, Kim YH, Kwon JY. Reliability of Society of Fetal Urology and Onen grading system in fetal hydronephrosis. Obstet Gynecol Sci. 2019; 62:87–92.

27. Kim SY, Kim MJ, Yoon CS, Lee MS, Han KH, Lee MJ. Comparison of the reliability of two hydronephrosis grading systems: the Society for Foetal Urology grading system vs. the Onen grading system. Clin Radiol. 2013; 68:e484–90.

28. Swenson DW, Darge K, Ziniel SI, Chow JS. Characterizing upper urinary tract dilation on ultrasound: a survey of North American pediatric radiologists’ practices. Pediatr Radiol. 2015; 45:686–94.

29. Suson KD, Preece J. Do current scientific reports of hydronephrosis make the grade? J Pediatr Urol. 2020; 16:597.

30. Aksu N, Yavascan O, Kangin M, Kara OD, Aydin Y, Erdogan H, et al. Postnatal management of infants with antenatally detected hydronephrosis. Pediatr Nephrol. 2005; 20:1253–9.

31. Yalcinkaya F, Ozcakar ZB. Management of antenatal hydronephrosis. Pediatr Nephrol. 2020; 35:2231–9.

32. Djahangirian O, Young I, Dorgalli C, Bissaillon A, Tran NA, Walia A, et al. Safe discharge parameters for patients with isolated antenatal hydronephrosis. J Pediatr Urol. 2018; 14:321.

33. Kohata E, Kimata T, Onuma C, Yamanouchi S, Tsuji S, Ohashi A, et al. Natural course of isolated mild congenital hydronephrosis: a 2-year prospective study at a single center in Japan. Int J Urol. 2019; 26:643–7.

34. Dias CS, Silva JM, Pereira AK, Marino VS, Silva LA, Coelho AM, et al. Diagnostic accuracy of renal pelvic dilatation for detecting surgically managed ureteropelvic junction obstruction. J Urol. 2013; 190:661–6.

35. Gatti JM, Broecker BH, Scherz HC, Perez-Brayfield MR, Kirsch AJ. Antenatal hydronephrosis with postnatal resolution: how long are postnatal studies warranted? Urology. 2001; 57:1178.

36. Matsui F, Shimada K, Matsumoto F, Takano S. Late recurrence of symptomatic hydronephrosis in patients with prenatally detected hydronephrosis and spontaneous improvement. J Urol. 2008; 180:322–5.

37. Coelho GM, Bouzada MC, Pereira AK, Figueiredo BF, Leite MR, Oliveira DS, et al. Outcome of isolated antenatal hydronephrosis: a prospective cohort study. Pediatr Nephrol. 2007; 22:1727–34.

38. Riccabona M, Avni FE, Blickman JG, Dacher JN, Darge K, Lobo ML, et al. Imaging recommendations in paediatric uroradiology: minutes of the ESPR workgroup session on urinary tract infection, fetal hydronephrosis, urinary tract ultrasonography and voiding cystourethrography, Barcelona, Spain, June 2007. Pediatr Radiol. 2008; 38:138–45.

39. Duong HP, Piepsz A, Collier F, Khelif K, Christophe C, Cassart M, et al. Predicting the clinical outcome of antenatally detected unilateral pelviureteric junction stenosis. Urology. 2013; 82:691–6.

40. Thomas DF. Prenatal diagnosis: what do we know of long-term outcomes? J Pediatr Urol. 2010; 6:204–11.

41. Madden-Fuentes RJ, McNamara ER, Nseyo U, Wiener JS, Routh JC, Ross SS. Resolution rate of isolated low-grade hydronephrosis diagnosed within the first year of life. J Pediatr Urol. 2014; 10:639–44.

42. Sarhan OM, Helaly AE, Al Otay A, Ghanbar MA, Nakshabandi Z. Isolated low grade prenatally detected unilateral hydronephrosis: do we need long term follow-up? Int Braz J Urol. 2018; 44:812–8.

43. Thornburg LL, Pressman EK, Chelamkuri S, Hulbert W, Rabinowitz R, Mevorach R. Third trimester ultrasound of fetal pyelectasis: predictor for postnatal surgery. J Pediatr Urol. 2008; 4:51–4.

44. Mesrobian HG, Mirza SP. Hydronephrosis: a view from the inside. Pediatr Clin North Am. 2012; 59:839–51.
45. Chertin B, Pollack A, Koulikov D, Rabinowitz R, Hain D, Hadas-Halpren I, et al. Conservative treatment of ureteropelvic junction obstruction in children with antenatal diagnosis of hydronephrosis: lessons learned after 16 years of follow-up. Eur Urol. 2006; 49:734–9.

46. Ismaili K, Hall M, Piepsz A, Alexander M, Schulman C, Avni FE. Insights into the pathogenesis and natural history of fetuses with renal pelvis dilatation. Eur Urol. 2005; 48:207–14.

47. Arora S, Yadav P, Kumar M, Singh SK, Sureka SK, Mittal V, et al. Predictors for the need of surgery in antenatally detected hydronephrosis due to UPJ obstruction: a prospective multivariate analysis. J Pediatr Urol. 2015; 11:248.
48. Hodhod A, Capolicchio JP, Jednak R, Eid H, El-Doray AE, El-Sherbiny M. Is the renal pyramidal thickness a good predictor for pyeloplasty in postnatal hydronephrosis? J Pediatr Urol. 2018; 14:277.

49. Rickard M, Lorenzo AJ, Braga LH. Renal parenchyma to hydronephrosis area ratio (PHAR) as a predictor of future surgical intervention for infants with high-grade prenatal hydronephrosis. Urology. 2017; 101:85–9.

50. Killi I, Avlan D, Taskinlar H, Kara PP, Apaydin FD, Delibas A, et al. Effective predictors for surgical decision in antenatal hydronephrosis: a prospective multiparameter analysis. Turk J Urol. 2017; 43:361–5.

51. Lee JN, Kang JK, Jeong SY, Lee SM, Cho MH, Ha YS, et al. Predictive value of cortical transit time on MAG3 for surgery in antenatally detected unilateral hydronephrosis caused by ureteropelvic junction stenosis. J Pediatr Urol. 2018; 14:55.

52. Longpre M, Nguan A, Macneily AE, Afshar K. Prediction of the outcome of antenatally diagnosed hydronephrosis: a multivariable analysis. J Pediatr Urol. 2012; 8:135–9.

53. Harper L, Bourquard D, Grosos C, Abbo O, Ferdynus C, Michel JL, et al. Cortical transit time as a predictive marker of the need for surgery in children with pelvi-ureteric junction stenosis: preliminary study. J Pediatr Urol. 2013; 9(6 Pt B):1054–8.

54. Piepsz A, Tondeur M, Nogarede C, Collier F, Ismaili K, Hall M, et al. Can severely impaired cortical transit predict which children with pelvi-ureteric junction stenosis detected antenatally might benefit from pyeloplasty? Nucl Med Commun. 2011; 32:199–205.

55. Schlotmann A, Clorius JH, Clorius SN. Diuretic renography in hydronephrosis: renal tissue tracer transit predicts functional course and thereby need for surgery. Eur J Nucl Med Mol Imaging. 2009; 36:1665–73.

56. Song SH, Park S, Chae SY, Moon DH, Park S, Kim KS. Predictors of renal functional improvement after pyeloplasty in ureteropelvic junction obstruction: clinical value of visually assessed renal tissue tracer transit in 99mTc-mercaptoacetyltriglycine renography. Urology. 2017; 108:149–54.

57. Kadioglu A. Renal measurements, including length, parenchymal thickness, and medullary pyramid thickness, in healthy children: what are the normative ultrasound values? AJR Am J Roentgenol. 2010; 194:509–15.

58. Rodriguez LV, Lock J, Kennedy WA, Shortliffe LM. Evaluation of sonographic renal parenchymal area in the management of hydronephrosis. J Urol. 2001; 165:548–51.

59. Szymanski KM, Al-Said AN, Pippi Salle JL, Capolicchio JP. Do infants with mild prenatal hydronephrosis benefit from screening for vesicoureteral reflux? J Urol. 2012; 188:576–81.

60. Tombesi MM, Alconcher LF. Short-term outcome of mild isolated antenatal hydronephrosis conservatively managed. J Pediatr Urol. 2012; 8:129–33.

61. Lee JH, Choi HS, Kim JK, Won HS, Kim KS, Moon DH, et al. Nonrefluxing neonatal hydronephrosis and the risk of urinary tract infection. J Urol. 2008; 179:1524–8.

62. Yavascan O, Aksu N, Anil M, Kara OD, Aydin Y, Kangin M, et al. Postnatal assessment of growth, nutrition, and urinary tract infections of infants with antenatally detected hydronephrosis. Int Urol Nephrol. 2010; 42:781–8.

63. Lidefelt KJ, Herthelius M. Antenatal hydronephrosis: infants with minor postnatal dilatation do not need prophylaxis. Pediatr Nephrol. 2008; 23:2021–4.

64. Braga LH, Mijovic H, Farrokhyar F, Pemberton J, DeMaria J, Lorenzo AJ. Antibiotic prophylaxis for urinary tract infections in antenatal hydronephrosis. Pediatrics. 2013; 131:e251–61.

65. Braga LH, Easterbrook B, Jegatheeswaran K, Lorenzo AJ. From research question to conducting a randomized controlled trial on continuous antibiotic prophylaxis in prenatal hydronephrosis: a rational stepwise process. Front Pediatr. 2016; 4:27.

66. Estrada CR, Peters CA, Retik AB, Nguyen HT. Vesicoureteral reflux and urinary tract infection in children with a history of prenatal hydronephrosis: should voiding cystourethrography be performed in cases of postnatally persistent grade II hydronephrosis? J Urol. 2009; 181:801–7.
67. Merguerian PA, Herz D, McQuiston L, Van Bibber M. Variation among pediatric urologists and across 2 continents in antibiotic prophylaxis and evaluation for prenatally detected hydronephrosis: a survey of American and European pediatric urologists. J Urol. 2010; 184(4 Suppl):1710–5.

68. Silay MS, Undre S, Nambiar AK, Dogan HS, Kocvara R, Nijman RJ, et al. Role of antibiotic prophylaxis in antenatal hydronephrosis: a systematic review from the European Association of Urology/European Society for Paediatric Urology Guidelines Panel. J Pediatr Urol. 2017; 13:306–15.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download