Abstract
Various approaches have been attempted in translational moyamoya disease research. One promising material for modeling and treating this disease is vascular progenitor cells, which can be acquired and expanded from patient peripheral blood. These cells may provide a novel experimental model and enable us to obtain insights regarding moyamoya disease pathogenesis. We briefly present the recent accomplishments in regard to the studies of vascular progenitor cells in moyamoya disease.
Moyamoya disease (MMD) is a specific cerebrovascular disease affecting the termini of the internal carotid arteries, typically bilaterally9). Thus, a natural approach is to investigate the vessel wall cells of MMD patients in search of significant findings that are relevant to MMD pathophysiology and treatment. We propose that this research interest must be driven in two directions, endothelial-lineage cells and smooth muscle-type (mural) cells, as both cells are indispensable to completely healthy vasculature. In reality, it is not easy to obtain these cells directly from patients, except for tiny specimens that are obtained during direct bypass surgery and tissues from autopsy cases. Therefore, vascular progenitor cells seem to be a valuable cell type for disease research because they can be derived from patient peripheral blood.
Endothelial progenitor cells (EPCs) have been an important topic in research evaluating several vascular diseases, including heart disease and ischemic cerebrovascular disease; however, the concept of smooth muscle progenitor cells (SPCs) has only recently been introduced. In this review, we present the up-to-date accomplishments with regard to vascular progenitor cell studies in moyamoya disease.
Circulating EPCs originate from the bone marrow, and they seem to take part in both postnatal vasculogenesis and vascular homeostasis7). EPCs are usually characterized by the surface protein expression of CD31 [an endothelial cell marker, also known as PECAM-1 (platelet endothelial cell adhesion molecule-1)], CD34 (a hematopoietic or progenitor marker), and vascular endothelial growth factor receptor-2 [VEGFR-2, also known as KDR [kinase insert domain receptor)]. Several investigators consider the stem cell marker CD133 to be a more precise EPC marker. CD133+/CD34+/VEGFR-2+ cells are considered to represent a more primitive EPC type with high proliferative potential. In identifying EPCs, the usefulness of other markers such as CD45 (the leukocyte common antigen) and CD14 (a myelomonocytic cell marker) is controversial3). The expression of von Willebrand factor, uptake of acetylated low-density lipoprotein, and enhanced endothelial nitric oxide synthase expression after shear-stress exposure can also be demonstrated14). Functionally, EPCs exhibit tremendous proliferative capacity and can be mobilized to vasculogenesis sites1). Additionally, they have a role in vascular maintenance at vascular injury sites.
SPCs from human peripheral blood were first isolated and cultured by Simper et al.12) in platelet-derived growth factor (PDGF) BB-enriched media. SPC-type outgrowth cells show a "hill-and-valley" appearance, while EPC-type outgrowth cells show a "cobblestone" appearance (Fig. 1)1214). In addition to the striking morphological differences in the outgrowth cells, they are characterized by positive staining with smooth muscle actin-α, smooth muscle myosin heavy chain (MHC) and calponin, which are smooth muscle-specific. In a previous experiment, these cells had 4- to 5-fold greater proliferative potential compared with endothelial outgrowth cells12).
For EPC culture from peripheral blood, the mononuclear cell fraction is isolated with density gradient centrifugation. The cells are plated in culture dishes that are coated with fibronectin and are cultured in endothelial cell growth medium that is enriched with vascular endothelial growth factor (VEGF). For SPC cultures, PDGF BB-enriched media seems to be essential. In a previous experiment, PDGF-BB was added at a concentration of 5 ng/mL since the seventh day of culturing6).
Two studies present opposing opinions on the relation between EPC circulation and moyamoya disease810). Rafat et al.10) demonstrated increased circulating hematopoietic stem cell and EPC levels (healthy controls vs. moyamoya disease subjects, 0.09% vs. 0.29% for CD34+CD133+ cells; 0.02% vs. 0.11% for CD34+CD133+VEGFR-2+ cells) from an adult moyamoya disease population (mean age, 35.7±15.9 years). In contrast, Kim et al.8) studied circulating EPCs from a pediatric moyamoya disease population (mean age, 7.5 years; age range, 2-13 years). On the day when the blood was collected (day 0), Kim et al.8) showed that the percentages of circulating cells that were positive for CD34, CD133, and KDR were significantly lower in the study subject population (normal controls vs. subjects, 8.48% vs. 2.97% for CD34; 0.56% vs. 0.09% for CD133; and 8.40% vs. 1.04% for KDR; all p less than 0.05). Rafat et al.10) presented data regarding double (CD34+CD133+ cells) and triple positive cells (CD34+CD133+VEGFR-2+ cells), while Kim et al.8) provided the numbers of cells that were positive for the respective markers. Thus, the differences in the datasets and ages of the study subjects make direct comparison of the two studies difficult. We acknowledge that the CD133+ cells made up less than 1% of the peripheral blood mononuclear cells in both studies. In general, there are more driving cues for vascular progenitor cell recruitment in patients with ischemic cerebrovascular diseases. Failure to show such an adequate response might be responsible for early and severe manifestations in pediatric MMD patients.
Jung et al.5) compared the numbers of colony-forming units (CFU, also called as 'cell clusters') of EPCs and outgrowth cells between MMD patients and healthy controls, and they demonstrated that the CFU numbers decreased (MMD patients vs. controls, 32.4±26.7 vs. 65.2±18.3, p<0.001) and the outgrowth cells were more frequently isolated in the MMD patients (33.3% vs. 10.4%, p=0.025). Notably, the outgrowth cells were isolated in all patients (n=3) who underwent revascularization surgery. Additionally, they found that low EPC-CFU number (less than 30) was more prevalent in the patients with more advanced MMD (p=0.001). They also showed that conditioned media from EPCs of MMD patients significantly induced less tube formation, which reflected impaired paracrine function in the patients' EPCs. Three-day cultures of mononuclear cells were used to produce the conditioned media, and human umbilical vein endothelial cells were employed for the tubule formation assay.
Pediatric MMD patients also showed reduced EPC cell cluster formation8). From a different perspective, Kim et al.8) conducted a tubule formation assay with late EPCs (i.e., cells with cobblestone morphology) from MMD patients and normal controls, and they showed that the patient cells had reduced tubule formation capability. A senescence-associated β-galactosidase assay revealed a higher percentage of senescent cells among the EPCs from the patients.
Recently, cells expressing CD34 and VEGFR2 were found in the thickened intima of supraclinoid internal carotid arteries that were collected from adult MMD patients13). These findings suggest that bone marrow-derived circulating EPCs may participate in development of the occlusive arterial lesions in MMD.
MMD histopathology is characterized by smooth muscle cell hyperplasia in the intima. The origin of the smooth muscle cells is one of the key questions regarding MMD pathophysiology. We established SPCs from the peripheral blood of MMD patients6). For cell culture, well plates that were coated with type I collagen were used. The fetal bovine serum concentration was set at 10% without VEGF enrichment. After 5 days, non-adherent cells were removed, and fresh culture medium was applied. At 1 week, PDGF-BB was added to the culture medium. After the initial cell cluster formation, two patterns of outgrowth cells appeared, which included SPCs (hill-and-valley pattern) and EPCs (cobblestone appearance). Fluorescence-activated cell sorter (FACS) analysis demonstrated that SPCs were present in 20% of the observed MMD patients, EPCs were present in 68% of the patients, and a mixed pattern was observed in 12% of the patients. Among the normal controls, FACS analysis revealed that SPCs were present in 30%, EPCs were present in 40%, and a mixed population was present in 40% of the subjects. A higher concentration of PDGF-BB led to failure of late outgrowths. VEGF enrichment also led to a failure of SPC outgrowth amongst the MMD patients.
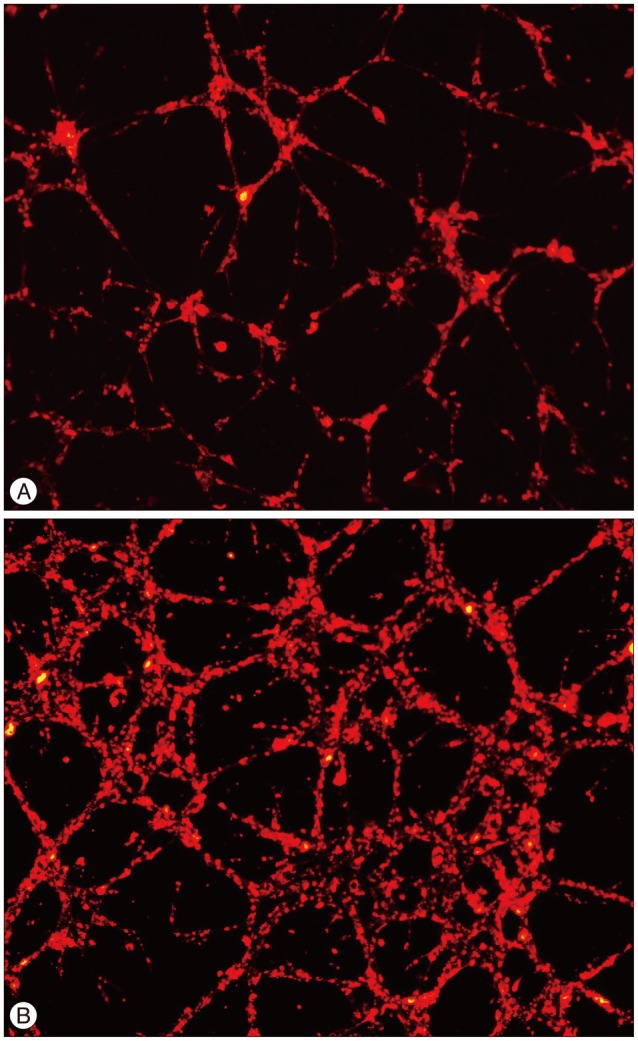
SPCs from the patients had lower PDGF receptor α expression (MMD patients vs. controls, 38.5%±2.6% vs. 66.1%±8.2%, p=0.0286)6). PDGF receptor β expression was not significantly different between the groups (87.7±3.1% vs. 96.6±1.2%, p= 0.1143). SPCs from the patients also had lower MHC (42.8±18.6% vs. 96±1.8%; p=0.0087) and calponin expression levels (87.1± 8.2% vs. 99.8±0.1%; p=0.0519). It was presumed that a defect in the cell maturation process might have occurred in the SPCs from the MMD patients. Interestingly, the tubule formation assay identified more irregularly arranged and thickened tubules with the patient SPCs, which is reminiscent of the pathologic findings of the MMD patient cerebral arteries (Fig. 2).
An Affymetrix gene chip mRNA microarray revealed 286 differentially expressed genes in SPCs from MMD patients with a >1.5-fold change and a t-test p value<0.016). Gene ontology (GO) analysis for the 124 up-regulated transcripts identified 19 terms (p<0.05), which included responses to endogenous stimulus, anterior/posterior pattern formation, and ephrin receptor signaling. Meanwhile, the GO analysis for the 162 down-regulated transcripts identified 26 terms (p<0.01), including cell adhesion, cell migration regulation, innate immune response, enzyme-linked receptor protein signaling pathways, and vasculature development.
It is known that circulating EPC number increases significantly after various injury types, including myocardial infarction, coronary artery bypass grafting, and stroke2411). Studies evaluating EPCs from MMD patients showed differences in EPC mobilization between adult and pediatric patients810), which might explain differences in the disease presentation and severity between the two age groups. Additionally, EPCs from MMD patients were found to be functionally impaired, whereby they failed to form tubules and showed defects in paracrine functions510). Further studies are under way to define the specific causes and possible solutions for these functional impairments.
It can be hypothesized that the thickened intima in MMD patients' cerebral arteries may (at least partly) result from malfunctioning EPCs and SPCs during the vascular repair and maintenance process. Although studies on SPCs are scarce, a study revealed some distinctive characteristics in relation to the cell culture finding, immaturity in terms of expression markers, defective tubule formation, and differential gene expression that were relevant to vascular development. Further studies are required to specify these characteristics in the setting of disease pathogenesis. Moreover, in vivo studies and animal models that mimic the disease are also required.
Vascular progenitor cells provide novel experimental models in MMD research. Still, there is much to learn in regard to MMD and vascular progenitor cells. Thus, research that utilizes these particular cells seems to be promising, although the establishment and maintenance of progenitor cells requires significant manpower, facilities, and funds.
Acknowledgements
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number : HI12C0066).
References
1. Asahara T, Murohara T, Sullivan A, Silver M, van der Zee R, Li T, et al. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997; 275:964–967. PMID: 9020076.

2. Chu K, Jung KH, Lee ST, Park HK, Sinn DI, Kim JM, et al. Circulating endothelial progenitor cells as a new marker of endothelial dysfunction or repair in acute stroke. Stroke. 2008; 39:1441–1447. PMID: 18356550.

3. Fadini GP, Losordo D, Dimmeler S. Critical reevaluation of endothelial progenitor cell phenotypes for therapeutic and diagnostic use. Circ Res. 2012; 110:624–637. PMID: 22343557.

4. Gill M, Dias S, Hattori K, Rivera ML, Hicklin D, Witte L, et al. Vascular trauma induces rapid but transient mobilization of VEGFR2(+)AC133(+) endothelial precursor cells. Circ Res. 2001; 88:167–174. PMID: 11157668.

5. Jung KH, Chu K, Lee ST, Park HK, Kim DH, Kim JH, et al. Circulating endothelial progenitor cells as a pathogenetic marker of moyamoya disease. J Cereb Blood Flow Metab. 2008; 28:1795–1803. PMID: 18612318.

6. Kang HS, Moon YJ, Kim YY, Park WY, Park AK, Wang KC, et al. Smooth-muscle progenitor cells isolated from patients with moyamoya disease : novel experimental cell model. J Neurosurg. 2014; 120:415–425. PMID: 24160477.

7. Khakoo AY, Finkel T. Endothelial progenitor cells. Annu Rev Med. 2005; 56:79–101. PMID: 15660503.

8. Kim JH, Jung JH, Phi JH, Kang HS, Kim JE, Chae JH, et al. Decreased level and defective function of circulating endothelial progenitor cells in children with moyamoya disease. J Neurosci Res. 2010; 88:510–518. PMID: 19774676.

9. Kim SK, Cho BK, Phi JH, Lee JY, Chae JH, Kim KJ, et al. Pediatric moyamoya disease : an analysis of 410 consecutive cases. Ann Neurol. 2010; 68:92–101. PMID: 20582955.
10. Rafat N, Beck GCh, Peña-Tapia PG, Schmiedek P, Vajkoczy P. Increased levels of circulating endothelial progenitor cells in patients with Moyamoya disease. Stroke. 2009; 40:432–438. PMID: 19095988.

11. Shintani S, Murohara T, Ikeda H, Ueno T, Honma T, Katoh A, et al. Mobilization of endothelial progenitor cells in patients with acute myocardial infarction. Circulation. 2001; 103:2776–2779. PMID: 11401930.

12. Simper D, Stalboerger PG, Panetta CJ, Wang S, Caplice NM. Smooth muscle progenitor cells in human blood. Circulation. 2002; 106:1199–1204. PMID: 12208793.

13. Sugiyama T, Kuroda S, Nakayama N, Tanaka S, Houkin K. Bone marrow-derived endothelial progenitor cells participate in the initiation of moyamoya disease. Neurol Med Chir (Tokyo). 2011; 51:767–773. PMID: 22123479.

14. Urbich C, Heeschen C, Aicher A, Dernbach E, Zeiher AM, Dimmeler S. Relevance of monocytic features for neovascularization capacity of circulating endothelial progenitor cells. Circulation. 2003; 108:2511–2516. PMID: 14581410.

Fig. 1
Vascular progenitor cells. Values on the y axis quantify the intensity of fluorescein isothiocyanate staining (FITC log), and on the x axis the values represent the number of cells counted. A : Early endothelial progenitor cells (EPCs), also called colony-forming units or cell clusters (×200). This cell cluster is composed of a central core of round cells that are surrounded by spindle shaped cells. B : Late EPCs, also known as endothelial outgrowth cells (×40). The cells are arranged in cobblestone-like formations. C : Smooth muscle progenitor cells (SPCs) (×40). These cells appear elon-gated and have a typical hill-and-valley appearance. D : Fluorescence-activated cell sorter analysis of the cells shown in C. The cells express smooth muscle myosin heavy chain (MHC) and calponin, which are smooth-muscle specific, with little expression of CD31 and fibroblast-specific protein 1 (FSP-1). αSMA : smooth muscle actin-alpha.
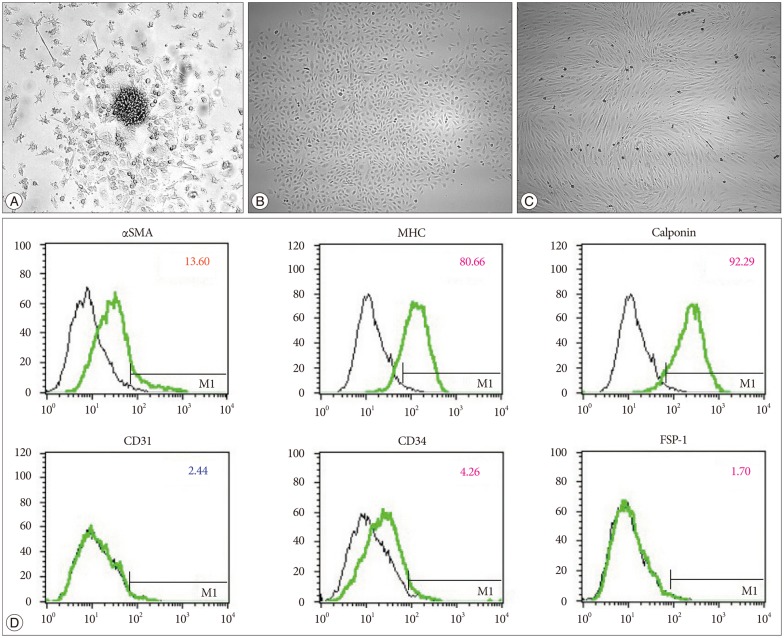
Fig. 2
Vascular progenitor cells. Values on the y axis quantify the intensity of fluorescein isothiocyanate staining (FITC log), and on the x axis the values represent the number of cells counted. A : Early endothelial progenitor cells (EPCs), also called colony-forming units or cell clusters (×200). This cell cluster is composed of a central core of round cells that are surrounded by spindle shaped cells. B : Late EPCs, also known as endothelial outgrowth cells (×40). The cells are arranged in cobblestone-like formations. C : Smooth muscle progenitor cells (SPCs) (×40). These cells appear elon-gated and have a typical hill-and-valley appearance. D : Fluorescence-activated cell sorter analysis of the cells shown in C. The cells express smooth muscle myosin heavy chain (MHC) and calponin, which are smooth-muscle specific, with little expression of CD31 and fibroblast-specific protein 1 (FSP-1). αSMA : smooth muscle actin-alpha.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download