INTRODUCTION
MATERIALS AND METHODS
Patient enrolment
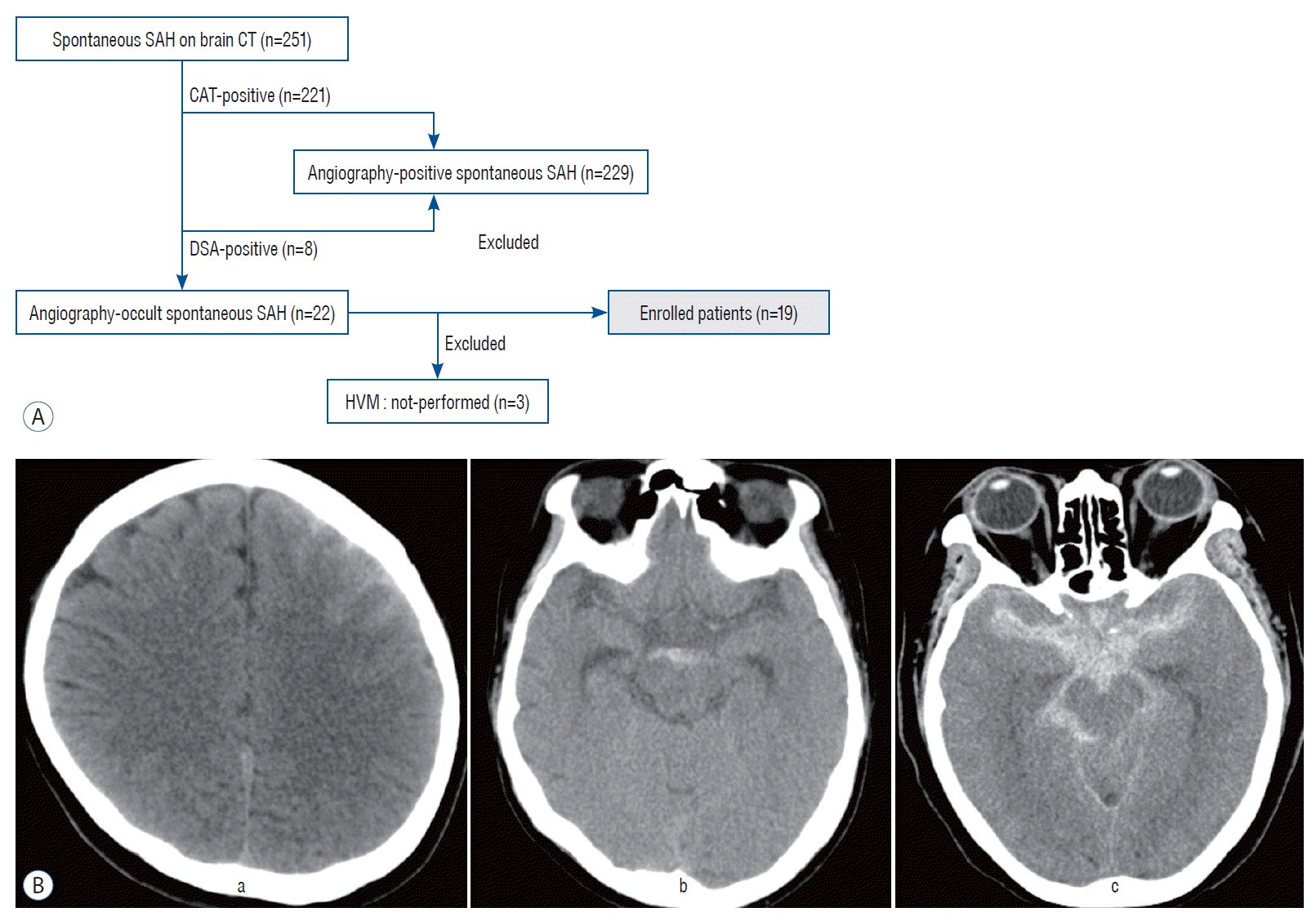 | Fig. 1.A : A flowchart representing the process of patient recruitment. B : The representative case illustration of three types of subarachnoid hemorrhage (SAH). A typical case of convexal type of SAH shows hemorrhage isolated in one or several adjacent sulci of left cerebral hemisphere without blood at the basal cisterns, interhemispheric fissure ventricle or parenchyma (a). Perimensencephalic type of SAH case demonstrates hemorrhage loculated in cisternal space of midbrain and the absence of a clot in other space (b). Diffuse type SAH indicates hemorrhage distributed widely in basal, interhemispheric, sylvian and perimesencephalic cisterns, which resembles a diffuse SAH caused by aneurysmal rupture (c). CT : computed tomography, CTA : computed tomography angiography, DSA : digital subtraction angiography, HVM : high-resolution vessel wall magnetic resonance imaging. |
Classification of SAH
Clinical and radiologic data
Protocol of 3D-HVM
Table 1.
Statistical analysis
RESULTS
General demographics
Table 2.
Convexal or perimesencephalic AOsSAH
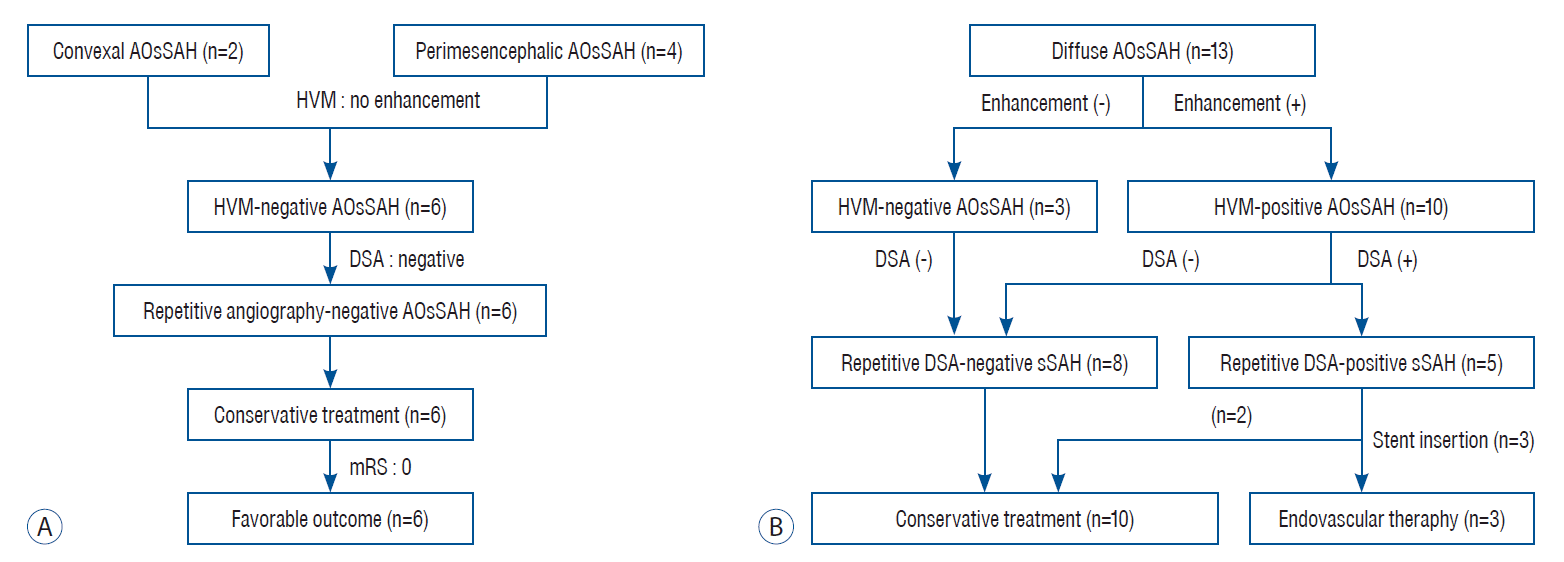 | Fig. 2.A flowchart of the enrolled patients. A : A flowchart showing evaluation and treatment process of convexal and perimesencephalic type subarachnoid hemorrhage patients. B : A flowchart showing evaluation and treatment process of diffuse type subarachnoid hemorrhage patients. AOsSAH : angiographically occult spontaneous subarachnoid hemorrhage, HVM : high-resolution vessel wall magnetic resonance imaging, DSA : digital subtraction angiography, sSAH : spontaneous subarachnoid hemorrhage. |
Diffuse AOsSAH
Table 3.
HVM : high-resolution vessel wall magnetic resonance imaging, AOsSAH : angiographically occult spontaneous subarachnoid hemorrhage, SAH : subarachnoid hemorrhage, mRS : modified Rankin Scale, M : male, D : diffuse type of SAH, BA : basilar artery, R/O : rule-out, MCA : middle cerebral artery, Rt : right, M1 : first segment of the middle cerebral artery, ICA : internal cerebral artery, BBA : blood blister-like aneurysm, F : female
Table 4.
Illustrative cases
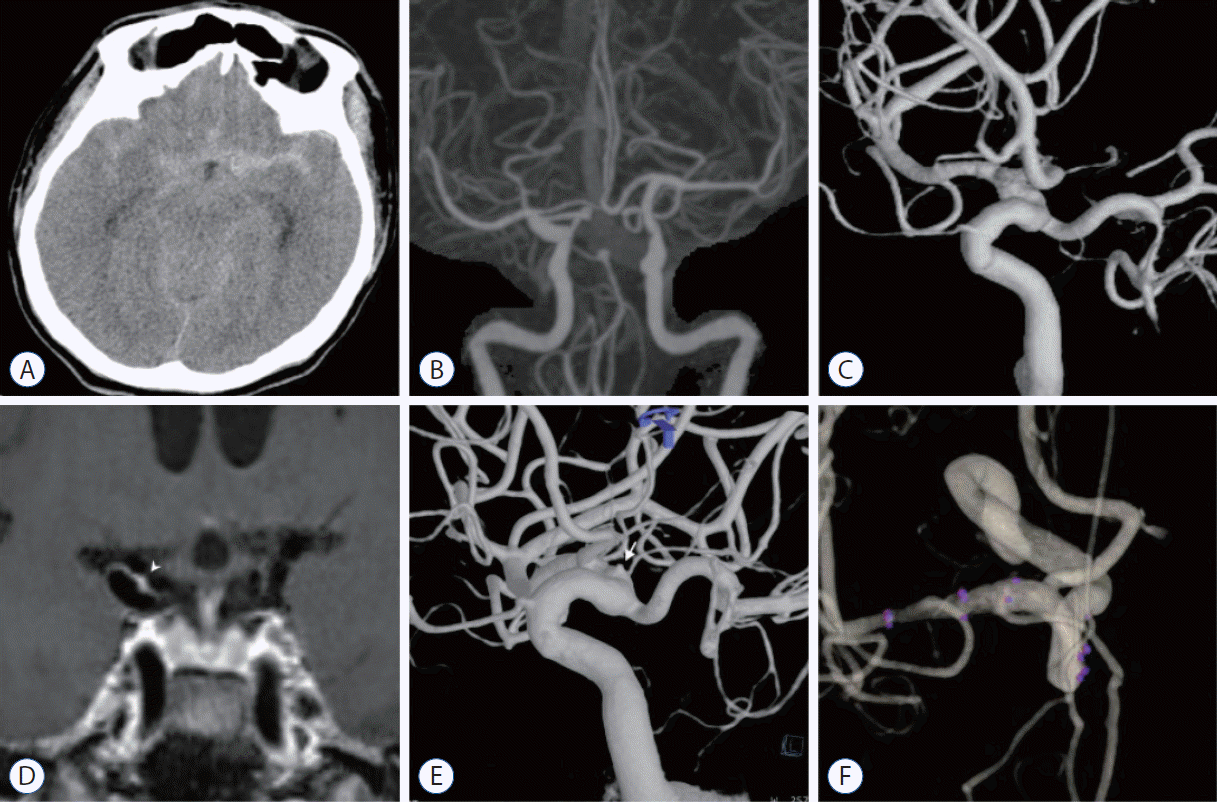 | Fig. 3.A case of ruptured blood blister-like aneurysm on right internal cerebral artery. A : A 62-year-old man was admitted in the emergency room complaining thunderclap worst headache. Brain computed tomography (CT) scan image shows diffuse type subarachnoid hemorrhage. The hemorrhage amount is slightly larger in left sylvian fissure than right side. B : The initial CT angiography shows no definitive arteriopathy causing subarachnoid hemorrhage. C : Six-vessel complete angiography taken on the first day of admission failed to discover any definitive vascular cause. D : Three-dimensional high-resolution vessel wall magnetic resonance imaging (MRI) taken on the fifth day of admission demonstrates strong focal enhancement on the dorsomedial wall of right side distal internal cerebral artery (white arrowhead). This finding suggests possibility of ruptured arteriopathy at this region. E : The second week angiography also failed to show any vasculopathy. The third week angiography high resolution 3-dimensional reconstruction guided by the finding from vessel wall MRI shows a blood blister-like aneurysm on the same location of strong enhancement (white arrow). This lesion was treated with overlapped laser cut stents. F : Six months follow-up angiography reveals complete healing of the ruptured blood blister-like aneurysm. |
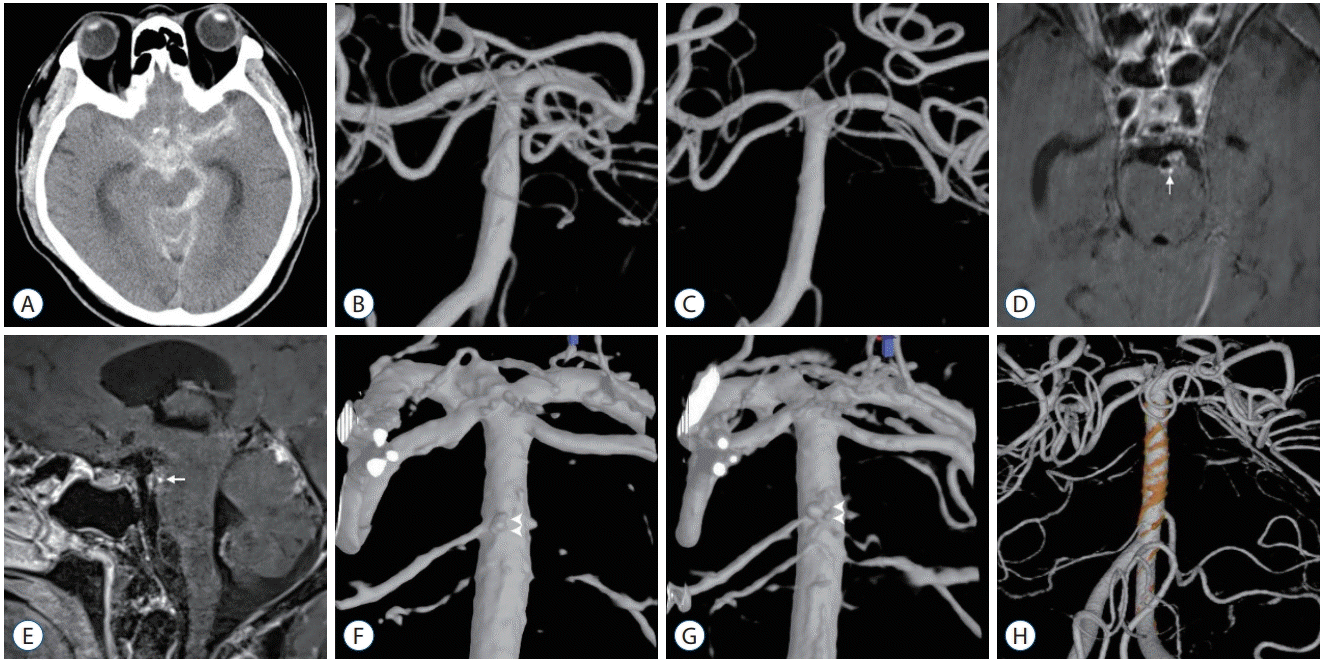 | Fig. 4.A case of ruptured perforator or blood blister-like aneurysm on posterior wall of distal basilar artery. A-C : Initial evaluation images showing diffuse subarachnoid hemorrhage on computed tomography scan and no definitive arteriopathy on first and second angiography. D and E : Three-dimensional high-resolution vessel wall magnetic resonance imaging (MRI) taken on the 10th day of admission demonstrates strong nodular enhancement on the posterior wall of basilar artery (white arrow). F : Re-do high-resolution 3-dimensional reconstruction of second angiography according to the vessel wall MRI finding reveals a tiny aneurysm on the posterior wall of the basilar artery at the concordant location of nodular enhancement on vessel wall MRI (white arrowheads). G : Third angiography shows definitive aneurysm on the same location (white arrowheads). H : This image shows the result of endovascular treatment using overlapped braided stents. |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download