INTRODUCTION
MATERIALS AND METHODS
Clinical characteristics of patients with recurrent high-grade glioma
Table 1.
Gamma knife radiosurgery for recurrent highgrade gliomas
Statistical analysis
RESULTS
Prognostic factors related with progression-free survival and overall survival
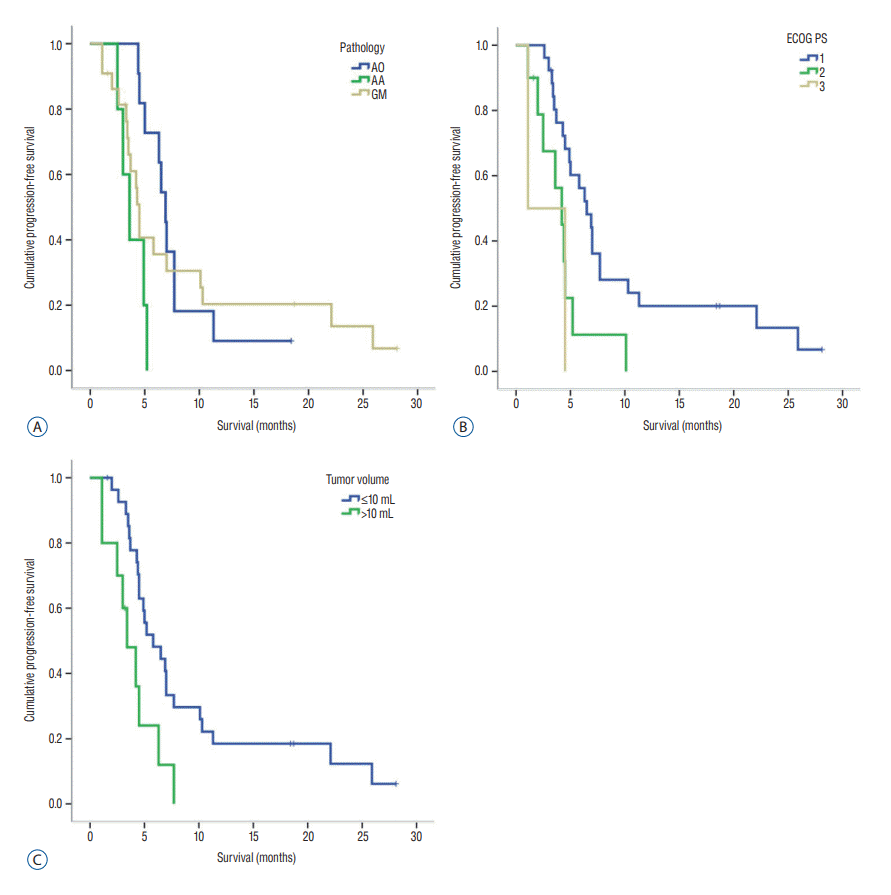 | Fig. 1.Kaplan-Meier curve related to progression-free survival. A : Median PFS was significantly longer in patients with anaplastic oligodendroglioma than in those with glioblastoma (p=0.001). B : Survival of patients with ECOG PS 1 was statistically significantly different compared to those with ECOG PS 3 (p=0.024). C : Survival of patients with tumor volume less than 10 mL was statistically significantly different compared to those with tumor volume over 10 mL (p=0.008). AO : anaplastic oligodendroglioma, AA : anaplastic astrocytoma, GM : glioblastoma; PFS : progression-free survival, ECOG : the Eastern Cooperative Oncology Group, PS : performance status. |
 | Fig. 2.A : Kaplan-Meier curve related with overall survival. Patients with ECOG PS 1 and 2 had better OS compared to those with ECOG PS 3 (p=0.000 and 0.042, respectively). B : Patients with tumor volume less than 10 mL had better OS compared to those with tumor volume over 10 mL (p=0.002). ECOG : the Eastern Cooperative Oncology Group, PS : performance status, OS : overall survival. |
Table 2.
Table 3.
Case presentation 1 : GKR for recurred glioblastoma
 | Fig. 3.GKR for recurred glioblastoma. A : Brain MRI showed the recurred homogenous enhanced lesion on the parietal lobe. B : Methionine-PET image showed a hot uptake lesion. C : GKR was performed for this lesion. The tumor volume was 2.05 mL, and the marginal radiation dose was 18 Gy with 50% isodose. D : Follow-up brain MRI after 24 months showed a decreased enhancing lesion without any neurologic symptoms. GKR : gamma knife radiosurgery, MRI : magnetic resonance images, PET : positron emission tomography. |
Case presentation 2 : GKR for recurred anaplastic oligodendroglioma
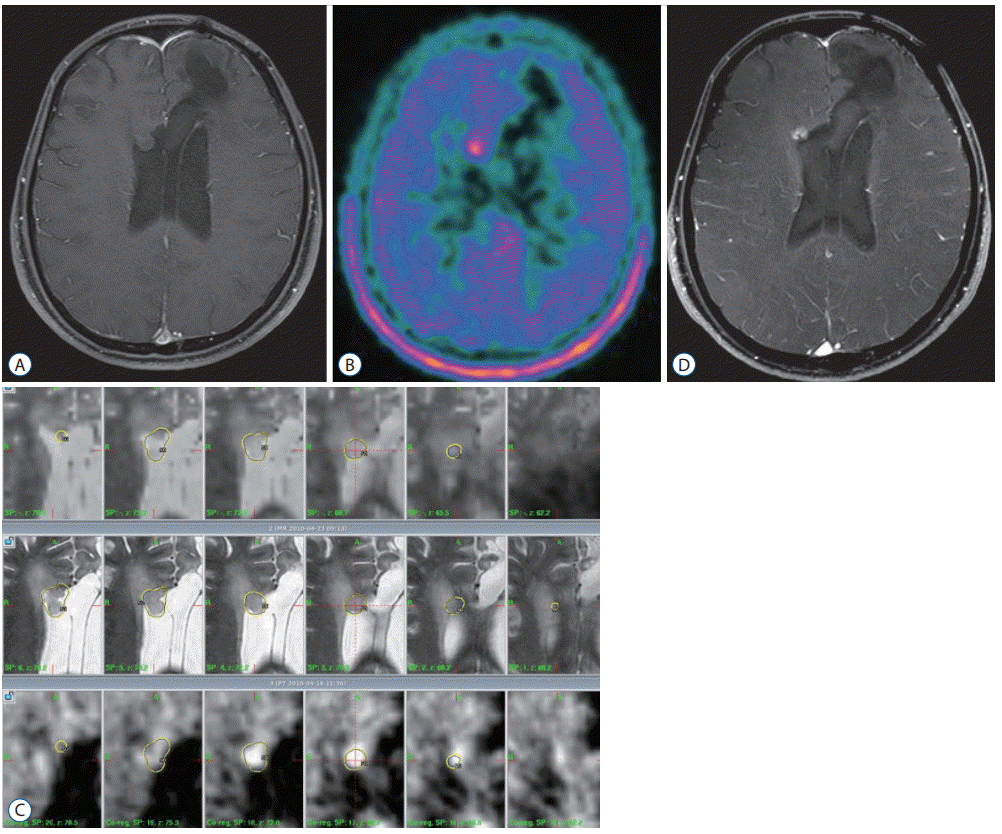 | Fig. 4.GKR for recurred anaplastic oligodendroglioma. A : Brain MRI showed a non-enhancing lesion adjacent to the right frontal horn. B : Methionine-PET image showed a hot uptake lesion. C : GKR was performed. The tumor volume was 2.0 mL, and the marginal radiation dose was 18 Gy with 50% isodose. D : Follow-up brain MRI after 11.3 months showed a stable enhancing lesion without any neurologic symptoms. GKR : gamma knife radiosurgery, MRI : magnetic resonance images, PET : positron emission tomography. |




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download