1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015; 65:5–29.

2. Han HH, Park JW, Na JC, Chung BH, Kim CS, Ko WJ. Epidemiology of prostate cancer in South Korea. Prostate Int. 2015; 3:99–102.

3. Chopra S, Srivastava A, Tewari A. Robotic radical prostatectomy: the new gold standard. Arab J Urol. 2012; 10:23–31.

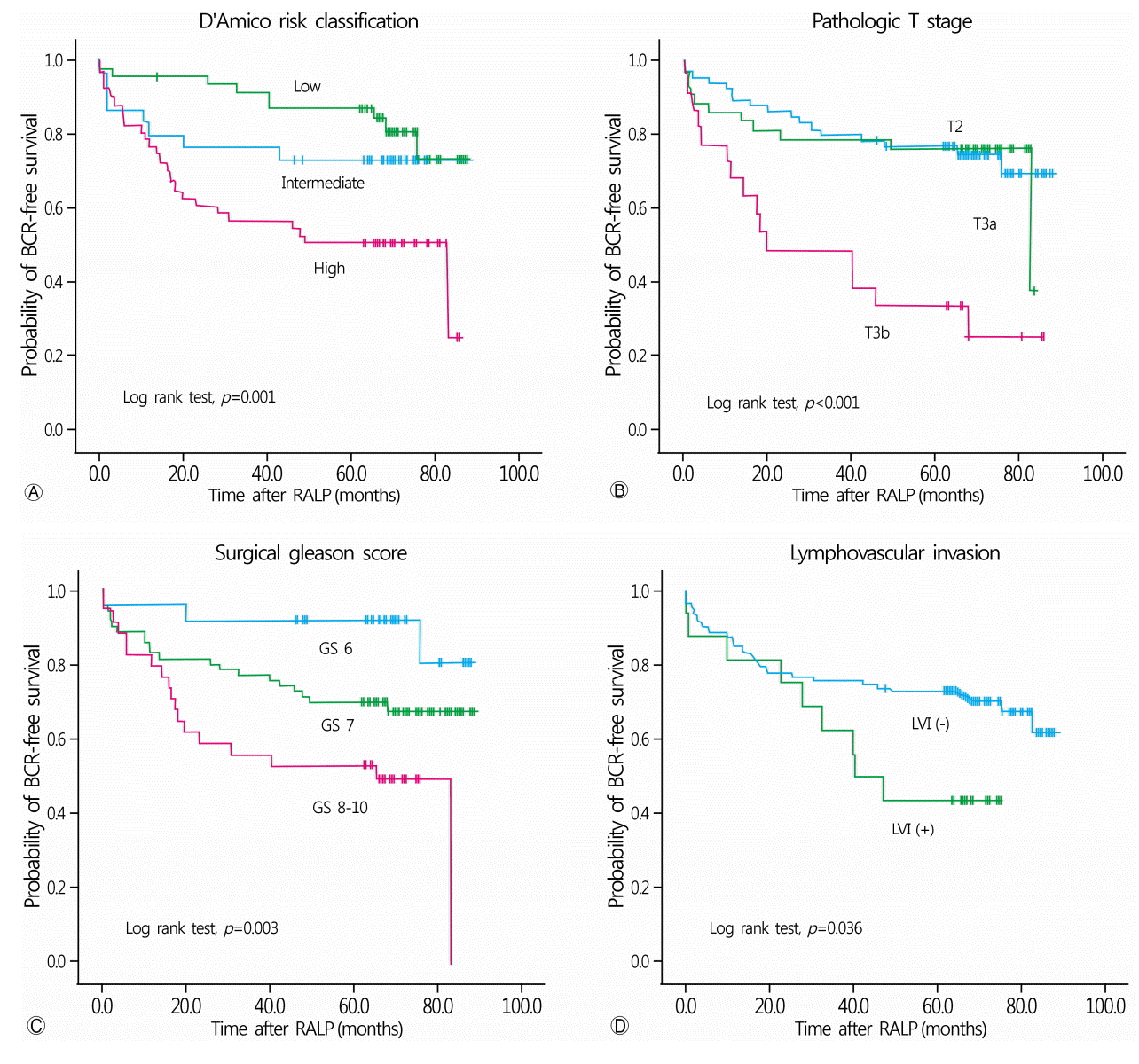
4. D’Amico AV, Moul J, Carroll PR, Sun L, Lubeck D, Chen MH. Cancer-specific mortality after surgery or radiation for patients with clinically localized prostate cancer managed during the prostate-specific antigen era. J Clin Oncol. 2003; 21:2163–72.

5. Luciani LG, Mattevi D, Mantovani W, Cai T, Chiodini S, Vattovani V, et al. Retropubic, laparoscopic, and robot-assisted radical prostatectomy: a comparative analysis of the surgical outcomes in a single regional center. Curr Urol. 2017; 11:36–41.

6. Nossiter J, Sujenthiran A, Charman SC, Cathcart PJ, Aggarwal A, Payne H, et al. Robot-assisted radical prostatectomy vs laparoscopic and open retropubic radical prostatectomy: functional outcomes 18 months after diagnosis from a national cohort study in England. Br J Cancer. 2018; 118:489–94.

7. Haglind E, Carlsson S, Stranne J, Wallerstedt A, Wilderäng U, Thorsteinsdottir T, et al. Urinary incontinence and erectile dysfunction after robotic versus open radical prostatectomy: a prospective, controlled, nonrandomised trial. Eur Urol. 2015; 68:216–25.

8. Kang YJ, Kim HS, Jang WS, Kwon JK, Yoon CY, Lee JY, et al. Impact of lymphovascular invasion on lymph node metastasis for patients undergoing radical prostatectomy with negative resection margin. BMC Cancer. 2017; 17:321.

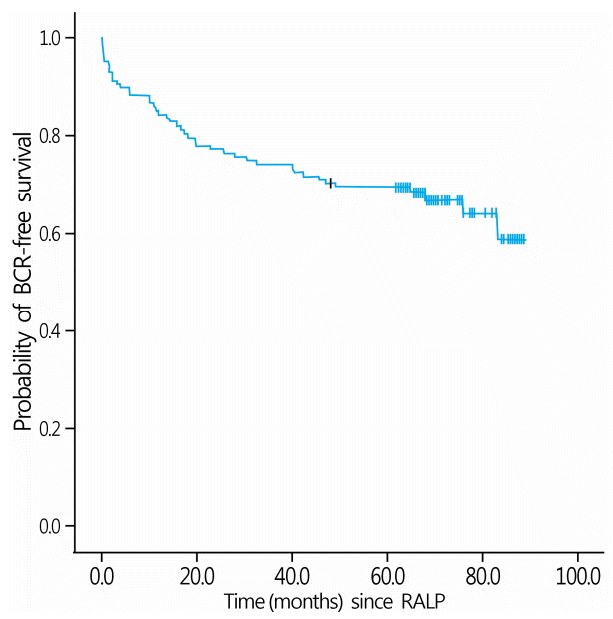
9. Menon M, Bhandari M, Gupta N, Lane Z, Peabody JO, Rogers CG, et al. Biochemical recurrence following robot-assisted radical prostatectomy: analysis of 1384 patients with a median 5-year follow-up. Eur Urol. 2010; 58:838–46.

10. Rajan P, Hagman A, Sooriakumaran P, Nyberg T, Wallerstedt A, Adding C, et al. Oncologic outcomes after robot-assisted radical prostatectomy: a large European single-centre cohort with median 10-year follow-up. Eur Urol Focus. 2018; 4:351–9.

11. Sooriakumaran P, Haendler L, Nyberg T, Gronberg H, Nilsson A, Carlsson S, et al. Biochemical recurrence after robot-assisted radical prostatectomy in a European single-centre cohort with a minimum follow-up time of 5 years. Eur Urol. 2012; 62:768–74.

12. Lee SH, Seo HJ, Lee NR, Son SK, Kim DK, Rha KH. Robot-assisted radical prostatectomy has lower biochemical recurrence than laparoscopic radical prostatectomy: systematic review and meta-analysis. Investig Clin Urol. 2017; 58:152–63.

13. Abdel Raheem A, Kim DK, Santok GD, Alabdulaali I, Chung BH, Choi YD, et al. Stratified analysis of 800 Asian patients after robot-assisted radical prostatectomy with a median 64 months of follow up. Int J Urol. 2016; 23:765–74.

14. Diaz M, Peabody JO, Kapoor V, Sammon J, Rogers CG, Stricker H, et al. Oncologic outcomes at 10 years following robotic radical prostatectomy. Eur Urol. 2015; 67:1168–76.

15. Shao IH, Chang YH, Hou CM, Lin ZF, Wu CT. Predictors of short-term and long-term incontinence after robot-assisted radical prostatectomy. J Int Med Res. 2018; 46:421–9.

16. Kadono Y, Ueno S, Kadomoto S, Iwamoto H, Takezawa Y, Nakashima K, et al. Use of preoperative factors including urodynamic evaluations and nerve-sparing status for predicting urinary continence recovery after robot-assisted radical prostatectomy: nerve-sparing technique contributes to the reduction of postprostatectomy incontinence. Neurourol Urodyn. 2016; 35:1034–9.
17. Honda M, Kawamoto B, Morizane S, Hikita K, Muraoka K, Sejima T, et al. A prognostic model for predicting urinary incontinence after robot-assisted radical prostatectomy. Int J Med Robot. 2017; 13:doi: 10.1002/rcs.1780.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download