Abstract
Background
Nonalcoholic fatty liver disease dramatically improves after bariatric surgery, primarily due to improvements in hepatic insulin sensitivity. Since the procedure for gastric cancer surgery is very similar to that for bariatric surgery, we investigated changes in fatty liver following gastrectomy for gastric cancer according to the type of surgery.
Methods
We evaluated hepatic steatosis in 212 early gastric cancer patients using Hounsfield units (HUs) on non-contrast computed tomography preoperatively and 6, 12, and 24 months after surgery. We compared the preoperative and postoperative liver-to-spleen HU ratio according to the type of surgery: Billroth I, Billroth II, and total gastrectomy with Roux-en-Y reconstruction.
Results
The initial results (liver/spleen HUs and the liver-to-spleen HU ratio) did not significantly differ according to surgical group. After surgery, only patients who underwent total gastrectomy with Roux-en-Y exhibited significant changes in the liver-to-spleen HU ratio at 6 months. In 26 patients who had higher initial HU levels of the spleen than the liver, the liver-to-spleen HU ratio significantly increased from 0.836 to 1.115 at 6 months, 1.109 at 12 months, and 1.102 at 24 months (p<0.01).
Conclusion
Significant changes in hepatic steatosis were found in even normal patients (with higher liver than spleen HU values) who underwent total gastrectomy with Roux-en-Y. Patients who initially had fatty liver also showed a significant increase in the liver-to-spleen HU ratio. These results suggest that total gastrectomy with Roux-en-Y reconstruction can have a positive effect on the improvement of hepatic steatosis.
Many studies have suggested that bariatric surgery has a remarkable effect on clinical manifestations of type 2 diabetes (T2DM). Buchwald et al. [1] reported that 82% of patients in their meta-analysis achieved resolution of clinical and laboratory manifestations of T2DM in the first 2 years after bariatric surgery, while 62% remained free of T2DM for more than 2 years after bariatric surgery. Furthermore, the rates of T2DM resolution differed by surgery type [1,2]. A meta-analysis showed T2DM resolution rates according to surgical type: 48% in laparoscopic adjustable gastric banding, 84% in Roux-en Y gastric bypass (RYGB), and 98% in biliopancreatic diversion [2].
In addition to T2DM, nonalcoholic fatty liver disease (NAFLD) also improved after bariatric surgery [3,4]. The spectrum of NAFLD ranges from simple hepatic steatosis to more severe nonalcoholic steatohepatitis, fibrosis, end-stage liver disease, and hepatocellular carcinoma [5]. The prevalence of NAFLD is between 10% and 25% in the general Korean population [6]. NAFLD in Korea is closely associated with several components of metabolic syndrome including obesity, hypertension, T2DM, and dyslipidemia, as also seen in Western countries [6]. Insulin resistance and hyperinsulinemia might play roles in the pathogenesis of NAFLD in patients with obesity and of normal body weight [4,6]. The mechanism underlying improvement of NAFLD after bariatric surgery suggests early improvement of hepatic insulin sensitivity [4,7].
According to the Korea National Cancer Incidence Database from 1999 to 2012, stomach cancer was one of the leading primary cancers [8]. In particular, stomach cancer is predicted to be the most common cancer in males. The age-standard mortality rate per 100,000 cases of stomach cancer was 7.8 in Korea in 2015 [8]. The treatment of choice for stomach cancer is surgery, with a procedure very similar to that of bariatric surgery. As a result, we compared changes in fatty liver following gastric cancer surgery based on which surgery.
Ethical statements: This study was approved by the Institutional Review Board of Kosin University Gospel Hospital (KUGH 2016-12-001). The informed consent was waived because this design is a retrospective study.
A retrospective review was conducted with the medical records of 374 patients who underwent gastric cancer surgery in Kosin University Gospel Hospital from 1 January to 31 December of 2013. Among the 374 patients, 212 had early gastric cancer.
We only enrolled early gastric cancer patients who underwent computed tomography (CT) scan before surgery and 6, 12, and 24 months after surgery. We evaluated hepatic steatosis using Hounsfield units (HUs) on non-contrast CT images. Hepatic attenuation only and liver-to-spleen (LS) ratio in HUs were calculated prior to the operation and at 6, 12, and 24 months after surgery. We compared the preoperative and postoperative results according to reconstruction method: Billroth I, Billroth II, and Roux-en Y.
Body weights and heights were measured using standardized techniques and equipment with participants wearing light indoor clothing without shoes. Body mass index (BMI) was calculated as weight divided by height squared (kg/m2). Blood samples were obtained after at least 8 hours of fasting. CT scans were performed with SOMATOM Sensation or SOMATOM Definition Flash (Siemens, Munich, Germany).
An endocrinologist who was educated by a radiologist measured hepatic and splenic CT attenuation on non-enhanced CT scans. Fat accumulation of the liver decreases the attenuation of CT [9]. On three cross-sections taken at different hepatic levels, the hepatic CT attenuation was assessed by selecting three circular regions of interest with an area of at least 2 cm2. By excluding areas of hepatic vascular and biliary structures, the largest possible region was chosen. Hepatic CT attenuation was calculated as the mean of the three measurements. Splenic CT attenuation was calculated through the same method.
The presence of fatty liver disease was defined as CT evidence of hepatic steatosis. The LS ratio was used to normalize hepatic attenuation with splenic attenuation [9]. CT steatosis was defined as a LS ratio less than one, and hepatic attenuation divided by splenic CT attenuation less than one, which has previously been proven to correlate with the presence of steatosis in liver biopsy and to be particularly specific for the presence of steatosis [10].
Differences in baseline characteristics were evaluated by one-way analysis of variance in the three surgical groups. Furthermore, paired t-tests were used to compare the changes of CT steatosis with follow-up in each surgery group. All data are presented as means with standard deviations. A p-value <0.05 was considered statistically significant. All analyses were performed using SPSS, version 23.0 (IBM Corp., Armonk, NY, USA).
This study was composed of a total of 212 early gastric cancer patients with a mean age of 58.43±10.53 years. Among them, 62.3% (n=132) underwent surgery with Billroth I, 22.2% (n=47) with Billroth II, and 15.5% (n=33) with total gastrectomy with Roux-en Y bypass surgery.
Preoperative baseline characteristics were not significantly different among surgical groups except for weight and BMI (Table 1). HUs of liver, HUs of spleen, and LS ratio were not significantly different among the groups.
After gastric cancer surgery, all groups exhibited a reduction in BMI (Table 2). In total gastrectomy in the Roux-en Y group, total cholesterol, low-density lipoprotein, and triglyceride levels significantly decreased 6 months after surgery. High-density lipoprotein cholesterol increased but was not statistically significant (Table 2).
Fig. 1 shows the changes in LS ratio with hepatic steatosis according to type of surgery. No significant changes were shown after surgery in the Billroth I or Billroth II operation group. However, there was significant increase in the LS ratio in the Roux-en Y group from 1.140 to 1.233 at 6 months, 1.223 at 12 months, and 1.261 at 24 months after surgery compared to the preoperation values (p=0.03).
In this study, most participants exhibited no hepatic steatosis before surgery. We selected 26 patients who had LS ratio <1, indicating the presence of hepatic steatosis [10], before surgery. Fig. 2 shows the changes in LS ratio after surgery. The LS ratio increased from 0.836 to 1.115 at 6 months, 1.109 at 12 months, and 1.102 at 24 months after surgery (p<0.001). Their initial mean BMI was 25.88±3.61 kg/m2, which is not considered obesity [11,12]. The mean reduction in BMI after gastric cancer surgery was 4.26±1.26 kg/m2 (Table 3).
Fatty liver disease is a predisposing condition of T2DM and increases insulin resistance [13]. Meta-analysis revealed that the improvement or resolution rate of hepatic steatosis after bariatric surgery was close to 90% [3]. The subjects in the included studies were severely obese, with BMIs ranging from 43.9 to 56 kg/m2 before bariatric surgery, classifying them as obesity class III according to the World Health Organization (WHO) [11,12]. After surgery, the BMIs remarkably decreased as a percentage reduction of mean BMI, ranging from 19.11% to 41.76%. These results pertaining to resolution of hepatic steatosis after bariatric surgery might be influenced by weight reduction. Therefore, remission of T2DM after bariatric surgery could be partially explained by resolution of hepatic steatosis.
The methods for bariatric and stomach cancer surgeries are very similar, and there have been reports that improvements in T2DM after gastric cancer surgery are similar to those achieved with bariatric surgery. One retrospective study reported that T2DM was resolved in 15.1% and improved in 30.4% of patients after stomach cancer surgery [14]. Another study revealed a better effect with T2DM. In patients who underwent Billroth II surgery, 85% exhibited improvement, while 88% of patients who underwent Roux-en-Y showed improvement [15]. However, to the best of our knowledge, no study has addressed fatty liver changes after stomach cancer surgery.
In this study, only 26 patients had fatty liver (LS ratio <1) before surgery. Their LS ratio was dramatically improved 6 months after stomach cancer surgery, and the improvement was sustained for 24 months. Almost all participants had normal body weight before surgery. Their preoperative BMI was lower than that of patients with obesity, defined as BMI of 25 kg/m2 or greater [11,12]. The participants did not achieve the hepatic steatosis criteria by CT LS ratio before gastric cancer surgery. Interestingly, hepatic CT attenuation and LS ratio increased after surgery even though they remained within the normal range. These results suggest that normal-weight patients and not only fatty liver patients can experience improvement after stomach cancer surgery. The reduction of mean BMI after surgery in this study was much smaller than in a previous meta-analysis [3]. These results suggest that improvement of hepatic steatosis after gastric surgery is due to another mechanism in addition to weight reduction. Our observations are consistent with those from previous studies. A number of studies has shown that improvement in the manifestations of metabolic syndrome including NAFLD after bariatric surgery is not solely dependent on weight [7,16-18]. Bojsen-Moller et al. [7] reported improvement in basal hepatic insulin sensitivity within 1 week after RYGB in obese T2DM patients, long before any significant weight reduction was achieved [19]. They suggested that this immediate improvement of hepatic insulin sensitivity could be a response to calorie restriction after bariatric surgery. They also showed an increment of glucagon-like peptide-1 (GLP-1) secretion that reached almost 5-fold peak concentration and a 12-fold area under the curve after oral glucose exposure with RYGB, which might be one mechanism for weight-independent improvement after surgery. The relationship between GLP-1 and hepatic steatosis has been addressed in several in vivo and in vitro studies [20-22]. These studies have suggested that GLP-1 acts directly on hepatocytes through activation of metabolic signaling pathways including the insulin signaling and AMP-activated protein kinase (AMPK) pathways. Therefore, improvement of insulin sensitivity is achieved. The AMPK pathway is associated with hepatic lipogenesis [21]. GLP-1 binds to its receptor and enhances the increase in cyclic AMP, a known activator of AMPK [23]. This process accelerates the phosphorylation of AMPK and reduces lipogenesis in hepatocytes [21]. This could explain the improvement of hepatic steatosis after gastric cancer surgery even though a much lower reduction of BMI was achieved compared to previous meta-analysis.
Previous studies have shown that bypassing the proximal gut for delivery of nutrients can enhance GLP-1 secretion and create differences in degree of improvement of fatty liver and T2DM after bariatric surgery according to surgery type [1,24,25]. RYGB or biliopancreatic diversion exhibited better results than sleeve gastrectomy [1]. Therefore, the importance of bypassing the proximal intestine has been demonstrated [24,25]. Our study results are consistent with other studies, as total gastrectomy with the Roux-en Y group exhibited significant changes in the LS ratio as well as the lipid profile compared to other surgery groups. This is a remarkable result, considering that the length of the bypass limb in the Roux-en-Y group (usually 30–40 cm) is very short compared to that in bariatric surgery (usually from the duodenum to 50–100 cm distal to the ligament of Treitz) [26]. This finding indicates that bypassing the proximal intestine even by a very short length could improve metabolic disease. Recently, several studies have suggested that GLP-1 and bile acid are related to this response [27].
Due to the retrospective design of this study, data collection was limited. Liver biopsy to confirm the states of hepatic steatosis was not performed. There is a limitation in not being able to collect data on whether patients have underlying diseases such as T2DM, hypertension, hyperlipidemia, drinking alcohol, and taking drugs related to underlying diseases. These data could influence as confounding variables. In addition, there is no patient with distal gastrectomy with Roux-en Y anastomosis, so the difference according to the anastomosis method cannot be confirmed. In the Billroth II patient group, there was no change in the L to S ratio even though the weight loss was the greatest before and 6 months after surgery. In this study, it was not possible to determine whether alcohol consumption or an underlying medical condition had an effect. Tests for insulin resistance, sensitivity, and GLP-1 level were not included. In addition, this study involved stomach cancer surgery, not bariatric surgery. Therefore, the participants were early gastric cancer patients with initial relatively normal body weight and normal hepatic steatosis state. Although these were the conditions, there were significant changes in hepatic steatosis after surgery.
Recently, the 5-year survival rates and remission rates were shown to be very high in early gastric cancer patients [28]. Therefore, surgical improvement of cancer remission and metabolic disease can affect quality of life and mortality rates in early gastric cancer patients with metabolic disease. In the case of gastrectomy patients, it can be inferred that metabolic diseases like obesity, NAFLD, and T2DM tend to improve.
Stomach cancer surgery results in significant improvements of hepatic steatosis in early gastric cancer patients with fatty liver. Especially, total gastrectomy with Roux-en Y can increase liver HUs and LS ratios even in normal patients who exhibited no fatty liver before surgery. These results can help clinicians determine the effectiveness of the treatment of metabolic disease in postoperative patients with early gastric cancer with metabolic disease.
References
1. Buchwald H, Estok R, Fahrbach K, Banel D, Jensen MD, Pories WJ, et al. Weight and type 2 diabetes after bariatric surgery: systematic review and meta-analysis. Am J Med. 2009; 122:248–56.

2. Padwal R, Klarenbach S, Wiebe N, Birch D, Karmali S, Manns B, et al. Bariatric surgery: a systematic review and network meta-analysis of randomized trials. Obes Rev. 2011; 12:602–21.

3. Mummadi RR, Kasturi KS, Chennareddygari S, Sood GK. Effect of bariatric surgery on nonalcoholic fatty liver disease: systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2008; 6:1396–402.

4. Mathurin P, Hollebecque A, Arnalsteen L, Buob D, Leteurtre E, Caiazzo R, et al. Prospective study of the long-term effects of bariatric surgery on liver injury in patients without advanced disease. Gastroenterology. 2009; 137:532–40.

5. Liou I, Kowdley KV. Natural history of nonalcoholic steatohepatitis. J Clin Gastroenterol. 2006; 40 Suppl 1:S11–6.
6. Park SH. Current status of liver disease in Korea: nonalcoholic fatty liver disease. Korean J Hepatol. 2009; 15 Suppl 6:S34–9.

7. Bojsen-Moller KN, Dirksen C, Jorgensen NB, Jacobsen SH, Serup AK, Albers PH, et al. Early enhancements of hepatic and later of peripheral insulin sensitivity combined with increased postprandial insulin secretion contribute to improved glycemic control after Roux-en-Y gastric bypass. Diabetes. 2014; 63:1725–37.

8. Jung KW, Won YJ, Oh CM, Kong HJ, Cho H, Lee DH, et al. Prediction of cancer incidence and mortality in Korea, 2015. Cancer Res Treat. 2015; 47:142–8.

9. Kodama Y, Ng CS, Wu TT, Ayers GD, Curley SA, Abdalla EK, et al. Comparison of CT methods for determining the fat content of the liver. AJR Am J Roentgenol. 2007; 188:1307–12.

10. Jacobs JE, Birnbaum BA, Shapiro MA, Langlotz CP, Slosman F, Rubesin SE, et al. Diagnostic criteria for fatty infiltration of the liver on contrast-enhanced helical CT. AJR Am J Roentgenol. 1998; 171:659–64.

11. WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004; 363:157–63.
12. Obesity: preventing and managing the global epidemic: report of a WHO consultation. World Health Organ Tech Rep Ser. 2000; 894:i–xii. 1–253.
13. Adams LA, Waters OR, Knuiman MW, Elliott RR, Olynyk JK. NAFLD as a risk factor for the development of diabetes and the metabolic syndrome: an eleven-year follow-up study. Am J Gastroenterol. 2009; 104:861–7.

14. Kim JW, Cheong JH, Hyung WJ, Choi SH, Noh SH. Outcome after gastrectomy in gastric cancer patients with type 2 diabetes. World J Gastroenterol. 2012; 18:49–54.

15. Kang KC, Shin SH, Lee YJ, Heo YS. Influence of gastrectomy for stomach cancer on type 2 diabetes mellitus for patients with a body mass index less than 30 kg/m(2). J Korean Surg Soc. 2012; 82:347–55.

16. Clark JM, Alkhuraishi AR, Solga SF, Alli P, Diehl AM, Magnuson TH. Roux-en-Y gastric bypass improves liver histology in patients with non-alcoholic fatty liver disease. Obes Res. 2005; 13:1180–6.

18. Barker KB, Palekar NA, Bowers SP, Goldberg JE, Pulcini JP, Harrison SA. Non-alcoholic steatohepatitis: effect of Roux-en-Y gastric bypass surgery. Am J Gastroenterol. 2006; 101:368–73.

19. Dirksen C, Jorgensen NB, Bojsen-Moller KN, Jacobsen SH, Hansen DL, Worm D, et al. Mechanisms of improved glycaemic control after Roux-en-Y gastric bypass. Diabetologia. 2012; 55:1890–901.

20. Gupta NA, Mells J, Dunham RM, Grakoui A, Handy J, Saxena NK, et al. Glucagon-like peptide-1 receptor is present on human hepatocytes and has a direct role in decreasing hepatic steatosis in vitro by modulating elements of the insulin signaling pathway. Hepatology. 2010; 51:1584–92.

21. Ben-Shlomo S, Zvibel I, Shnell M, Shlomai A, Chepurko E, Halpern Z, et al. Glucagon-like peptide-1 reduces hepatic lipogenesis via activation of AMP-activated protein kinase. J Hepatol. 2011; 54:1214–23.

22. Svegliati-Baroni G, Saccomanno S, Rychlicki C, Agostinelli L, De Minicis S, Candelaresi C, et al. Glucagon-like peptide-1 receptor activation stimulates hepatic lipid oxidation and restores hepatic signalling alteration induced by a high-fat diet in nonalcoholic steatohepatitis. Liver Int. 2011; 31:1285–97.

23. Viollet B, Foretz M, Guigas B, Horman S, Dentin R, Bertrand L, et al. Activation of AMP-activated protein kinase in the liver: a new strategy for the management of metabolic hepatic disorders. J Physiol. 2006; 574(Pt 1):41–53.

24. Pournaras DJ, Aasheim ET, Bueter M, Ahmed AR, Welbourn R, Olbers T, et al. Effect of bypassing the proximal gut on gut hormones involved with glycemic control and weight loss. Surg Obes Relat Dis. 2012; 8:371–4.

25. Lindqvist A, Spegel P, Ekelund M, Mulder H, Groop L, Hedenbro J, et al. Effects of ingestion routes on hormonal and metabolic profiles in gastric-bypassed humans. J Clin Endocrinol Metab. 2013; 98:E856–61.

26. Korner J, Inabnet W, Febres G, Conwell IM, McMahon DJ, Salas R, et al. Prospective study of gut hormone and metabolic changes after adjustable gastric banding and Roux-en-Y gastric bypass. Int J Obes (Lond). 2009; 33:786–95.

Fig. 1.
Changes in the liver-to-spleen (LS) ratio according to type of surgery. a)p<0.05 compared to the preoperative values.
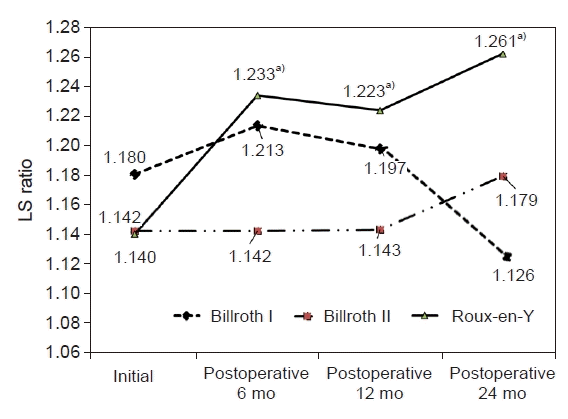
Fig. 2.
Changes in the liver-to-spleen (LS) ratio among patients who had hepatic steatosis before surgery. a)p<0.05 compared to the preoperative values.
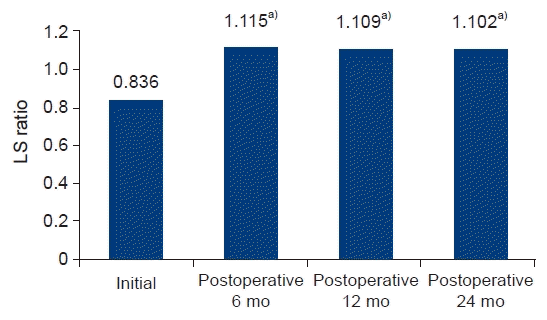
Table 1.
Baseline characteristics of the study participants according to the type of surgery
Values are presented as number (%) or mean±standard deviation..
BMI, body mass index; HbA1c, hemoglobin A1c; HDL, high-density lipoprotein; LDL, low-density lipoprotein; GOT, glutamic oxaloacetic transaminase; GPT, glutamic pyruvic transaminase; ALP, alkaline phosphatase; γ-GTP, gamma glutamyl transpeptidase; HUs, Hounsfield units.
Table 2.
Serial changes of baseline characteristics after 6 months according to the surgery type
Table 3.
Serial changes of baseline characteristics after surgery in 26 patients who had hepatic steatosis before surgery




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download