Abstract
Background
Postoperative nausea and vomiting (PONV), postanesthetic shivering and pain are common postoperative patient complaints that can result in adverse physical and psychological outcomes. Some antiemetics are reported to be effective in the management of postoperative pain and shivering, as well as PONV. We evaluated the efficacy of dexamethasone added to ramosetron on PONV, shivering and pain after thyroid surgery.
Methods
One hundred and eight patients scheduled for thyroid surgery were randomly allocated to three different groups: the control group (group C, n = 36), the ramosetron group (group R, n = 36), or the ramosetron plus dexamethasone group (group RD, n = 36). The patients were treated intravenously with 1 and 2 ml of 0.9% NaCl (group C); or 2 ml of 0.15 mg/ml ramosetron plus 1 ml of 0.9% NaCl (group R); or 2 ml of 0.15 mg/ml ramosetron plus 1 ml of 5 mg/ml dexamethasone (group RD) immediately after anesthesia.
Results
Incidence of nausea and the need for rescue antiemetics, verbal rating scale (VRS) 1 hour pain value, ketorolac consumption, and incidence of shivering were significantly lower in group R and group RD, than in group C (P < 0.05). Moreover, these parameters were significantly lower in group RD than in group R (P < 0.05).
Conclusions
Combination of ramosetron and dexamethasone significantly reduced not only the incidence of nausea and need for rescue antiemetics, but also the VRS 1 hour pain value, ketorolac consumption, and the incidence of shivering compared to ramosetron alone in patients undergoing thyroid surgery.
Go to : 
Postoperative nausea and vomiting (PONV), shivering, and pain are frequent and distressing to patients recovering from general anesthesia. PONV occurs in 20% to 30% general surgery patients, and in up to 70% to 80% in high risk surgical patients [1,2]. PONV after thyroidectomy is especially common and vomiting may increase the risk of postoperative bleeding, and neck hematoma. Postanesthetic shivering (PAS) occurs in the postoperative period in up to 76% of patients and varies according to age, sex, anesthetic drugs, and the type of surgery [3,4].
PAS increases tissue oxygen consumption by 200% to 500%, carbon dioxide production, blood pressure and stroke volume [5,6]. Thus, patients with already limited myocardial oxygen supply may develop further compromised myocardial function.
The antiemetic efficacy of selective serotonin 5-hydroxytryptamine type 3 (5-HT3) receptor antagonists has been well established in the prevention and treatment of PONV. Research suggests that the serotonergic system has a role in the control of PAS and pain [7,8,9].
Combinations of different classes could be a more effective treatment modality, given the multifactorial etiologies of PONV, PAS, and pain.
Ramosetron is a newly developed 5-HT3 antagonist with higher receptor affinity and a longer duration of action than older agents in its class [7,10]. Dexamethasone enhances antiemetic efficacy, as well as reduces PAS and pain [3,11,12].
Until the present time, the majority of studies testing the combined effects of ramosetron and dexamethasone solely examined the prevention of PONV [13,14,15]. Therefore, we designed a prospective, randomized, double-blinded study to compare the efficacy of ramosetron with or without dexamethasone in order to reduce postoperative pain and shivering as well as PONV in patients undergoing thyroid surgery.
Go to : 
Approval from the Institutional Review Board at our institution and written informed consent from the study patients was obtained. One hundred and eight elective thyroid surgery patients aged between 20-65 years, with American Society of Anesthesiologists classification I-II were enrolled in the study.
The exclusion criteria were as follows: history of allergy to any drugs used in the study, gastrointestinal disease, motion sickness, diabetes mellitus, obesity (body mass index ≥ 35 kg/m2), renal or hepatic functional impairment, pregnancy, menstruation, use of antiemetic drugs or a systemic steroid 24 h before surgery, and chronic pain disorder. A total of 108 patients scheduled for thyroid surgery were randomly (envelope randomization) allocated to three groups: the control group (group C, n = 36), the ramosetron group (group R, n = 36), or the ramosetron plus dexamethasone group (group RD, n = 36). Each patient received 2 syringes immediately after the induction of anesthesia. Anesthetic nurses who were not involved in the study prepared these syringes. Patients in Group C received 1 and 2 ml of 0.9% NaCl intravenously (i.v.), those in Group R received 2 ml of 0.15 mg/ml ramosetron plus 1 ml of 0.9% NaCl, those in Group RD received 2 ml of 0.15 mg/ml ramosetron plus 1 ml of 5 mg/ml dexamethasone. All patients, investigators collecting the postoperative data, and nurses involved in the postoperative care of patients were blinded to the randomization. All patients were premedicated with midazolam 2-3 mg before arriving to the operating room. Heart rate, non-invasive blood pressure, pulse oximeter, electrocardiogram, and end-tidal carbon dioxide were recorded during surgery. Tympanic temperature was measured using a Thermoscan® (infrared tympanic thermometer IRT4020, Braun, Germany) immediately before induction of anesthesia, and just before extubation. Anesthesia was induced with a slow (30-60 s) i.v. bolus dose of remifentanil 1 µg/kg, followed by propofol 1-2 mg/kg in all groups, desflurane at an initial end-tidal concentration of 1 minimum alveolar concentration in 50% oxygen/air. The desflurane concentration was titrated in response to hemodynamic changes. Desflurane and remifentanil administration were stopped at the end of the procedure. Antagonism of muscle relaxation was achieved with pyridostigmine 0.2 mg/kg and glycopyrrolate 0.008 mg/kg i.v., and when spontaneous ventilation of the patient was adequate, the endotracheal tube was subsequently extubated. After the operation, the patients were transferred to the post anesthesia care unit (PACU), post- operatively. Primary outcome measures included the incidence of nausea, vomiting, and rescue antiemetic drug use within 48 h postoperatively. Nausea was defined as a subjectively unpleasant sensation associated with an awareness of the urge to vomit; vomiting was defined as the forceful expulsion of gastric contents through the mouth. Except for the expulsion of gastric contents, retching is similar to vomiting in all aspects. Therefore, it was considered as vomiting. The severity of nausea was assessed by a verbal rating scale (VRS) with choices ranging from 0 (none) to 100 (most severe). When moderate or severe nausea (VRS score > 40) or vomiting was present, patients were asked if they required rescue antiemetics, and metoclopramide 10 mg was i.v. administered. Patients were specifically advised to call the nurse as soon as they felt any symptoms, not only of PONV, but also of any side effects, such as headache, dizziness, and drowsiness. Secondary outcome measures were the incidence of PAS, severity of postoperative pain VRS (0-100) during 1 h in PACU after surgery, and the total amount of rescue analgesic administered for 48 h post-operatively.
Shivering was defined as readily detectable fasciculation or tremors of the face, trunk, or limbs of a minimum of 10 seconds duration. PAS was treated with a forced-air warming blanket. Pethidine was not administered to treat shivering because it could affect PONV.
If patients asked for analgesics or experienced pain with VRS exceeding 40, they received a bolus of ketorolac 30 mg i.v.. Calculation of sample size was performed using power analysis and sample size calculation software for Windows 2008 (Kaysville, Utah). The degree of freedom and effect size were 2 and 0.3, respectively. A sample size of 36 patients per group was needed to demonstrate a significant difference with a power of 80% and an α- coefficient of 0.05. The results were presented as means ± SD or the number of patients (%). Group comparisons of age, body weight, height, duration of surgery, tympanic membrane temperature, VRS pain score at 1 h after surgery, and ketorolac consumption during the study period, were conducted using one-way analysis of variance (ANOVA). Post-hoc comparisons were performed by applying the Scheffe's test to the significance level. The χ2 test or Fisher's exact test was used to analyze categorical data, such as the smoking status, history of PONV, incidence of PAS, nausea, vomiting, and the use of rescue antiemetics. A P value of < 0.05 was considered statistically significant.
Go to : 
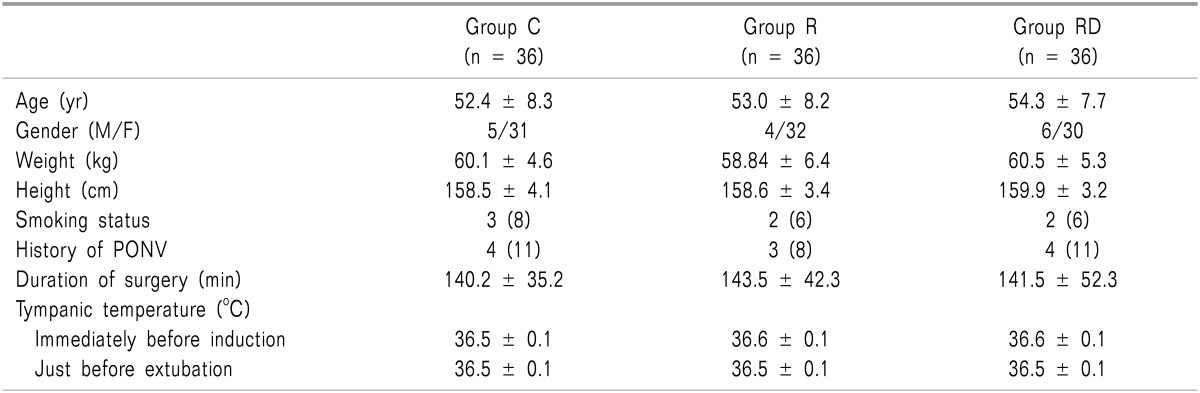
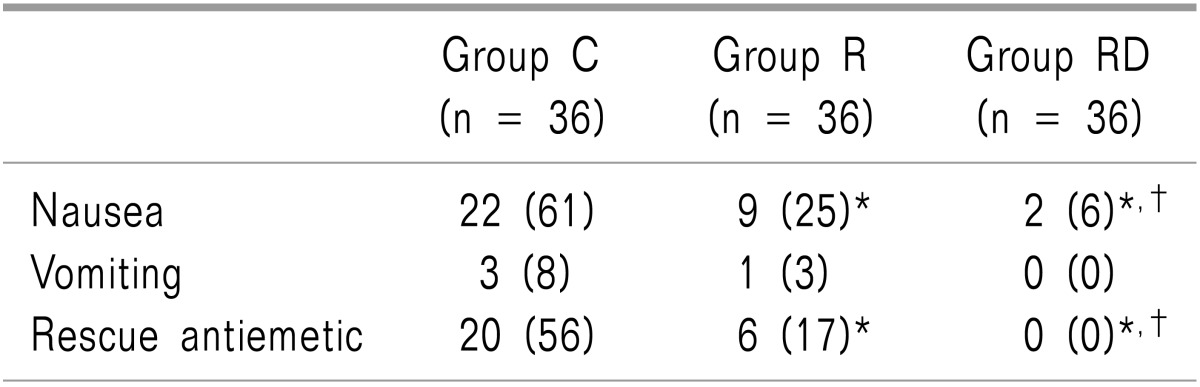
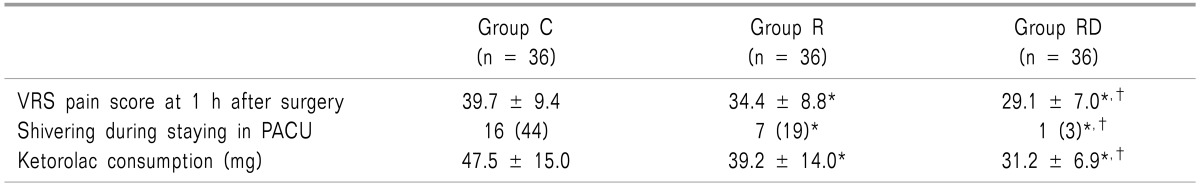
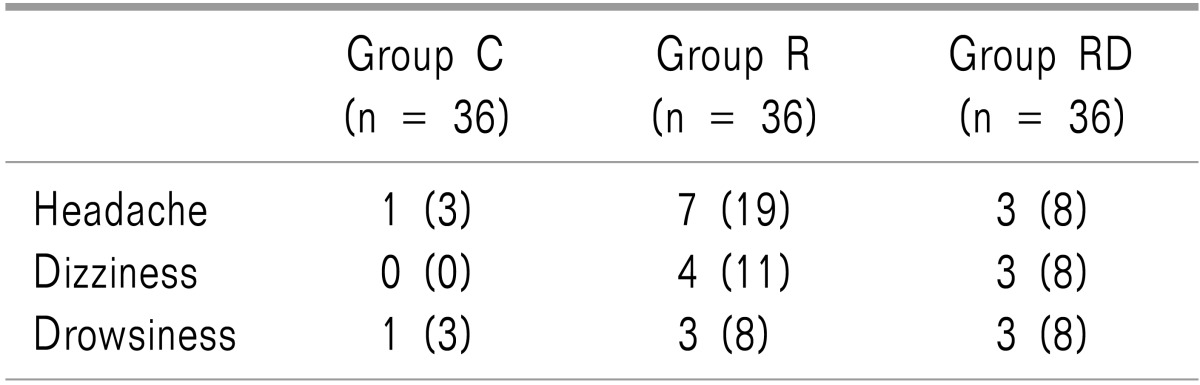
Patient characteristics including the history of PONV, tympanic temperature, as well as operative data were similar among the groups (Table 1). The incidence of nausea and the need for rescue antiemetics were significantly lower in groups R and RD than group C. However, group RD treatment was more efficacious than group R treatment (P < 0.05; Table 2). The VRS 1 hour pain value, ketorolac consumption, and incidence of shivering were significantly lower in groups R and RD, than in group C (P < 0.05). However, group RD decreased more significantly than group R (P < 0.05; Table 3). The incidence of the most common adverse events from 5-HT3 receptor antagonist, such as headache, dizziness and drowsiness were similar among the 3 groups (Table 4). There were no clinically serious adverse events attributed to dexamethasone.
Go to : 
We demonstrated that the combination of ramosetron with dexamethasone was significantly more effective in reducing the incidence of PONV, PAS, and postoperative pain, compared with ramosetron monotherapy.
PONV, PAS and pain after surgery are common postoperative patient complaints, often resulting in adverse physical and psychological outcomes.
Multifactorial etiologies of PONV have been identified. None of the currently available antiemetics are capable of completely eliminating the risk of PONV, hence it is rational to use a combination of antiemetics with different mechanisms of action. There is evidence that combination therapy acts synergistically [10,16,17,18]. The most commonly evaluated combination of antiemetics is the 5-HT3 antagonist with droperidol or dexamethasone. However, because of the potential association droperidol has with arrhythmia, the Food and Drug Administration (FDA) has warned against its use [19]. Therefore, we studied ramosetron, a new selective 5-HT3 antagonist that possesses more potent and prolonged-acting properties than the previously developed 5-HT3 receptor antagonists, such as ondanestron and granisetron plus dexamethasone.
Among the antiemetic drugs, 5-HT3 antagonists have been well-reported and are frequently used in clinical field [7,10,13,14,15]. Additionally, they have been effectively given to ease PAS [7,8,20,21]. Although its mechanism of 5-HT3 receptor antagonists is still not completely understood at this point, it is probably related to the inhibition of serotonin re-uptake in the preoptic anterior hypothalamic region. And it influences both heat production and heat loss pathways [22]. Iqbal et al. [9] compared granisetron and pethidine for the prevention of PAS. They found that use of granisetron before induction of anesthesia was effective in preventing PAS without adverse effects that are caused by pethidine, such as respiratory depression, hypotension and PONV.
It has been reported that 5-HT-3 receptors are involved in nociceptive pathways and bind to opioid µ receptors exhibiting agonist activity, resulting in a peripheral antinociceptive effect [23,24]. Descending serotonergic neurons from the rostral ventromedial medulla facilitate nociceptive signaling in models of cancer-induced bone pain, inflammatory pain, and neuropathic pain.
The effects of dexamethasone in reducing the risk of PONV, PAS and severity of postoperative pain have been documented [7,11,12,25]. The mechanisms for the improved effects of combination therapy of dexamethasone have not been elucidated, but the suggested mechanisms are due to dexamethasone's strong anti-inflammatory effects on neck structures by prostaglandin synthesis inhibition and a decrease in serotonin turnover in the central nervous system. It may thus reduce the ascending parasympathetic impulses to the vomiting center of the surgical sites [26,27]; the suppression of release of inflammatory mediators that induce hyperalgesia, such as tumor necrosis factor-α, interleukin-1β, and interleukin-6 [28]; and decrease the temperature gradient and shivering by inhibition of release of vasoconstrictor and pyrogenic cytokines [29]. Moreover, significant postoperative beneficial effects minus serious adverse effects, from single-dose i.v. dexamethasone, such as reducing fatigue, duration of convalescence, and vocal dysfunction which is an important issue for patients undergoing thyroid surgery have also been shown [11,30]. Therefore, it seems reasonable to add dexamethasone when we use i.v. ramosetron before thyroid surgery.
There are some limitations to this study. Firstly, we did not collect PONV data separately during the 48 hours' time interval, so we could not identify early and late effects, respectively. Secondly, postoperative VRS pain score was assessed for only 1 hour after surgery, hence the effect beyond 1 hour remained undetermined. Further well-designed studies are warranted to evaluate the early and late effect of ramosetron and dexamethasone combination on PONV, and postoperative pain.
We concluded that the combination of ramosetron and dexamethasone after thyroid surgery was a simple, safe, and effective method to reduce the incidence of PONV, PAS, and postoperative pain, as compared to ramosetron alone.
Go to : 
ACKNOWLEDGEMENTS
This paper was written as part of Konkuk University's research support program for its faculty on sabbatical leave in 2013.
Go to : 
References
1. Apfel CC, Läärä E, Koivuranta M, Greim CA, Roewer N. A simplified risk score for predicting postoperative nausea and vomiting: conclusions from cross-validations between two centers. Anesthesiology. 1999; 91:693–700. PMID: 10485781.
2. López-Olaondo L, Carrascosa F, Pueyo FJ, Monedero P, Busto N, Sáez A. Combination of ondansetron and dexamethasone in the prophylaxis of postoperative nausea and vomiting. Br J Anaesth. 1996; 76:835–840. PMID: 8679359.

3. Yared JP, Starr NJ, Hoffmann-Hogg L, Bashour CA, Insler SR, O'Connor M, et al. Dexamethasone decreases the incidence of shivering after cardiac surgery: a randomized, double-blind, placebo-controlled study. Anesth Analg. 1998; 87:795–799. PMID: 9768772.

4. Eberhart LH, Döderlein F, Eisenhardt G, Kranke P, Sessler DI, Torossian A, et al. Independent risk factors for postoperative shivering. Anesth Analg. 2005; 101:1849–1857. PMID: 16301273.

5. Zwischenberger JB, Kirsh MM, Dechert RE, Arnold DK, Bartlett RH. Suppression of shivering decreases oxygen consumption and improves hemodynamic stability during postoperative rewarming. Ann Thorac Surg. 1987; 43:428–431. PMID: 3105477.

6. Macintyre PE, Pavlin EG, Dwersteg JF. Effect of meperidine on oxygen consumption, carbon dioxide production, and respiratory gas exchange in postanesthesia shivering. Anesth Analg. 1987; 66:751–755. PMID: 3111305.

7. Song YK, Lee C. Effects of ramosetron and dexamethasone on postoperative nausea, vomiting, pain, and shivering in female patients undergoing thyroid surgery. J Anesth. 2013; 27:29–34. PMID: 22965329.

8. Asl ME, Isazadefar K, Mohammadian A, Khoshbaten M. Ondansetron and meperidine prevent postoperative shivering after general anesthesia. Middle East J Anaesthesiol. 2011; 21:67–70. PMID: 21991735.
9. Iqbal A, Ahmed A, Rudra A, Wankhede RG, Sengupta S, Das T, et al. Prophylactic granisetron vs pethidine for the prevention of postoperative shivering: a randomized control trial. Indian J Anaesth. 2009; 53:330–334. PMID: 20640142.
10. Yang SY, Jun NH, Choi YS, Kim JC, Shim JK, Ha SH, et al. Efficacy of dexamethasone added to ramosetron for preventing postoperative nausea and vomiting in highly susceptible patients following spine surgery. Korean J Anesthesiol. 2012; 62:260–265. PMID: 22474554.

11. Feroci F, Rettori M, Borrelli A, Lenzi E, Ottaviano A, Scatizzi M. Dexamethasone prophylaxis before thyroidectomy to reduce postoperative nausea, pain, and vocal dysfunction: a randomized clinical controlled trial. Head Neck. 2011; 33:840–846. PMID: 20737495.

12. Jakobsson J. Preoperative single-dose intravenous dexamethasone during ambulatory surgery: update around the benefit versus risk. Curr Opin Anaesthesiol. 2010; 23:682–686. PMID: 20847689.

13. Jeon Y, Kim H, Kwak KH. Comparison of ramosetron, dexamethasone, and a combination of ramosetron and dexamethasone for the prevention of postoperative nausea and vomiting in Korean women undergoing thyroidectomy: a double-blind, randomized, controlled study. Curr Ther Res Clin Exp. 2010; 71:78–88. PMID: 24683252.

14. Jo YY, Lee JW, Shim JK, Lee WK, Choi YS. Ramosetron, dexamethasone, and their combination for the prevention of postoperative nausea and vomiting in women undergoing laparoscopic cholecystectomy. Surg Endosc. 2012; 26:2306–2311. PMID: 22358123.

15. Ryu JH, Chang JE, Kim HR, Hwang JW, Oh AY, Do SH. Ramosetron vs. ramosetron plus dexamethasone for the prevention of postoperative nausea and vomiting (PONV) after laparoscopic cholecystectomy: prospective, randomized, and double-blind study. Int J Surg. 2013; 11:183–187. PMID: 23313969.

16. Sanchez-Ledesma MJ, López-Olaondo L, Pueyo FJ, Carrascosa F, Ortega A. A comparison of three antiemetic combinations for the prevention of postoperative nausea and vomiting. Anesth Analg. 2002; 95:1590–1595. PMID: 12456422.

17. Kim EJ, Ko JS, Kim CS, Lee SM, Choi DH. Combination of antiemetics for the prevention of postoperative nausea and vomiting in high-risk patients. J Korean Med Sci. 2007; 22:878–882. PMID: 17982239.

18. Heffernan AM, Rowbotham DJ. Postoperative nausea and vomiting--time for balanced antiemesis? Br J Anaesth. 2000; 85:675–677. PMID: 11094578.
19. Habib AS, Gan TJ. Food and drug administration black box warning on the perioperative use of droperidol: a review of the cases. Anesth Analg. 2003; 96:1377–1379. PMID: 12707137.

20. Powell RM, Buggy DJ. Ondansetron given before induction of anesthesia reduces shivering after general anesthesia. Anesth Analg. 2000; 90:1423–1427. PMID: 10825334.

21. Kim MS, Kim DW, Woo SH, Yon JH, Lee S. Effect of ramosetron on shivering during spinal anesthesia. Korean J Anesthesiol. 2010; 58:256–259. PMID: 20498774.

22. Alfonsi P. Postanaesthetic shivering: epidemiology, pathophysiology, and approaches to prevention and management. Drugs. 2001; 61:2193–2205. PMID: 11772130.
23. Memiş D, Turan A, Karamanlioğ B, Süt N, Pamukçu Z. The prevention of pain from injection of rocuronium by ondansetron, lidocaine, tramadol, and fentanyl. Anesth Analg. 2002; 94:1517–1520. PMID: 12032018.

24. Memiş D, Turan A, Karamanlioglu B, Kaya G, Pamukçu Z. The prevention of propofol injection pain by tramadol or ondansetron. Eur J Anaesthesiol. 2002; 19:47–51. PMID: 11913803.

25. Hval K, Thagaard KS, Schlichting E, Raeder J. The prolonged postoperative analgesic effect when dexamethasone is added to a nonsteroidal antiinflammatory drug (rofecoxib) before breast surgery. Anesth Analg. 2007; 105:481–486. PMID: 17646509.

26. Fredrikson M, Hursti T, Fürst CJ, Steineck G, Börjeson S, Wikblom M, et al. Nausea in cancer chemotherapy is inversely related to urinary cortisol excretion. Br J Cancer. 1992; 65:779–780. PMID: 1586608.

27. Aapro MS, Plezia PM, Alberts DS, Graham V, Jones SE, Surwit EA, et al. Double-blind crossover study of the antiemetic efficacy of high-dose dexamethasone versus high-dose metoclopramide. J Clin Oncol. 1984; 2:466–471. PMID: 6539363.

28. Sapolsky RM, Romero LM, Munck AU. How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocr Rev. 2000; 21:55–89. PMID: 10696570.

29. Jansen NJ, van Oeveren W, van den Broek L, Oudemansvan Straaten HM, Stoutenbeek CP, Joen MC, et al. Inhibition by dexamethasone of the reperfusion phenomena in cardiopulmonary bypass. J Thorac Cardiovasc Surg. 1991; 102:515–525. PMID: 1656149.

30. Sánchez-Rodríguez PE, Fuentes-Orozco C, González-Ojeda A. Effect of dexamethasone on postoperative symptoms in patients undergoing elective laparoscopic cholecystectomy: randomized clinical trial. World J Surg. 2010; 34:895–900. PMID: 20151129.

Go to : 




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download