Abstract
Stevens-Johnson syndrome (SJS) is a rare but life-threatening skin reaction disease and carbamazepine is one of its most common causes. We report a case of SJS secondary to carbamazepine in a patient with previous pruritus due to carbamazepine which was given for treatment of trigeminal neuralgia. We would like to caution all providers that carbamazepine readministration should be avoided in the patient with a previous history of SJS or adverse skin reaction. In addition, we strongly recommend gradual titration when initiating treatment with carbamazepine.
Stevens-Johnson syndrome (SJS), defined by widespread blisters arising in macules and/or flat atypical targets-shaped lesions, are diseases with homogenous clinical characteristics and a potentially lethal outcome [1]. SJS is usually associated with some types of anticonvulsants, including carbamazepine, lamotarigine, phenobarbotal, phenytoin and valproic acid [2]. Clinically, these diseases present as erythema, necrosis, and extensive sloughing of the epidermis; mucous involvement; and systemic symptoms.
The pathogenesis of these diseases has not yet been established. Dysregulation of the immunologic reaction is thought to be one of the most important causes. The death of keratinocytes due to apoptosis is currently thought to be the major mechanism [3]. SJS can be characterized as a hypersensitivity syndrome because of the preexistence of pharmacogenetic and immunologic abnormalities to the administered drug. It is quite difficult to prevent SJS because drug hypersensitivity reactions occur in an unpredictable manner.
Carbamazepine, which is widely used to treat seizure disorder, bipolar disorder, trigeminal neuralgia, and chronic pain, is one of most common causes of drug hypersensitivity reactions [4]. The reported frequency of serious carbamazepine hypersensitivity reaction is between 1/1,000 and 1/10,000 new exposures to the drug [5].
The object of this article was to report a case of SJS secondary to carbamazepine in a patient with trigeminal neuralgia.
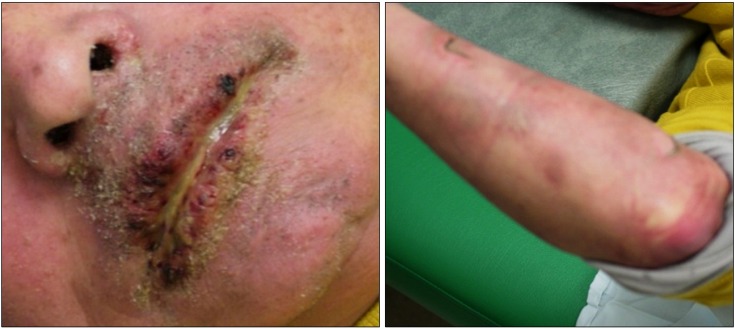
A 68-year-old male presented with left mandibular area pain due to third trigeminal neuralgia. He had underdone conventional radiofrequency lesioning in the left third branch of the trigeminal nerve for trigeminal neuralgia one year previously. Following the procedure he had been without pain until quite recently. However, at the time of his presentation, he was reporting pain in the same area of 40/100 using the VAS (Visual Analogue Scale) score. We decided to proceed with medical treatment including carbamazepine 200 mg three times a day, tramadol (37.5 mg)/acetaminophen (325 mg) combination three times a day, and nortriptyline 10 mg one time a day because his pain was mild. On his second day of medication, a mild fever and general weakness had occurred. On his fourth day after beginning the medication, an oral rash and bullae on the cheek, neck, forearm and leg were found. On the fifth day, the patient visited the emergency room and was admitted with a presumptive diagnosis of SJS. Further history revealed that he had discontinued medication including carbamazepine 200 mg one year previously on his own due to pruritus after taking the medication once. The medical history was otherwise unremarkable. On physical examination, the patient displayed pruritic and stinging erythema and painful crursted erosions on both lips, with several flaccid and ruptured bullae on the left cheek, neck, forearm, and leg (Fig. 1). Pharyngitis was present but he was able to swallow some food. Eye discomfort and genital mucosa involvement were not present. Nikolsky's sign was positive. Laboratory examination revealed mild leukocytosis, and alanine aminotransferase, C-reactive protein, and potassium were mildly elevated. Carbamazepine was withdrawn and prednisolone 125 mg/day was started. A skin biopsy performed on the third day after admission, demonstrated subepidermal bullae formation with epidermal necrosis and minimal perivascular lymphohistocytic infiltration in the upper dermis and the correct diagnosis of SJS was established. Steroid treatment was given for 5 days and gradually withdrawn. On the eighth day the patient was discharged in good condition, with generalized desquamation and incomplete peeling of the skin on the neck, forearm, and face. Two weeks after discharge, he underwent conventional radiofrequency lesioning of the left third branch of the trigeminal nerve. Two weeks later, his pain was nearly gone.
Trigeminal neuralgia involves severe, lancinating pain, triggered by non-nociceptive stimuli, in the distribution of one or more divisions of the trigeminal nerve. The primary medications used to treat the painful symptoms of trigeminal neuralgia are anticonvulsants. The first-line treatment is carbamazepine [6].
Carbamazepine has been strongly associated with SJS. Although SJS has multiple etiologies, it is commonly triggered by viral infections (herpes simplex virus is the infectious agent more commonly involved) and neoplasias (carcinomas and lymphomas). However, the most common cause is the use of medications. Among the drugs implicated more often are allopurinol, antibiotics, anticonvulsants, and non-steroid anti-inflammatories [7]. Recently, in a seven-year study, Devi et al. concluded that anticonvulsants were the cause implicated most in SJS especially in the first eight weeks of treatment, and the main drug responsible (more than 80%) was carbamazepine [8]. The increased number of prescriptions of carbamazepine for the control of pain may be the reason for the increased frequency of SJS due to carbamazepine. An additional article reported on the association between SJS and analgesics. The authors observed an association for acetaminophen (paracetamol) with a relative risk of 1.2. In addition, tramadol showed a high relative risk, but there was also a high percentage of co-medication of the drug with other highly suspected drugs (57%), suggesting potential confounding by co-medication [9].
At present, the mechanisms by which SJS develops are not well understood. SJS usually occurs during the first course of drug ingestion (without prior sensitization). T-cells are already present in the body before drug exposure and elicit a robust immune reaction on carbamazepine antigenic stimulation [10].
Typically, the initial presentation is marked by symptoms of fever, myalgia, and general weakness for 1 to 3 days before the development of cutaneous lesions. The skin lesions are symmetrically distributed on the face and upper trunk areas. The rash spreads rapidly and is usually maximal within four days, sometimes within hours. The initial skin lesions are usually poorly defined macules with darker purpuric centers that coalesce [11].
Diagnosis is clinical. However, skin biopsy helps confirm the diagnosis, usually excluding bullous diseases not related to drug therapy. Early on, there is full-thickness epidermal necrosis and detachment, with an only slightly altered underlying dermis. Immunofluorescence studies only help exclude other bullous disease. Anemia and lymphopenia are frequent, but eosinophilia is rare. Neutropenia suggests a poor prognosis [11].
The main therapeutic action in SJS is early recognition of the drug reaction and withdrawal of the drug, since any delay can be seriously deleterious to the patient. There is no universally accepted, definitively effective, specific treatment for acute SJS other than supportive care. Glucocorticoids have not been proven useful in SJS. They may be beneficial in the early stage of SJS but may increase the risk of infection, prolong wound healing, or even increase mortality, if bullous eruption or mucosal erosion is fully developed. Although some retrospective studies have suggested that intravenous immunoglobulin may be effective in stopping the progression of SJS, other studies showed limited benefit on the mortality rate or progression of the disease [7].
The patient in this case was given the same drug (carbamazepine) twice, but the degree of his cutaneous reaction was greater with the second exposure, when he developed SJS. On his first exposure to the medication, he took one dose of carbamazepine 200 mg. The second time, he took carbamazepine 600 mg a day for 5 days. We assumed that SJS did not occur on the first administration of carbamazepine because the patient stopped taking the medication after a single dose, which is not enough dosage to trigger hypersensitivity reaction. Huang et al. also reported a case of fetal toxic epidermal necrolysis induced by carbamazepine treatment in a patient who previously had carbamazepine-induced SJS [12]. It has been reported that higher daily doses of some drugs are associated with an increased risk of SJS compared to lower doses, as is the case for allopurinol [13]. However there is as yet no evidence about the relationship between carbamazepine dosage and SJS.
It is important to note that carbamazepine readministration should be avoided in patients with a previous history of SJS or adverse skin reaction to carbamazepine through the obtaining of an accurate medical history. In addition we advise a gradual titration at the start of treatment with anticonvulsant.
References
1. Auquier-Dunant A, Mockenhaupt M, Naldi L, Correia O, Schröder W, Roujeau JC, et al. Correlations between clinical patterns and causes of erythema multiforme majus, Stevens-Johnson syndrome, and toxic epidermal necrolysis: results of an international prospective study. Arch Dermatol. 2002; 138:1019–1024. PMID: 12164739.
2. Mockenhaupt M, Messenheimer J, Tennis P, Schlingmann J. Risk of Stevens-Johnson syndrome and toxic epidermal necrolysis in new users of antiepileptics. Neurology. 2005; 64:1134–1138. PMID: 15824335.

3. Paul C, Wolkenstein P, Adle H, Wechsler J, Garchon HJ, Revuz J, et al. Apoptosis as a mechanism of keratinocyte death in toxic epidermal necrolysis. Br J Dermatol. 1996; 134:710–714. PMID: 8733377.

4. Leeder JS. Mechanisms of idiosyncratic hypersensitivity reactions to antiepileptic drugs. Epilepsia. 1998; 39(Suppl 7):S8–S16. PMID: 9798756.

5. Tennis P, Stern RS. Risk of serious cutaneous disorders after initiation of use of phenytoin, carbamazepine, or sodium valproate: a record linkage study. Neurology. 1997; 49:542–546. PMID: 9270593.

6. Larsen A, Piepgras D, Chyatte D, Rizzolo D. Trigeminal neuralgia: diagnosis and medical and surgical management. JAAPA. 2011; 24:20–25. PMID: 21748954.
7. Letko E, Papaliodis DN, Papaliodis GN, Daoud YJ, Ahmed AR, Foster CS. Stevens-Johnson syndrome and toxic epidermal necrolysis: a review of the literature. Ann Allergy Asthma Immunol. 2005; 94:419–436. PMID: 15875523.

8. Devi K, George S, Criton S, Suja V, Sridevi PK. Carbamazepine--the commonest cause of toxic epidermal necrolysis and Stevens-Johnson syndrome: a study of 7 years. Indian J Dermatol Venereol Leprol. 2005; 71:325–328. PMID: 16394456.

9. Mockenhaupt M, Viboud C, Dunant A, Naldi L, Halevy S, Bouwes Bavinck JN, et al. Stevens-Johnson syndrome and toxic epidermal necrolysis: assessment of medication risks with emphasis on recently marketed drugs. The EuroSCAR-study. J Invest Dermatol. 2008; 128:35–44. PMID: 17805350.

10. Wu Y, Sanderson JP, Farrell J, Drummond NS, Hanson A, Bowkett E, et al. Activation of T cells by carbamazepine and carbamazepine metabolites. J Allergy Clin Immunol. 2006; 118:233–241. PMID: 16815161.

11. Roujeau JC, Stern RS. Severe adverse cutaneous reactions to drugs. N Engl J Med. 1994; 331:1272–1285. PMID: 7794310.

12. Huang LY, Liao WC, Chiou CC, Lou JP, Hu P, Ko FC. Fatal toxic epidermal necrolysis induced by carbamazepine treatment in a patient who previously had carbamazepine-induced Stevens-Johnson syndrome. J Formos Med Assoc. 2007; 106:1032–1037. PMID: 18194909.

13. Halevy S, Ghislain PD, Mockenhaupt M, Fagot JP, Bouwes Bavinck JN, Sidoroff A, et al. Allopurinol is the most common cause of Stevens-Johnson syndrome and toxic epidermal necrolysis in Europe and Israel. J Am Acad Dermatol. 2008; 58:25–32. PMID: 17919772.





 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download