Abstract
During the early stage of postherpetic neuralgia, an epidural block on the affected segment is helpful in controlling pain and preventing progression to a chronic state. The main neurologic complication following an epidural block is cord compression symptom due to an epidural hematoma. When neurologic complications arise from an epidural block for the treatment of postherpetic neuralgia, it is important to determine whether the complications are due to the procedure or due to the herpes zoster itself. We report a case of a patient who was diagnosed with herpes zoster myelitis during treatment for postherpetic neuralgia. The patient complained of motor weakness in the lower extremities after receiving a thoracic epidural block six times. Although initially, we believed that the complications were due to the epidural block, it was ultimately determined to be from the herpes zoster myelitis.
Go to : 
Postherpetic neuralgia can be treated with local anesthetics, antidepressants, antiepileptics, and epidural block on the affected dermatomes [1]. Although herpes zoster most commonly occurs along the thoracic distribution (51.2%), deterioration of motor function is a very rare complication (0.3%). Additionally, the deterioration of motor function in the thoracic distribution is less common than deterioration in the head, neck or lumbrosacral region [2]. In contrast, there are many neurologic complications resulting from an epidural block, in particular, an epidural hematoma causing spinal cord compression which results in a decrease in motor function. When neurologic complications develop after performing an epidural block on patients with postherpetic neuralgia, it is important to differentiate the complications caused by either the epidural block or the herpes zoster itself. We report a case of a patient with newly developed urinary retention and motor weakness of lower extremities during treatment for postherpetic neuralgia.
One month before visiting our pain clinic, a 68-year-old female patient with vesicles and pain on the left side around the navel was diagnosed with herpes zoster and received treatment at a local hospital. Although the skin rash improved, the patient, at times, felt squeezing sensations on her left flank. When the patient was referred to the pain clinic, the patient had a squeezing pain and scars of vesicles on the T10 dermatome level on the left side and her numeric rating scale (NRS) was 5 (out of 10). She felt the squeezing pain several times for approximately one minute each time. Aside from a history of hypertension, there was no other unusual medical history or a history of prior surgeries. She was 152 cm tall and 51.2 kg in weight. Laboratory examinations showed no signs of bacteria or viral infection. Complete blood cell count, prothrombin time and activated partial thrombin time tests showed normal results, and there were no abnormal results in the serologic tests (HBs Ag/Ab, HIV Ag/Ab, quantitive VDRL).
After diagnosis of postherpetic neuralgia, the patient received an epidural block once a week for a total of six weeks. We performed the epidural block with 8 ml of 0.8% mepivacaine with the paramedian approach to T10-T11 with the 'loss of resistance' method. Out of six total procedures, only the first procedure included 20 mg of triamcinolone with local anesthetics, and the rest were composed of local anesthetics only. Any signs or symptoms suggestive of dural puncture, such as cerebrospinal fluid (CSF) aspiration through the block needle, headache, paresthesia or sensory impairment were not observed during each epidural block. Pregabalin 150 mg and amitriptyline 5 mg were also prescribed for twice a day. After each epidural block, a pain free period continued for two or three days but she felt numbness like prior to the procedure. Additionally, the squeezing pain reappeared but the pain intensity was reduced to NRS 3. Thus, we expected full recovery by repetitive epidural blocks and by an additional root block or transforaminal epidural block.
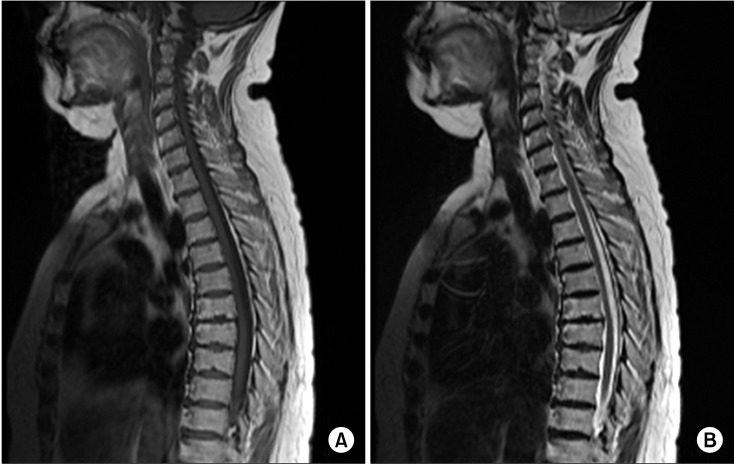
Two days after the sixth epidural block, the patient was admitted to the department of neurology, through the emergency department, because the patient felt continuous dizziness and motor weakness in both lower extremities. Five days prior to receiving the sixth epidural block, the patient began to feel slightly dizzy. However, the continual dizziness became extreme by the time the patient was admitted to the hospital. Motor weakness of the lower extremities was felt by the patient two days after the final epidural block. In addition, the patient had symptoms of nausea and vomiting. The patient also could not stand or walk alone. The patient's body temperature was 38.3℃ and she did not have any abnormalities on her skin. Physical examination showed a grade III motor weakness in both of the lower extremities, but the deep tendon reflex and cerebellar function tests were normal. Sensory impairment was not observed. She was referred to our pain clinic, because a complication during an epidural block was considered by neurologists. We reviewed the sixth epidural block, examining for any evidence of dural puncture, aspiration of blood and other abnormal events, but there were none. However, in order to be certain, we performed Magnetic Resonance Imaging (MRI) and CSF tests on the first day of admission. The MRI showed no signs of hematoma, abscess, or arachnoiditis (Fig. 1). No bacteria were detected upon CSF analysis. Considering the results, we were able to exclude the possibility of an epidural block complication. The CSF analysis results showed, however, a mild increase in the WBC count (49,000 count/mm3) and a protein level of 97 mg/dl. Virus serology test of the blood and the CSF showed negative anti-IgG and negative anti-IgM antibodies for herpes simplex virus (HSV) and herpes zoster. Two days after the patient was admitted, the patient complained of urinary difficulty and a Foley catheter was inserted, resulting in discharge of 1,200 ml of urine through the catheter. The patient felt squeezing pain on the T10 dermatome level on the left side during her entire stay at the hospital.
Considering her clinical progressions and the increase in protein level in the CSF analysis, herpes zoster myelitis was diagnosed by the neurologist and appropriate treatments commenced. Three days after the patient was admitted, the patient was given intravenous acyclovir 1.6 g and dexamethasone 20 mg daily for two weeks. The patient also received exercise rehabilitation treatment while she was on the medication. After receiving treatment for one week, the patient was able to walk but had difficulty in standing up. The patient showed gradual improvement and four weeks later made a full recovery in motor function. The patient's urinary difficulty improved through urinary training and she fully recovered in eight weeks.
Go to : 
Herpes zoster infection can cause various neurological complications such as postherpetic neuralgia, polyradiculoneuritis, transverse myelitis, vasculopathy, aseptic meningitis, leukoencephalopathy, dorsal root or cranial nerve ganglionitis, ventriculitis, necrotizing angiitis, meningoencephalitis, and so on [2]. Among these, herpes zoster myelitis is rare and appears at a frequency of approximately 0.3% [3]. The pathogenesis of herpes zoster myelitis is still not clearly understood but it is suspected that the virus affects the central nervous system directly or destroys the spinal nerve through an immune reaction mechanism [4]. Generally, the onset of herpes zoster myelitis is subacute and it takes more or less 8 days to 10 weeks from the time the skin rash appears to develop into a full clinical myelopathy [4,5]. Typical symptoms of this disease include paresthesia at a certain affected level, paraparesis, quadriparesis and impaired sphincter function. Although there are no standard diagnosis for herpes zoster myelitis, a past history of skin lesion in addition to myelopathy signs without supraspinal lesions is usually a possible indication of herpes zoster myelitis [6]. There have been reports indicating WBCs increase in the CSF findings in herpes zoster myelitis. Mild CSF pleocytosis and an elevated protein level associated with acute urinary retention suggest a spinal cord lesion, as already proposed by previous studies [7]. Although herpes zoster myelitis can be confirmed by an increase in herpes zoster antibodies in the serum or CSF, there have been reports of herpes zoster myelitis in situations in which there were no signs of herpes zoster antibodies [5,8]. When there is an increase in the signal intensity on the spinal cord in the T2 weighted image of an MRI, it is a strong indication of the diagnosis [9]. However, there have been documented cases of myelitis showing normal findings on the MRI [10]. Although it could be misdiagnosed as other spinal diseases when symptoms occur without prior skin lesions, herpes zoster antibody test or lesion localization using electromyogram can be useful [4]. In our case, the diagnosis of herpes zoster myelitis was inferred by a neurologist based on the presence of skin lesions, the onset time of neurologic symptoms, deterioration of the lower extremities motion, urinary retention and an increase in protein on the CSF analysis.
Clinicians often categorize patients with dizziness as those with peripheral vestibular disease versus those with central vestibular disease. Including migraines, central vestibular abnormality due to head trauma or infection, brain stem stroke and vertebrobasilar insufficiency, and cerebellar degeneration; these four disease entities can affect the central vestibular system [11]. In our case, dizziness with nausea and vomiting could be caused by a central vestibular system abnormality due to zoster virus infection.
Early treatment using antiviral agents, particularly acyclovir has proven to be effective for a better prognosis and steroids can also be used as a treatment. Patients normally begin to recover two weeks after the worst episode of neurologic complications. Although recovery of muscular strength is generally good, only partial recovery of sensory deficits is reported in several cases [12]. The fact that the patient's symptoms improved with the anti-viral treatment and steroid therapy supports a diagnosis of herpes zoster myelitis in our case.
There are several complications after an epidural block causing neurologic symptoms, including cord compression and arachidonitis. It is important to rule out spinal cord compression due to epidural abscess or hematoma, and intrinsic bacterial or fungal infections of the spinal cord [13]. Spinal tap, computerized tomography, MRI, myelography and blood tests may help in narrowing the diagnostic possibilities. In our case, we were able to exclude spinal cord compression because there was no evidence of bacterial infection in the CSF and blood test, and the MRI showed no abnormal findings. Arachnoiditis should also be considered as only one cause of neurologic symptoms after an epidural block. In this case, arachnoiditis can be excluded from a list of differential diagnoses because the neurologic complications caused by the arachnoiditis normally have a slow progression. In addition, in the case of arachnoiditis causing motor deficit, the abnormal configurations of nerve roots are detectable. Arachnoiditis has been treated with medications, physiotherapy, surgical dissection of scar tissue from the cauda equina, transcutaneous nerve stimulation, and injection of corticosteroids, but frequently with little benefit [14]. In our case, we could not find any abnormal findings on the MRI, and the patient showed good response to steroid therapy and antiviral agents.
Postherpetic neuralgia is one of the common diseases seen in pain clinics and an epidural block is used frequently as a treatment modality of postherpetic neuralgia. Because neurologic complications such as deterioration of motor function caused by herpes zoster are very rare, it is likely to be mistaken as a complication from the epidural block. In this case, the possibility of a complication due to the block was initially considered because the motor function deteriorated during the treatment. We believe that if herpes zoster myelitis is not considered as one of the possibilities among other differential diagnoses causing such neurological symptoms, a chance for treatment could be lost.
In conclusion, when neurologic complications develop after an epidural block in patients with postherpetic neuralgia, the possibility that herpes zoster myelitis as well as a complication caused by the epidural block intervention should both be considered, resulting in prompt examination and treatment.
Go to : 
References
1. Kost RG, Straus SE. Postherpetic neuralgia--pathogenesis, treatment, and prevention. N Engl J Med. 1996; 335:32–42. PMID: 8637540.

2. Mueller NH, Gilden DH, Cohrs RJ, Mahalingam R, Nagel MA. Varicella zoster virus infection: clinical features, molecular pathogenesis of disease, and latency. Neurol Clin. 2008; 26:675–697. PMID: 18657721.

3. de Silva SM, Mark AS, Gilden DH, Mahalingam R, Balish M, Sandbrink F, et al. Zoster myelitis: improvement with antiviral therapy in two cases. Neurology. 1996; 47:929–931. PMID: 8857721.

4. Yang IS, Lim GD, Shin SS. A case of herpes zoster myelitis - a case report. J Korean Pain Soc. 1997; 10:266–269.
5. Boiardi A, Ferrante P, Porta E, Sghirlanzoni A, Bussone G. Herpes zoster myelitis: nervous system complications. Ital J Neurol Sci. 1986; 7:617–622. PMID: 3804716.

6. Oda Y, Terai T, Yukioka H, Fujimori M. A case of herpes zoster myelitis occurring during epidural analgesia. Anesth Analg. 1995; 81:646–648. PMID: 7653839.

7. Gibbon N. A case of herpes zoster with involvement of the urinary bladder. Br J Urol. 1956; 28:417–421. PMID: 13383174.
8. Schoenhuber R, Bortolotti P, Panzetti P, Guerzoni MC, Colombo A. Lumbosacral herpes zoster myelitis. Ital J Neurol Sci. 1986; 7:541–543. PMID: 3804709.

9. Baik JS, Kim WC, Heo JH, Zheng HY. Recurrent herpes zoster myelitis. J Korean Med Sci. 1997; 12:360–363. PMID: 9288637.

10. Baethge BA, King JW, Husain F, Embree LJ. Herpes zoster myelitis occurring during treatment for systemic lupus erythematosus. Am J Med Sci. 1989; 298:264–266. PMID: 2801761.

11. Furman JM, Whitney SL. Central causes of dizziness. Phys Ther. 2000; 80:179–187. PMID: 10654064.

12. Ong OL, Churchyard AC, New PW. The importance of early diagnosis of herpes zoster myelitis. Med J Aust. 2010; 193:546–547. PMID: 21034391.

13. Cameron CM, Scott DA, McDonald WM, Davies MJ. A review of neuraxial epidural morbidity: experience of more than 8,000 cases at a single teaching hospital. Anesthesiology. 2007; 106:997–1002. PMID: 17457132.
14. Haisa T, Todo T, Mitsui I, Kondo T. Lumbar adhesive arachnoiditis following attempted epidural anesthesia--case report. Neurol Med Chir (Tokyo). 1995; 35:107–109. PMID: 7753309.
Go to : 




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download