This article has been
cited by other articles in ScienceCentral.
Abstract
Background
Nefopam is a centrally acting analgesic that is used to control pain. The aim of this study was to find an appropriate dose of nefopam that demonstrates an analgesic effect when administered in continuous infusion with fentanyl at the end of laparoscopic cholecystectomy.
Methods
Ninety patients scheduled for laparoscopic cholecystectomy were randomly assigned to receive analgesia with fentanyl alone (50 µg, Group 1, n = 30), or with fentanyl in combination with nefopam 20 mg (Group 2, n = 30) or in combination with nefopam 40 mg (Group 3, n = 30) at the end of surgery. Pain and side effects were evaluated at 10 minutes, 30 minutes, 1 hour, 2 hours, 6 hours, and 12 hours after arrival in the post-anesthesia care unit (PACU).
Results
Pain was statistically significantly lower in Groups 2 and 3 than in Group 1 at 10 minutes, 2 hours, and 6 hours after arrival in the PACU. Nausea was statistically significantly lower in Group 2 than in Groups 1 and 3 at 10 minutes after arrival in the PACU. Shivering was statistically significantly lower in Groups 2 and 3 than in Group 1 at 10 minutes after arrival in the PACU.
Conclusions
Nefopam is a drug that can be safely used as an analgesic after surgery, and its side effects can be reduced when fentanyl 50 µg is injected with nefopam 20 mg.
Go to :

Keywords: fentanyl, laparoscopic cholecystectomy, nefopam, pain
INTRODUCTION
In the early 1970s, nefopam was used as a myorelaxant to treat spasticity [
1]. Although the drug's analgesic properties have been identified since then, its mechanism of analgesia has not been completely elucidated. The mechanism discovered to date involves controlling the reuptake of serotonin, norepinephrine, and dopamine as a centrally acting, non-opioid analgesic [
2,
3].
There have been dozens of studies that have examined various methods to control pain after surgery, and there have been attempts to induce synergistic effects by mixing drugs [
4-
6]. The advantages of combination treatment include reducing the side effects and improving the quality of analgesia by decreasing the dose of each drug [
4]. A mixture of non-steroidal anti-inflammatory drugs (NSAIDs), which work on the arachidonic acid cascade in peripheral sites, and opioids, which work centrally on specific receptors, has been effectively used in a variety of surgeries [
7,
8]. However, the use of NSAIDs is not desirable in patients with underlying hepatic or renal failure or in patients with a high risk of renal or hemorrhagic complications. In contrast, nefopam can be safely used as an analgesic adjuvant with opioids because nefopam neither alters renal or hepatic function nor causes hemorrhagic complications at usual doses [
9,
10].
Nefopam has been widely used in many countries, although there is limited clinical data on its use in the treatment of acute and chronic pain. Studies comparing nefopam and opioids reported that nefopam 20 mg is equipotent to morphine 6-12 mg and meperidine 50 mg in surgical settings [
11,
12]. Some authors have reported that nefopam has a morphine-sparing effect of 30-50% [
13,
14].
The intensity of postoperative pain depends on the type of surgery performed. Therefore, this study was conducted to evaluate the effect of nefopam in laparoscopic surgery. The purpose of this study was to compare the analgesic efficacy and side effects of intravenous administration of a continuous infusion of nefopam at various doses, in combination with fentanyl, at the end of surgery in patients undergoing laparoscopic cholecystectomy, in order to establish a more effective and safe dose of nefopam.
Go to :

MATERIALS AND METHODS
This study was approved by the Institutional Review Board, and all participants provided written informed consent. The study investigated 90 patients aged 18 to 65 who underwent laparoscopic cholecystectomy under general anesthesia. The patients were classified as ASA physical status I or II.
Preoperative and intraoperative exclusion criteria included pregnancy, kidney or hepatic diseases, chronic medication with analgesic or sedative drugs, a history of alcohol or drug abuse, and intraoperative administration of opioids other than remifentanil.
On the day before surgery, patients were instructed to familiarize themselves with the visual analogue scale (VAS) for pain (0 = no pain, 10 = worst possible pain).
Patients were premedicated with 3 mg of intramuscular midazolam 30 minutes before surgery. After patients arrived in the operating room, a non-invasive blood pressure monitor, electrocardiograph, pulse oximeter, and bispectral index (BIS) sensors (BIS VISTA®, Aspect Medical Systems, Inc., Newton, MA, USA) were attached.
For anesthesia, effect-site concentration was adjusted with propofol 4 µg/ml and remifentanil 2 ng/ml using target-controlled infusion (TCI). At the loss of consciousness, rocuronium 0.8 mg/kg was administered intravenously and intubation was performed. Anesthesia was maintained using TCI and was controlled with propofol 2-4 µg/ml and remifentanil 1-3 ng/ml to maintain BIS between 40-60. In addition, controlled respiration was managed so that the capnogram remained in the range of 32-36 mmHg.
When the surgery was complete and the fascia was about to be closed, the patients received the following medications intravenously through a peripheral line using an infusion pump: Group 1 was given 50 µg fentanyl, Group 2 was given 50 µg fentanyl and 20 mg nefopam, and Group 3 was given 50 µg fentanyl and 40 mg nefopam. First, fentanyl was mixed with normal saline to a total volume of 20 ml. This mixture was then infused for 5 minutes. Next, nefopam was mixed with normal saline to a total volume of 20 ml. This mixture was then infused for 15 minutes.
After surgery, all patients were injected with pyridostigmine 10 mg and glycopyrrolate 0.4 mg. Patients were transferred to the PACU when they breathed spontaneously and responded to questions after they had recovered from muscle relaxation and had been extubated. In the PACU, patients were given 3 L/min of O2 via nasal cannula and were covered with a blanket. The temperature of the PACU was maintained at 26℃. The initial assignment of the patients to the three groups had been made by an anesthesiologist who did not evaluate the patients in the PACU. In addition, an observer of this experiment was an anesthesiologist who was blinded to the assignment of the patients to the groups.
After arrival of the patients in the PACU, their pain intensity was evaluated using the VAS. Pain and side effects were evaluated at 10 minutes, 30 minutes, 1 hour, 2 hours, 6 hours, and 12 hours after patient arrival in the PACU. If VAS scores were reported as greater than 4, ketorolac 30 mg was intravenously administered.
In addition, the degree of nausea, vomiting, shivering, respiratory depression, and dry mouth were evaluated. Scores were classified as 0 = none; 1 = minor, no treatment necessary; or 2 = treatment required to evaluate side effects. Respiratory depression was defined as a respiratory rate of less than 10/minute and a sedation score of 3. Sedation scores were classified as 0 = no sedation, 1 = intermittent drowsiness, 2 = patient drowsy but could be aroused verbally, and 3 = impossible to arouse the patient verbally.
If a nausea or vomiting score of 2 was reported, ramosetron 0.3 mg was intravenously administered, and if a shivering score of 2 was reported, a forced air-warming blanket was applied. Pethidine was not administered to treat shivering because of its potential effect on postoperative nausea and vomiting (PONV). In the presence of respiratory depression, naloxone 0.04 mg was intravenously administered.
Results were represented as mean ± SD or the number of patients (%). Statistical analyses were performed using SPSS version 13.0 (SPSS Inc., Chicago, IL, USA). A one-way ANOVA was used for comparisons of age, weight, height, duration of operation, duration of anesthesia, pain and analgesic consumption (ketorolac) during the study period among the groups. Post hoc comparisons were performed by applying the Scheffe correction to the significance level. A Chi-square test was used for comparisons of gender, ASA class, severity of side effects, and the number of rescue anti-emetic drugs administered to the groups. P values of < 0.05 were considered statistically significant.
Go to :

RESULTS
There were no statistically significant differences in age, weight, height, duration of operation, duration of anesthesia, gender, or ASA class among the groups (
Table 1). In the comparison of postoperative pain and ketorolac consumption according to time after surgery, the results from Groups 2 and 3 were statistically significantly less than those of Group 1 at 10 minutes, 2 hours, and 6 hours after arrival in the PACU, whereas no statistically significant difference was observed between Groups 2 and 3. There were no statistically significant differences among the groups at 30 minutes, 1 hour, and 12 hours after arrival in the PACU (
Fig. 1,
Table 2).
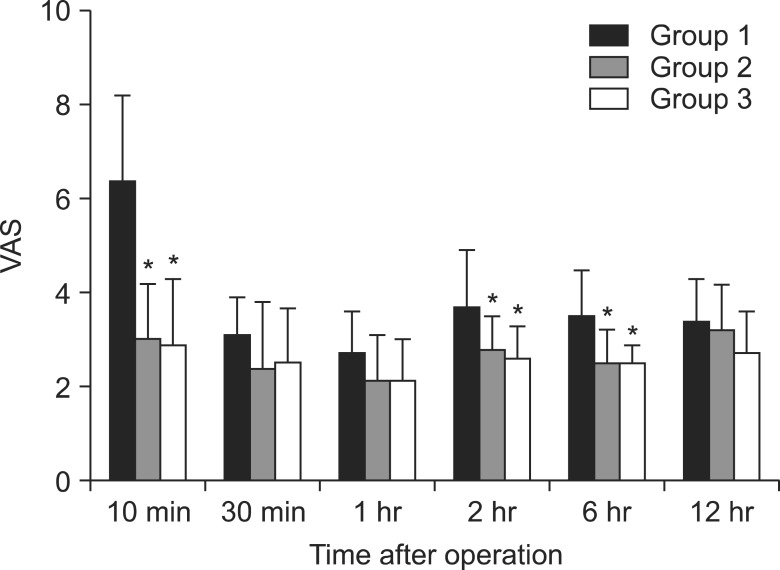 | Fig. 1Postoperative visual analogue scale at different times. Groups 2 and 3 showed significantly lower pain scores than Group 1 at 10 minutes, 2 hours, and 6 hours after arrival in the PACU. *P < 0.05 compared to Group 1. 
|
Table 1
Demographic and Anesthetic Data of Patients
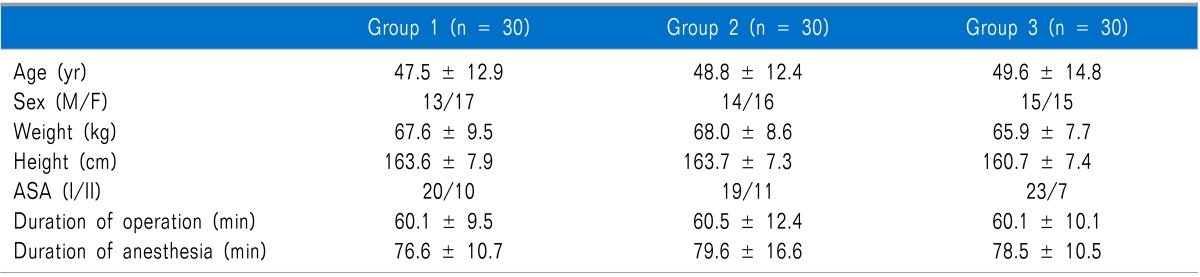

Table 2
Postoperative Ketorolac Consumption at Different Times
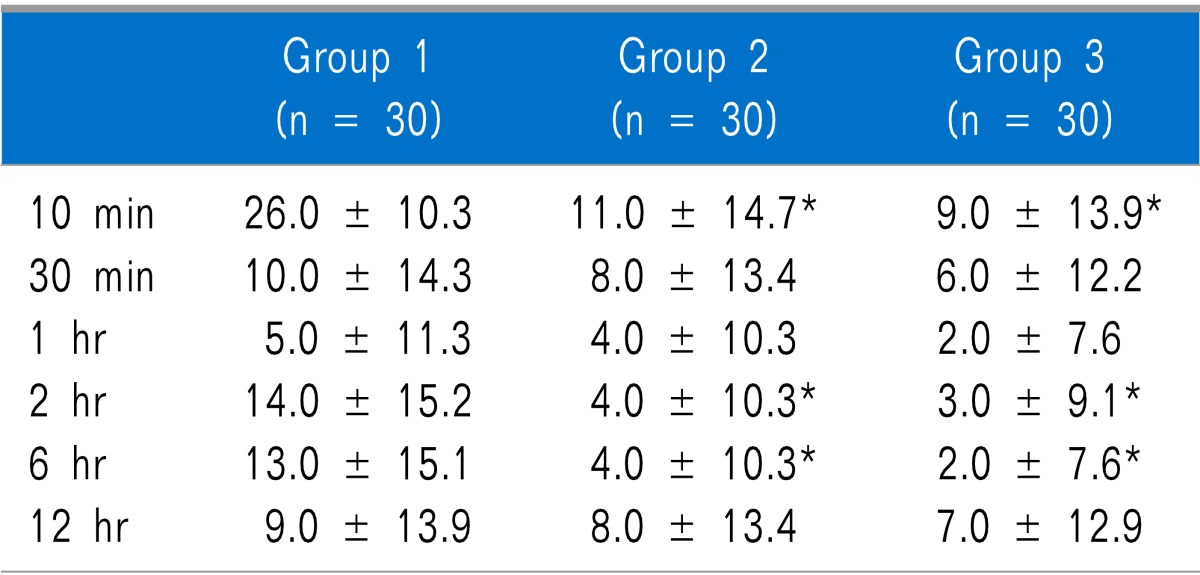

Nausea was statistically significantly lower in Group 2 than in Groups 1 and 3 at 10 minutes after arrival in the PACU, but there was no statistically significant difference between Groups 1 and 3. There were no significant differences among the groups at 30 minutes, 1 hour, 2 hours, 6 hours, and 12 hours after arrival in the PACU (
Table 3). As for vomiting, there were no statistically significant differences among the groups at any time (
Table 3). Shivering was statistically significantly lower in Groups 2 and 3 than in Group 1 at 10 minutes after arrival in the PACU, but there was no statistically significant difference between Groups 2 and 3. There were no significant differences among the groups at 30 minutes, 1 hour, 2 hours, 6 hours, and 12 hours after arrival in the PACU (
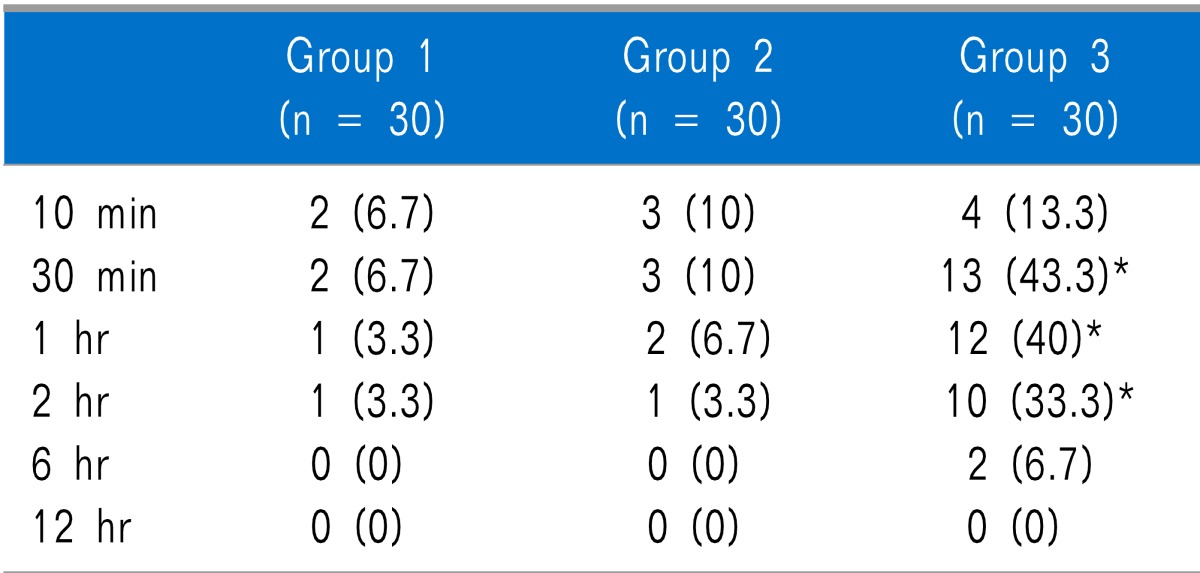
Table 4). As for respiratory depression, there were no statistically significant differences between the groups at any time. Dry mouth was statistically significantly higher in Group 3 than in Groups 1 and 2 at 30 minutes, 1 hour, and 2 hours after arrival in the PACU, but there was no statistically significant difference between Groups 1 and 2. There were no statistically significant differences among the groups at 10 minutes, 6 hours, and 12 hours after arrival in the PACU (
Table 5).
Table 3
Incidence of Nausea, Vomiting and Use of Rescue Anti-emetics at Different Time
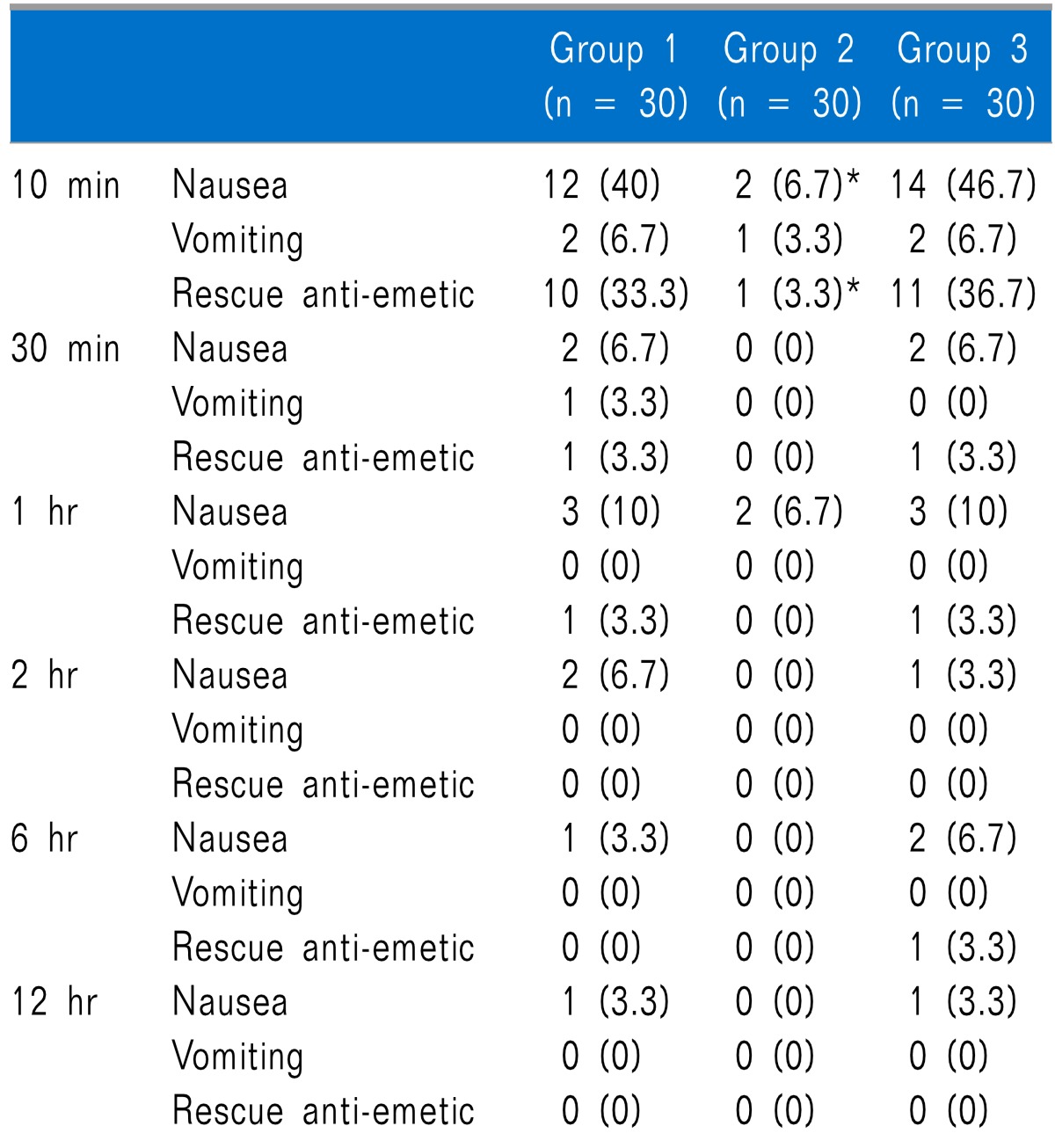

Table 4
Incidence of Shivering at Different Time
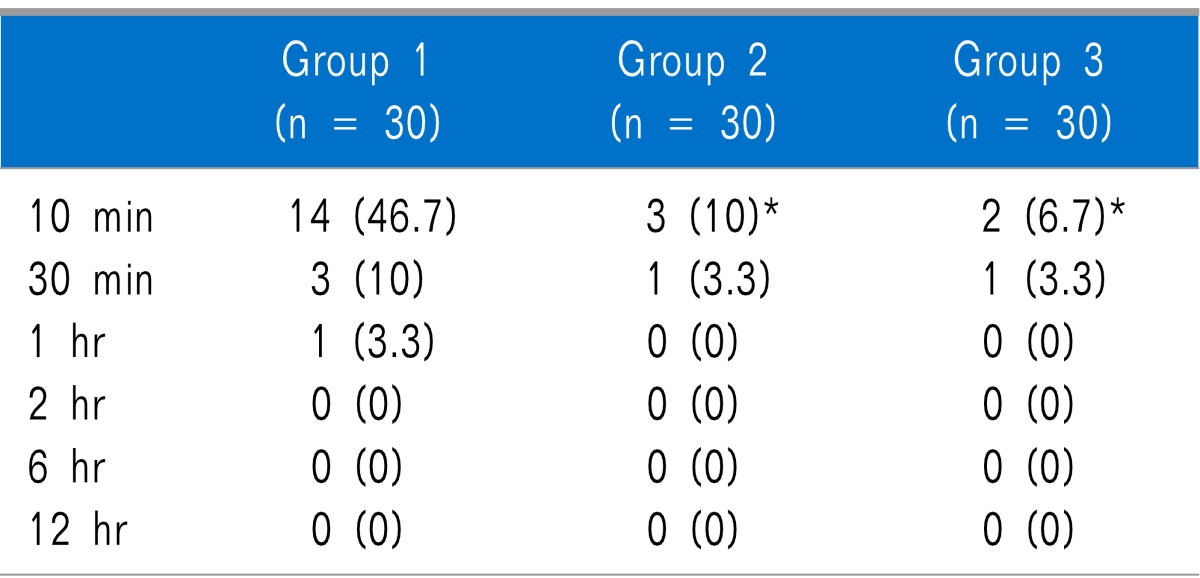

Table 5
Incidence of Dry Mouth at Different Time

Go to :

DISCUSSION
This study aimed to evaluate the optimal dose of nefopam in combination with fentanyl at the end of laparoscopic cholecysyectomy. With respect to its effects on pain reduction and side effects, the more effective combination was discovered to be 50 µg fentanyl and 20 mg nefopam.
Anesthesiologists have tested many different approaches in an attempt to reduce pain and side effects in patients after surgery. However, no method has been discovered that can completely eliminate pain without side effects. The current study attempted to find a way to achieve this through the use of nefopam. As nefopam is known to reduce opioid dosage [
15], this study compared fentanyl alone versus fentanyl in combination with nefopam by continuous infusion.
Although the usual intravenous dose of nefopam recommended by pharmaceutical firms is 20 mg, a median effective analgesic dose (ED
50, analgesic efficacy in 50% of patients) has yet to be defined [
15]. However, Van Elstraete and Sitbon [
16] recently reported that the ED
50 in patients undergoing tonsillectomy under general anesthesia is 21.7 mg. Therefore, as various studies regarding the dose of nefopam are needed, this study compared the degree of pain and side effects in three groups of patients who were administered fentanyl 50 µg alone, or fentanyl in combination with nefopam 20 mg or nefopam 40 mg.
Although the degree of pain was reduced in both groups that received nefopam, since there was no difference between the groups receiving nefopam 20 mg and nefopam 40 mg, it can be said that a dose of nefopam 20 mg with fentanyl 50 µg via continuous infusion at the end of laparoscopic surgery was sufficient. There was no difference in the degree of pain at 30 minutes and 1 hour after arrival in the PACU, which may be attributed to the administration of ketorolac injections in patients with VAS scores greater than 4, and the difference between pain scores at 2 hours and 6 hours after arrival in the PACU may have been due to the synergistic effect of ketorolac and nefopam.
It is known that the injection of nefopam at the end of surgery improves postoperative analgesia and reduces morphine consumption [
17,
18]. In addition, Laboureyras et al. [
19] reported that in an experiment using rats, one injection of nefopam before surgery reduced not only postoperative pain but also long-term pain vulnerability. In the present study, the incidence of nausea was statistically significantly lower in Group 2 than in Groups 1 and 3 at 10 minutes after arrival in the PACU. There was no difference between the groups after 30 minutes after arrival in the PACU, because ramosetron was administered to patients with severe symptoms.
Although nefopam is known as a drug with emetic properties, in a study of patients undergoing elective hepatic resection, the pain score and the incidence of nausea were found to be lower in the group receiving nefopam with morphine for patient-controlled analgesia (PCA) than in the group receiving morphine alone for PCA [
14]. Mimoz et al. [
14] could not explain the exact reason why, after elective hepatic resection, nausea was reduced in the group administered medicine mixed with nefopam. However, the authors presumed that, in the group administered medicine mixed with propacetamol, the reduction in nausea was due to the morphine-sparing effect caused by the reduced use of morphine.
In the current study, however, it was found that although nausea was less prevalent in the group that received nefopam 20 mg than in the other groups, no difference was observed between the group that received nefopam 40 mg and the group that received fentanyl alone.
Since this study used ketorolac instead of opioid as a rescue drug, the findings cannot be explained by the opioid-sparing effect, and particularly, because nausea was not reduced in the group that received 40 mg nefopam, we cannot explain the exact reason for these findings. We may suppose that pain reduction after surgery reduces nausea, but in the group administered 40 mg nefopam, only pain was reduced, while nausea was not reduced; thus, we cannot draw any conclusions relating pain and nausea at this point. However, 40 mg nefopam may cause more side effects than 20 mg, so we can assume that in terms of its doses, nausea was more reduced at a dose of 20 mg nefopam. Thus, the use of nefopam 20 mg at the end of laparoscopic surgery is considered desirable.
It is known that nefopam reduces the shivering that commonly occurs after anesthesia [
20]. Nefopam is a benzoxazocine compound that is structurally related to orphenadrine and diphenhydramine [
10]. It reduces only the shivering threshold without influencing vasoconstriction or sweating [
21]. In both in vitro and in vivo studies, it has been reported that nefopam is related to the inhibition of catecholamine reuptake [
22]. Nefopam also possesses the properties of a noncompetitive
N-Methyl-
D-Aspartate (NMDA) receptor antagonist. Therefore, nefopam reduces shivering due to its related mechanism to the thermoregulatory response [
23,
24]. In this study, shivering was significantly lower in Groups 2 and 3 (in which nefopam was used) than in Group 1 (in which nefopam was not used).
Dry mouth, which is considered a pharmacological property of nefopam, was significantly higher in Group 3 than in Groups 1 and 2 at 30 minutes, 1 hour, and 2 hours after arrival in the PACU, The plasma half-life of nefopam is about 4 hours (range about 3 to 8 hours) and plasma peak concentrations are reached in 15 minutes [
10]. Therefore, 15 minutes after injection of nefopam, the drug will reach the plasma peak concentration and patients will experience severe dry mouth, which will decline after the drug's half-life is reached. The common side effects of nefopam are tachycardia and sweating [
10]. However, we did not assess these side effects in the current study since fentanyl and nefopam were injected when the fascia was closed, which was around the time when the patients began to awaken from anesthesia, and thus it was not clear if any observed tachycardia and sweating was caused by nefopam or other administered medicine or by recovery from anesthesia.
Nefopam can be considered a safe drug that can be used in controlling pain after surgery. Although there have been reports on its side effects, these are not significant when an appropriate dose is used. This study revealed that administration of fentanyl 50 µg with nefopam 20 mg effectively reduced postoperative pain and side effects. However, the intensity of postoperative pain depends on the type of surgery, and the results of this study are limited to laparoscopic cholecystectomy. Therefore, further research on the dose of nefopam, as well as on various kinds of opioids, and their doses, is needed, since research on dose of nefopam at the end of surgery is limited.
Go to :

ACKNOWLEDGEMENTS
This paper was supported by Wonkwang University in 2011.
Go to :

References
1. Klohs MW, Draper MD, Petracek FJ, Ginzel KH, Ré ON. Benzoxazocines: a new chemical class of centrally acting skeletal muscle relaxants. Arzneimittelforschung. 1972; 22:132–133. PMID:
5066986.
2. Gassel MM, Diamantopoulos E, Petropoulos V, Hughes AC, Ballesteros ML, Ré ON. Controlled clinical trial of oral and parenteral nefopam hydrochloride. A novel and potent analgesic drug. J Clin Pharmacol. 1976; 16:34–41. PMID:
1107359.

3. Piercey MF, Schroeder LA. Spinal and supraspinal sites for morphine and nefopam analgesia in the mouse. Eur J Pharmacol. 1981; 74:135–140. PMID:
6276187.

4. Kehlet H, Dahl JB. The value of "multimodal" or "balanced analgesia" in postoperative pain treatment. Anesth Analg. 1993; 77:1048–1056. PMID:
8105724.

5. Kim SH, Kim SI, Ok SY, Park SY, Kim MG, Lee SJ, et al. Opioid sparing effect of low dose ketamine in patients with intravenous patient-controlled analgesia using fentanyl after lumbar spinal fusion surgery. Korean J Anesthesiol. 2013; 64:524–528. PMID:
23814653.

6. Cheong YK, Son Y, Song YK, Kim TY. The analgesic effect of continuous intraarticular infusion of ropivacaine and fentanyl after arthroscopic shoulder surgery. Korean J Anesthesiol. 2006; 50:449–453.

7. Moffat AC, Kenny GN, Prentice JW. Postoperative nefopam and diclofenac. Evaluation of their morphine-sparing effect after upper abdominal surgery. Anaesthesia. 1990; 45:302–305. PMID:
2337215.

8. Parker RK, Holtmann B, Smith I, White PF. Use of ketorolac after lower abdominal surgery. Effect on analgesic requirement and surgical outcome. Anesthesiology. 1994; 80:6–12. PMID:
8291731.

9. Dordoni PL, Della Ventura M, Stefanelli A, Iannace E, Paparella P, Rocca B, et al. Effect of ketorolac, ketoprofen and nefopam on platelet function. Anaesthesia. 1994; 49:1046–1049. PMID:
7864317.

10. Heel RC, Brogden RN, Pakes GE, Speight TM, Avery GS. Nefopam: a review of its pharmacological properties and therapeutic efficacy. Drugs. 1980; 19:249–267. PMID:
6991238.
11. Sunshine A, Laska E. Nefopam and morphine in man. Clin Pharmacol Ther. 1975; 18:530–534. PMID:
1102231.

12. Tigerstedt I, Sipponen J, Tammisto T, Turunen M. Comparison of nefopam and pethidine in postoperative pain. Br J Anaesth. 1977; 49:1133–1138. PMID:
337983.

13. McLintock TT, Kenny GN, Howie JC, McArdle CS, Lawrie S, Aitken H. Assessment of the analgesic efficacy of nefopam hydrochloride after upper abdominal surgery: a study using patient controlled analgesia. Br J Surg. 1988; 75:779–781. PMID:
3167526.

14. Mimoz O, Incagnoli P, Josse C, Gillon MC, Kuhlman L, Mirand A, et al. Analgesic efficacy and safety of nefopam vs. propacetamol following hepatic resection. Anaesthesia. 2001; 56:520–525. PMID:
11412156.

15. Beloeil H, Delage N, Nègre I, Mazoit JX, Benhamou D. The median effective dose of nefopam and morphine administered intravenously for postoperative pain after minor surgery: a prospective randomized double-blinded isobolographic study of their analgesic action. Anesth Analg. 2004; 98:395–400. PMID:
14742377.

16. Van Elstraete AC, Sitbon P. Median effective dose (ED50) of paracetamol and nefopam for postoperative pain: isobolographic analysis of their antinociceptive interaction. Minerva Anestesiol. 2013; 79:232–239. PMID:
23241734.
17. Tramoni G, Viale JP, Cazals C, Bhageerutty K. Morphine-sparing effect of nefopam by continuous intravenous injection after abdominal surgery by laparotomy. Eur J Anaesthesiol. 2003; 20:990–992. PMID:
14690106.

18. Saghaei E, Moini Zanjani T, Sabetkasaei M, Naseri K. Enhancement of antinociception by co-administrations of nefopam, morphine, and nimesulide in a rat model of neuropathic pain. Korean J Pain. 2012; 25:7–15. PMID:
22259710.

19. Laboureyras E, Chateauraynaud J, Richebé P, Simonnet G. Long-term pain vulnerability after surgery in rats: prevention by nefopam, an analgesic with antihyperalgesic properties. Anesth Analg. 2009; 109:623–631. PMID:
19608840.

20. Taniguchi Y, Ali SZ, Kimberger O, Zmoos S, Lauber R, Markstaller M, et al. The effects of nefopam on the gain and maximum intensity of shivering in healthy volunteers. Anesth Analg. 2010; 111:409–414. PMID:
20529984.

21. Alfonsi P, Adam F, Passard A, Guignard B, Sessler DI, Chauvin M. Nefopam, a nonsedative benzoxazocine analgesic, selectively reduces the shivering threshold in unanesthetized subjects. Anesthesiology. 2004; 100:37–43. PMID:
14695722.

22. Girard P, Coppé MC, Verniers D, Pansart Y, Gillardin JM. Role of catecholamines and serotonin receptor subtypes in nefopam-induced antinociception. Pharmacol Res. 2006; 54:195–202. PMID:
16750379.

23. Fernández-Sánchez MT, Díaz-Trelles R, Groppetti A, Manfredi B, Brini AT, Biella G, et al. Nefopam, an analogue of orphenadrine, protects against both NMDA receptor-dependent and independent veratridine-induced neurotoxicity. Amino Acids. 2002; 23:31–36. PMID:
12373515.

24. Cheong YK, Kim TY, Lee SW. The preventive effect on postanesthetic shivering according to the dosages of ketamine. Korean J Anesthesiol. 2008; 55:277–281.

Go to :






 PDF
PDF Citation
Citation Print
Print







 XML Download
XML Download