INTRODUCTION
Desflurane is a halogenated, inhaled anesthetic agent with low blood and tissue solubility, which promotes rapid induction and early recovery [1]. However, a rapid increase in the concentration of desflurane above 1 minimal alveolar concentration (MAC) has been demonstrated to induce transient sympathetic hyperactivity and lead to increases in blood pressure (BP) and heart rate (HR) [2,3]. Cardiovascular stimulation might increase the risk of myocardial ischemia, specifically in patients with coronary artery disease, during inhalation of desflurane [4]. Clonidine, esmolol, fentanyl, alfentanil, and remifentanil have been reported to blunt the cardiovascular stimulation induced by desflurane [5–7].
Dexmedetomidine is a highly selective α2 adrenergic receptor agonist indicated for sedation and analgesia in both critical care and perioperative settings [8]. The use of dexmedetomidine as an anesthetic adjunct has been reported to attenuate intraoperative sympathetic tone and the hemodynamic response to endotracheal intubation [9–12]. This character of dexmedetomidine arises from the inhibition of sympathetic outflow at the α2 adrenergic receptor in postsynaptic neurons of the central nervous system [8]. Kersten et al. [13] reported that pretreatment with dexmedetomidine decreased the hemodynamic effects induced by desflurane in a canine model. However, there has been limited research on dexmedetomidine that focuses on the attenuation of the hemodynamic effects induced by desflurane in humans. Therefore, we conducted this investigation to examine the hemodynamic effects of dexmedetomidine infusion before anesthetic induction on desflurane-induced cardiovascular changes.
MATERIALS AND METHODS
Patients
This was a randomized control study conducted at a tertiary teaching hospital. We obtained approval for our study from the Institutional Review Board (IRB) and Ethics Committee (IRB no. 2016-06-308-005), registered our study at www.clinicaltrials.gov (ID. KCT0002157), enrolled a total of 60 patients of American Society of Anesthesiologists physical status classification I and II (age ≥ 19 and < 65 years), and scheduled elective surgery for them at our hospital between August and December 2016 under general anesthesia. We obtained written informed consent from all participants. They were randomly assigned into three groups, and each group of 20 patients received an allocated intervention, respectively (Fig. 1). The following patients were excluded: those who did not provide informed consent; those with histories of asthma, habitual abuse of hypnotics or sedatives, anemia, or hematocrit levels < 24%; those who required pre-anesthetic medication; those with uncontrolled hypertension (systolic blood pressure (SBP) ≥ 160 mmHg), pre-induction SBP ≤ 90 mmHg, pre-induction HR ≥ 100 or ≤ 45 beats/min, or uncontrolled diabetes (long-lasting blood glucose over 200 mg/dl); those with a risk of aspiration; those who were judged by the investigator unfit for our study; and pregnant women.
Fig. 1
CONSORT flow chart presenting the study protocol. CONSORT: consolidated standards of reporting trials, ASA PS: American Society of Anesthesiologists physical status, Hct: hematocrit, SBP: systolic blood pressure, HR: heart rate, DM: diabetes mellitus, IV: intravenous.
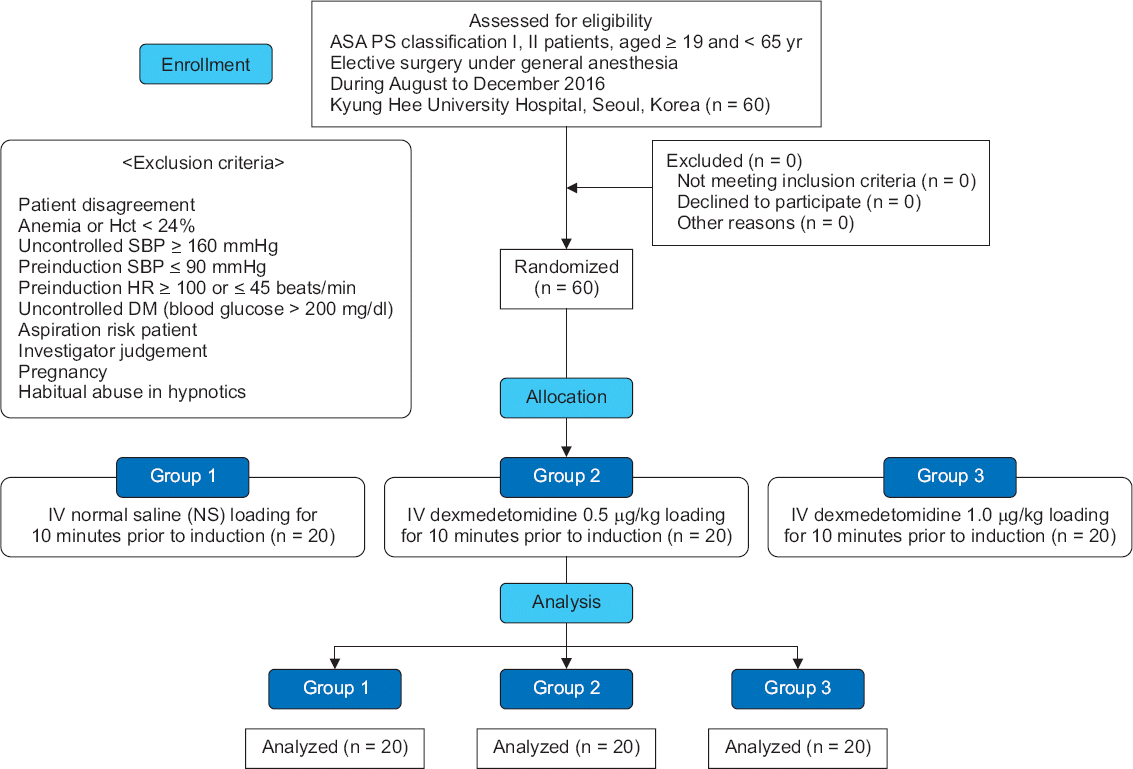
The patients enrolled in this study were randomly allocated into three groups on the basis of computer-generated random numbers. The researchers not participating in the study prepared either 50 ml normal saline (NS) (group 1) or dexmedetomidine (Precedex, Hospira, USA) solution diluted in 50 ml NS (group 2, 0.5 μg/kg; group 3, 1.0 μg/kg). Each group received 0.5 ml/kg of either of these solutions for 10 minutes through an automatic fluid infusion regulator before anesthetic induction.
Hemodynamic monitoring and anesthesia protocol
Upon arrival of a patient at the operating room, routine monitoring modalities for noninvasive BP monitoring, 3-lead electrocardiography, and peripheral oxygen saturation measurements were set up. After drug infusion for 10 minutes, injections of 40 mg lidocaine, 0.2 mg/kg etomidate, and 0.8 mg/kg rocuronium were administered for the induction of general anesthesia. The patients in each group inhaled 8 vol% of desflurane for anesthetic maintenance. Desflurane (Suprane, Baxter Healthcare, Puerto Rico) was administered at a vaporizer dial setting of 8% with a target end-tidal CO2 concentration of 30 mmHg for 5 minutes by manual ventilation using an anesthesia bag. The inspiratory and expiratory desflurane concentrations were measured at the Y-piece via a side-stream sampling tube and recorded every minute using the Dräger Fabius GS monitor (Dräger Medical AG & CO, Germany). Any adverse events, including hypotension (mean blood pressure [MBP] < 50 mmHg), hypertension (SBP > 190 mmHg), tachycardia (HR > 130 beats/min), or bradycardia (HR < 40 beats/min), during the induction of general anesthesia were also recorded. When hypertension or tachycardia occurred, a rescue dose of 0.5 μg/kg remifentanil was injected. When bradycardia occurred, a rescue dose of 0.5 mg atropine was injected. After 5 minutes of desflurane administration, tracheal intubation was performed after adequate muscle relaxation. Surgery was subsequently performed as scheduled.
Data analysis
Twenty patients in group 2 were included in the analysis; however, two patients had missing data from certain times because of sudden BP cuff malfunction. The research was conducted without filling in the missing data. With no exclusion, all 20 individuals in both groups 1 and 3 were included in each analysis (Fig. 1). MBP and HR were the primary outcome parameters in our study. Preoperative characteristics of the patients, including age, sex, height, weight, body mass index (BMI), and past medical and medication history were already recorded from electronic medical records. In the operating room, baseline hemodynamic parameters including MBP and HR were recorded after 1–2 minutes of rest. After initiating drug infusion for 10 minutes, the parameters were evaluated at 5 and 10 minutes. With inhalation of 8 vol% desflurane after anesthetic induction, we recorded the four main parameters for 5 minutes at 1-minute intervals. Inhalation was maintained at 8 vol% until the end of data collection. Finally, all data values were analyzed for comparison among the three groups.
Statistical analysis
All data were expressed as mean ± standard deviation (SD). Referring to existing literature that evaluated the hemodynamic effect of remifentanil in desflurane anesthesia [7], we performed a pilot study. The sample size was yielded with our values of HR 3 minutes after desflurane inhalation by G* power (Ver. 3.1.9.2, Franz Faul, Germany). The SDs of the three groups’ means derived from the variance of the three groups’ means, and also the common SD derived from the common variance, were calculated (27.2 and 45.81, respectively). Thus, the effect size was determined to be 0.59. As a result, the total sample size was calculated after inputting the value of the desired power, 95%, at 5% significance level. Assuming a 20% drop out during data collection, a total of 60 patients (20 in each group) was required.
We used IBM SPSS version 21.0 (IBM Corp., USA) for analysis. One-way analysis of variance (ANOVA) and chi-square tests were used to analyze demographic data among participants, such as age, sex, height, weight, BMI, past medical and medication history, and end-tidal desflurane concentration. Incidences of the adverse events among the groups during the study were also compared with the same method. Differences in the data at specific times among the three groups were evaluated with ANOVA. To prove the significant statistical differences, we performed the post-hoc test with Tukey correction. P values of < 0.05 were accepted as statistically significant.
RESULTS
There were no significant differences in the baseline demographic and clinical characteristics among the three groups. In addition, the mean end-tidal concentration of desflurane (vol%) showed no significant differences among the groups (Table 1).
Table 1
Baseline Demographics and End-tidal Concentration of Desflurane Inhalation
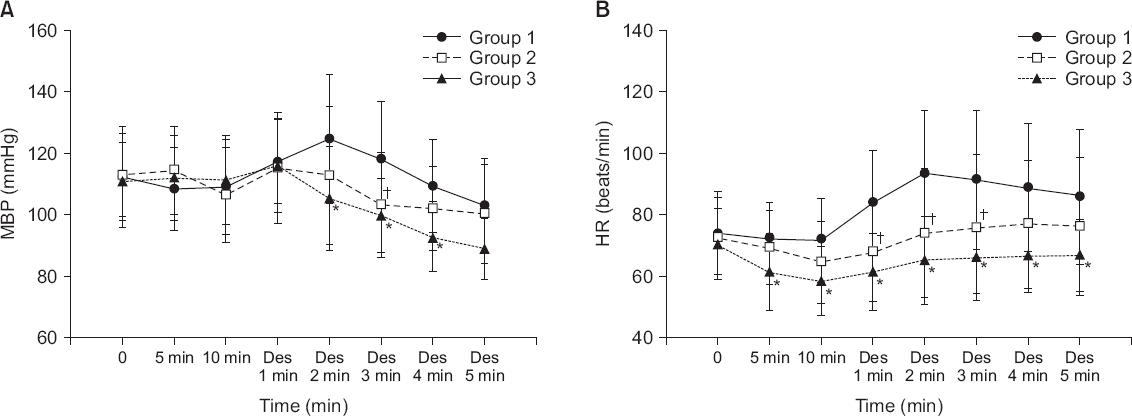
The baseline MBP values of groups 1, 2, and 3 were 112.1 ± 16.3, 112.7 ± 13.5, and 110.8 ± 13.2 mmHg, respectively (Fig. 2A). In group 1, the MBP values 2, 3 and 4 minutes after desflurane inhalation were 124.5 ± 21.2, 118.1 ± 18.5, and 109.2 ± 15.1 mmHg, respectively. In group 2, the MBP 3 minutes after desflurane inhalation was 103.1 ± 17.1 mmHg, which was significantly lower compared to that in group 1 (P = 0.037). In group 3, the MBP values 2, 3 and 4 minutes after desflurane inhalation were 105.2 ± 13.1, 99.5 ± 12.3, and 92.7 ± 10.8 mmHg, respectively, and were significantly lower compared to those in group 1 (P = 0.023, P = 0.021, and P = 0.031). The baseline HRs in groups 1, 2, and 3 were 73.9 ± 13.5, 72.3 ± 13.3, and 70.2 ± 11.5 beats/min, respectively (Fig. 2B). In group 1, the HR was significantly elevated above the baseline during the entire 5 minutes after desflurane inhalation (P = 0.043, P = 0.021, P = 0.033, P = 0.041, P = 0.038, respectively) and the MBP was significantly elevated above the baseline at 1, 2, and 3 minutes after desflurane inhalation (P = 0.045, P = 0.028, P = 0.039, respectively). However, in group 2, the increase in HR induced by desflurane inhalation was significantly suppressed compared to that in group 1 at 1, 2, and 3 minutes after desflurane inhalation (P = 0.021, P = 0.018, P = 0.027, respectively). In group 3, the increase in HR induced by desflurane inhalation was significantly suppressed compared to that in group 1 during the entire 5 minutes after desflurane inhalation (P = 0.012, P = 0.001, P = 0.007, P = 0.014, P = 0.021, respectively). Neither MBP nor HR were significantly different between groups 1 and 2 in the last 5 minutes after desflurane inhalation (P = 0.384).
Fig. 2
(A) Changes in MBP observed in the three groups during the study period. (B) Changes in HR observed in the three groups during the study period. Group 1: IV normal saline loading for 10 minutes prior to induction. Group 2: 0.5 hhhhhhhhg/kg IV dexmedetomidine loading for 10 minutes prior to induction. Group 3: 1.0 nnnnnnnng/kg IV dexmedetomidine loading for 10 minutes prior to induction. 5 min: 5 minutes after initiating drug infusion. 10 min: 10 minutes after initiating drug infusion. Des 1–5 min: 1–5 minutes after desflurane inhalation. MBP: mean blood pressure, HR: heart rate, IV: intravenous. *P < 0.05, significant difference between groups 1 and 3. †P < 0.05, significant difference between groups 1 and 2.
There were several adverse events during this study. The incidence of hypertension (SBP ≥ 190 mmHg) was 30% (n = 6), 5% (n = 1), and 10% (n = 2) in groups 1, 2, and 3, respectively, and 1 patient in group 1 presented with tachycardia (HR > 130 beats/min) during desflurane inhalation. Meanwhile, 2 patients in group 3 presented with bradycardia (HR < 40 beats/min) during the first 10-minute drug infusion. However, among the three groups, there were no severe hemodynamic collapses or significant differences in the incidences of these adverse effects (Table 2).
DISCUSSION
Our hypothesis was that dexmedetomidine, a potent selective α2 adrenergic receptor agonist, may suppress the transient sudden increases in BP and HR when desflurane concentration is rapidly increased over 1 MAC during anesthetic induction [14]. Our study showed that intravenous (IV) administration of dexmedetomidine over 10 minutes prior to anesthetic induction had blunting effects on transient cardiovascular changes induced upon inhalation of desflurane. In previous studies, transient sympathetic stimulation effects of desflurane developed 1–2 minutes after inhalation and continued for 3–4 minutes [2,7]. Our study showed the same results, and dexmedetomidine infusion before anesthetic induction blunted these cardiovascular changes in our study.
Our study compared two different loading doses of dexmedetomidine: 0.5 μg/kg and 1.0 μg/kg. There are few studies about different dose comparisons of dexmedetomidine on cardiovascular changes with desflurane. Some studies have compared the effects of different doses of preoperative dexmedetomidine on hemodynamic changes; however, most of them used dexmedetomidine as a single injection for premedication to validate the hemodynamic and sympathetic stabilizing effect during endotracheal intubation under volatile anesthesia [15,16]. Single injections of 1.0 and 2.0 μg/kg before induction showed major limitations including marked bradycardia or hypotension despite sufficient hemodynamic attenuation against stress responses [17], and we compensated with continuous infusion for 10 minutes before anesthetic induction. Gulabani et al. [18] compared two different doses of dexmedetomidine (0.5 and 1.0 μg/kg IV loading over 10 minutes), and they concluded that dexmedetomidine 1.0 μg/kg was superior to 0.5 μg/kg for blunting of the pressure response to laryngoscopic intubation. In our study, 0.5 and 1.0 μg/kg dexmedetomidine loading infusions were sufficient to effectively inhibit the cardiovascular stimulation induced by desflurane during the entire 5 minutes, and the HR and MBP remained closer to the baseline after administration of 0.5 μg/kg dexmedetomidine than after administration of a dose of 1.0 μg/kg.
Many studies have shown increases in plasma catecholamine concentrations after anesthetic induction with inhalation of desflurane [19,20]. These increases in plasma catecholamine concentrations are inhibited by dexmedetomidine [13,19,20]. It has been reported that the central α2 adrenoceptor located in medullary nuclei is the main sympathetic catecholamine outflow inhibition site [21,22]. Opioids and β-blockers have also demonstrated cardiovascular stabilization effects during inhalation anesthesia with desflurane [5]. However, opioids and β-blockers mainly have cardiac and vascular effects rather than central adrenal catecholamine outflow inhibition effects, so they cannot inhibit the increase in plasma catecholamine concentrations. Dexmedetomidine can inhibit sympathetic stimulation compared with opioids and β-blockers because of the inhibition of central adrenal catecholamine outflow.
It has been reported that a lower HR is associated with decreased myocardial ischemia and improved long-term outcomes in patients undergoing major vascular surgery [23]. In addition, dexmedetomidine attenuates the hemodynamic and neuroendocrinal response of surgical trauma during heart surgery of high-risk adults and children as well [9,10,15,24]. In many studies, sympathetic stimulation by volatile agents has been shown to be related to cardiac ischemia in patients of cardiovascular disease [4]. In our study, decreases in HR compared with that of the control group suggested that suppression of sympathetic activation by perioperative dexmedetomidine seems to specifically beneficial in patients with ischemic heart disease to prevent myocardial infarction [4,25]. The side effects of dexmedetomidine in high-risk patients or during surgery are severe hypotension and bradycardia requiring immediate treatments. In other studies in patients undergoing heart surgery, there were no significant differences in the incidences of such adverse events [9,10].
There are several limitations to this study. First, we compared only two different doses of dexmedetomidine: 0.5 μg/kg and 1.0 μg/kg. Comparison of more differentiated doses could have provided more detailed information about the optimal dose of dexmedetomidine to effectively attenuate the transient cardiovascular changes without any serious adverse events induced by desflurane. Second, measuring plasma catecholamine concentrations is quite invasive, time-consuming, and expensive. For these reasons, we could not analyze the trends of plasma norepinephrine, epinephrine, and vasopressin levels in terms of sympathetic stimulation with desflurane and the suppressive effect of dexmedetomidine. Third, this study was conducted only among healthy adults. Therefore, we could not know whether the outcomes were improved in patients with ischemic heart disease.
In conclusion, our randomized, prospective study demonstrates that infusion of dexmedetomidine for 10 minutes before general anesthetic induction has blunting effects on the transient cardiovascular changes induced by desflurane inhalation during the induction period.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download