INTRODUCTION
Cirrhosis has many possible manifestations, and its signs and symptoms may be either from a direct result of the failure of liver cells or secondary to the resultant portal hypertension. Portal hypertension leads to the development of new veins called collateral vessels. Collateral vessels develop at specific places, such as esophagus, stomach, abdominal wall and rectum. In particularly, collateral vessels located in sites other than the gastroesophageal region are referred as ectopic varices, and they are fragile and can rupture easily, resulting in a large amount of blood loss [1]. This complication can be serious and can occasionally lead to fatal result. However, given that the treatment options vary, no standard therapy has been established.
Collateral vessels located in rectum reported forth most develop site, but there is no report about intraoperative rectal variceal bleeding [1]. We describe the previously unreported occurrence of sudden massive rectal variceal bleeding after cross-clamping of the inferior vena cava and hepatic portal vein in the absence of venovenous bypass during living donor liver transplantation (LT).
Go to : 
CASE REPORT
A 52-year-old woman with known alcoholic cirrhosis, portal hypertension, esophageal and gastric varices, ascites and hepatic encephalopathy was admitted for a living donor LT. Before the surgery, the patient’s model for end-stage liver disease score was 37, and the Child-Pugh score was class C. Laboratory values included 6.4 g/dl hemoglobin, 1.21 mg/dl creatinine, 37.36 mg/dl total bilirubin, 3.85 INR, 44.7 s prothrombin time, 81.3 s partial thromboplastin time, and 41 × 103/μl platelets.
Anesthesia was induced with intravenous propofol 80 mg and rocuronium bromide 50 mg and maintained with a 50% oxygen/air mixture and desflurane 4 vol% with continuous infusion of remifentanil and cisatracurium. During the induction of general anesthesia, arterial cannulations were performed in the right radial artery and right femoral artery. A double-lumen 8F dialysis catheter was inserted into the left internal jugular vein, 9 Fr large bore catheter (Advanced Venous Access, Edwards Lifesciences, USA) and 7F triple-lumen catheters were inserted into the right internal jugular vein. Hemodynamic monitoring included the right radial and femoral arterial, central venous pressures, and cardiac output measurements (Flo Trac/Vigileo™, Edwards Lifesciences, USA) using radial artery catheter.
The LT operation started 105 minutes after the induction of anesthesia. During the anhepatic period, the hemodynamic profile was unstable (Table 1). During surgery, dopamine and dobutamine infusion were started and titrated following a fall in CO and cardiac index. With a maximum dosage of norepinephrine, dopamine and dobutamine were infused up to 20 μg/kg/min to maintain the mean arterial blood pressure between 60 and 70 mmHg. Blood products and fluids were rapidly infused with a rapid infusion system.
Table 1
Change of Vital Signs and Amount of Blood Products Used during Operation
An hour after cross-clamping of the inferior vena cava and hepatic portal vein, we transfused more blood products into the patient than the amount of blood that flew out from the surgical field, but there were still high stroke volume variation (> 15%) and low hemoglobin levels showed below 6 to 7 g/dl hemoglobin. Then, blood was noticed dripping down from the surgical drapes. Initially, we checked for disconnected peripheral intravenous lines, but this was not the case. Then blood was seen discharging out of the patient’s anus, to an extent that two surgical bed sheets and surgical drapes under the patient were drenched with blood, along with blood dripping onto the floor.
A colonoscopy examination was performed by a transplant surgeon. Colonoscopic finding revealed massive rectal bleeding from edematous and dilated rectal varices. The abdominal computed tomography (CT) taken before the surgery suggested the possibility of variceal bleeding on rectum. We tried a direct suture ligation but it was difficult due to the massive bleeding. Application of rectal pressure dressing with epinephrine (10 μg/ml, 40 ml) soaked gauze until second-look examinations. And vasopressin 0.4 units/min was infused to decrease portal blood flow and portal venous pressure. We decided to proceed with the LT and try the suture ligation again at the end of the surgery. After reperfusion, the femoral mean blood pressure dropped from 73 to 49 mmHg, and heart rate dropped from 85 to 60 beats/min. But it was normalized within 5 minutes after repeat bolus injection of epinephrine 10 μg twice at 3-minute intervals. Calcium gluconate and sodium bicarbonate were given intermittently, based on the results of arterial blood gas analyses. Without any complications during the intra-operative course after reperfusion, the hemodynamic status stabilized as surgery progressed. Vasopressin infusion was stopped. Norepinephrine and inotropics were gradually tapered down with continuous monitoring of the hemodynamic variables. At end of surgery, we examined the rectal bleeding site that showed shrunk rectal varices, compared with the previous view with the bleeding having stopped. We then decided to observe the progress of the patient.
During the operation, the patient was given about 20,500 ml of crystalloid, 1,000 ml of colloid, 5,000 ml of 5% albumin, 29 units of packed red blood cells, 2,100 ml of cell saved blood, 50 units of fresh frozen plasma, 8 units of apheresis platelets and 10 units of cryoprecipitate. Her total urine output was 1,080 ml.
The patient was transferred back to the intensive care unit after 14 hours and 10 minutes of surgery. Without any more of rectal bleeding during the post-operative course, she was transferred to the general ward on the second post-operative day in a stable condition.
Go to : 
DISCUSSION
Portal hypertension induced collateral vessels are fragile and can rupture easily, resulting in a large amount of blood loss, which can be fatal [1]. LT currently represents the ultimate therapy for bleeding collateral vessels in patients with liver cirrhosis, and it is the only therapy that cures both portal hypertension and the underlying liver disease. Early recognition of variceal bleeding may allow early managements before life-threatening hemorrhage occurs. However, we experienced massive bleeding of collateral vessels in rectum which is difficult to discern place during surgery.
Collateral vessels develop at specific places, such as esophagus, stomach, abdominal wall and rectum [1]. Ectopic varices are venous collaterals occurring anywhere in the abdomen except in the gastroesophageal region and represent up to 5% of all variceal bleeding [1,2]. There is considerable heterogeneity in the epidemiology of ectopic varices because of the modality of detection, etiology of portal hypertension, and significant interobserver variability [3].
Rectal varices are one of ectopic varices that are distinguished from hemorrhoids by their typical presence above the dentate line [4]. Rectal varices uncommonly bleed, but the bleeding can be massive enough to cause fatal results. Rectal varices have been reported in 10–40% of patients with liver cirrhosis undergoing colonoscopy [5,6]. These are dilated portosystemic collateral veins, which develop in patients with portal hypertension as a pathway for portal venous blood flow from the superior hemorrhoidal veins (portal) to and through the middle and inferior hemorrhoidal veins (systemic) [4].
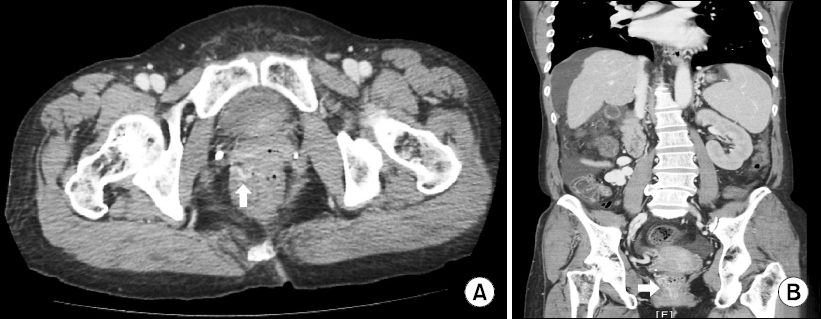
Guidelines have been established for the treatment of esophageal and gastric varices, whereas no unanimity of opinions has been obtained with regard to the treatment methods for ectopic varices [7]. Management of ectopic varices includes initiation of resuscitation in the form of replacing intravascular volume loss by crystalloids and blood transfusion and the administration of systemic vasopressin and octreotide. Once the patient is stabilized hemodynamically evaluate with emergency colonoscopy or intravenous contrast enhanced CT. Intravenous contrast enhanced CT may be preferable to colonoscopy in an unprepared acute setting and should be considered as the primary modality for diagnosis of rectal varices [4]. In our case, we also confirmed rectal varix with CT taken before surgery (Fig. 1). However, in our patient, occult bleeding from rectal varices did not present before surgery. So, she did not need any treatment for ectopic varix before surgery.
There are no randomized trials or set guidelines that dictate the management of bleeding ectopic varices [3]. The goal of treatment is to stop acute bleeding as soon as possible. The optimal therapeutic modality intervention for rectal varices involves medical, endoscopic, interventional radiological and surgical modalities. This depends on complications of the varices, the condition of the patient and the subsequent management plan.
Therapeutic endoscopic management with band ligation or endoscopic sclerotherapy is considered the urgent treatment [1]. In a case of refractory bleeding and for the prophylaxis of repeated bleeding, the portal hypertension which is underlying cause of bleeding must be treated. Treating methods include shunt procedure such as transjugular intrahepatic portosystemic shunt or a LT as the definitive therapy [1].
We tried to suture ligation for emergent control of bleeding but, failed. Therefore, we first decided to proceed with the LT and to make a second attempt at suture ligation at the end of the surgery. There were three reasons: First, the portal pressure, vessel size and varix wall tension are the major determinants of ectopic variceal hemorrhage [8]. There have been a few similar previous case reports of massive gastrointestinal bleeding from varices due to portal hypertension during the anhepatic phase when the hepatic portal vein was clamped [9,10]. It could be increased portal vein pressure by portal vein clamping in the anhepatic phase, leading to rectal varix bleeding. As such, the treatment of rectal varices bleeding after cross-clamping of the hepatic portal vein should be based on decompression of the portal pressure by the portal vein unclamping. Second, we tried to directly suture-ligate, but that was difficult due to massive bleeding by coagulopathy. LT currently represents the definitive treatment for severe portal hypertension and coagulopathy, resulting in bleeding varices and the transplantation provides an excellent outcome for the end stage liver disease. Third, varix bleeding was discovered in the anhepatic phase, LT could not be delayed.
It is known that direct surgery or local devascularization of the rectal varices is a useful minimally invasive procedure that does not take much time, and can be done even if the patient has Child B or C cirrhosis. However, in our case, emergency control of bleeding could not be obtained with suture ligation under severe portal hypertension and coagulopathy in anhepatic phase during the LT. Therefore, we tried to pressure dressing to achieve hemostasis and used vasopressin to decrease portal blood flow. At the end of the transplantation surgery, we found out that the rectal bleeding stopped. Possibly, reducing the portal pressure and correcting the coagulopathy might have helped to control the bleeding during surgery.
In summary, rectal variceal hemorrhage can occur due to an increase in portal vein pressure after cross-clamping of the inferior vena cava and portal vein during LT. Therefore, preoperative recognition of rectal varices may allow for surveillance and preemptive therapy before life-threatening hemorrhage occurs. Also, keep in mind that massive rectal bleeding could occur during LT under surgical drapes.
Go to : 




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download