Abstract
Objectives
The aim of this study was to investigate hearing and outer cells function in patients with psoriatic arthritis. Our investigation was a prospective case control study.
Methods
A total of 31 psoriatic arthritis patients (62 ears) and 31 healthy control subjects (62 ears) were enrolled in the study. We investigated hearing changes of patients and controls via pure tone audiometry, speech discrimination scores, tympanometry, acoustic reflex, and transient product otoacoustic emission.
Results
The mean age of psoriatic arthritis patients was 36.1±8.5 years (range, 14 to 62 years). The average age of the control group was 37.9±8.1 years (range, 16 to 62 years). There were statistically significant differences between pure tone audiometry in all frequencies and right and left emission at the 4.0 and 1.0 in psoriatic arthritis patients versus controls (P<0.05). This difference was evident, especially at high frequencies. There was no statistically significant difference between the ages and genders of the patient and control groups (P>0.05). Both audiological and otoacoustic emissions were not significantly different between right and left ear (P>0.05).
Conclusion
Based on the audiological and otoacoustic findings; it is likely that the cochlear outer hair cells become subtly damaged in psoriatic arthritis patients, consequently leading to changes in hearing thresholds. These data suggest that it is important to screen psoriatic arthritis patients for hearing changes with otoacoustic emissions and audiologic tests regularly.
Psoriatic arthritis (PsA) is an inflammatory arthritis interrelated with psoriasis (Ps) and 50% of PsA patients are seronegative for rheumatoid factor [1]. Frequently patients that are seronegative for rheumatoid factor are HLA-B27 positive and often exhibit extra-articular manifestations of the disease. The precise prevalence of PsA is unknown, but it is estimated to range from 0.02% and 0.42% in the general population and from 6% to 42% in patients with Ps. The average age of disease onset is reported to be between 36 to 40 years in most PsA studies [2]. Symptoms of PsA include pain, swelling or stiffness in one or more joints; sausage digits also known as dactylitis and pain in the feet and ankles, especially Achilles tendinitis or plantar fasciitis. Furthermore, nail changes with separation from the nail bed, ocular involvement, and aortic incompetence are all examples of extra-articular manifestations of PsA [3].
Recently it was found that bilateral sensorineural hearing loss (SHL) may be caused by autoimmune diseases such as mixed cryoglobulinemia, Cogan syndrome, Wegener granulomatosis, systemic sclerosis, giant cell arteritis, and relapsing polychondritis. The first autoimmune-mediated SHL was reported by McCabe [4] in 1979. After this time many studies have reported various autoimmune diseases that cause autoimmune ear disease (AIED) including rheumatoid arthritis, ankylosing spondylitis, Behçet disease, Sjögren syndrome, polyarteritis nodosa, and systemic lupus erythematosus [56789]. Even though many autoimmune diseases have been implicated in causing SHL, there is a paucity of data regarding hearing changes in patients with PsA.
To our best knowledge, there are no prospective studies about hearing in PsA. We were found only two case reports of hearing loss with PsA in PubMed. Srikumar et al. [10] reported a case of sudden onset SHL in a patient diagnosed with PsA. The other case study reported bilateral and asymmetric sensorineural deafness in a 13-year-old girl with juvenile PsA despite receiving treatment with etanercept [11].
Therefore, the aim of this study was to investigate hearing changes within in PsA patients by audiometric and otoacoustic emission (OAE) tests.
PsA patients receiving care from the Departments of Dermatology and Physical Therapy and Rehabilitation at the Dicle University Hospital from September 2012 to November 2013 were enrolled in the study. The number of patients was forty. However, five patients did not give consent form for performed audiological tests. One patient had history of ear surgery and three patients had a history of drug use (sulfasalazin) affecting hearing system. From these causes, we evaluated statistical analyses on thirty-one patients (62 ears). A total of 31 PsA patients made up the patient group and their ages ranged from 14 to 62 years. A total of 31 healthy controls were selected and their ages ranged from 16 to 62 years. We selected control patients from among accompanying persons who were healthy people without any discomfort. PsA was diagnosed according to the Classification Criteria for Psoriatic Arthritis (CASPAR) criteria [12]. Disease severity was evaluated by the Psoriasis Area and Severity Index (PASI) [13]. Thirty patients had used methotrexate and one patient had used tumor necrosis factor-alpha (TNF-α) inhibitors. The weekly dose of methotrexate was average 15 mg/kg and dose of etanercept was 50 mg/kg for body weight.
Exclusion criteria from the study were a history of trauma to the ears and/or head, barotrauma, ototoxic drug use (including sulfasalazine), previous otological surgery, and any otological disease such as otosclerosis, Meniere disease, or suppurative labyrinthitis. All participants received an otoscopic examination. Patients were removed from the study if the tympanic membrane was perforated or if there were signs of any other middle ear abnormality. Moreover, patients were excluded from the study if the tympanogram was flat or if there was an absence of acoustic reflexes. Overall, these exclusion criteria were used to select against patients with pre-existing inner ear pathology. Following ear, nose and throat examinations, all patients received pure tone audiometry (PTA) (Madsen OB 822 Clinical Audiometer), speech discrimination score (SDS) tympanometry (Clinical Middle Analyzer AZ 26, Interacoustic, Denmark, calibrated to ANSI S3.39-1987 standards), and transient evoked otoacoustic emissions (TEOAE) (Otodynamics Ltd., Herts, UK; EZ-screen, Interacoustic, Assens, Denmark) studies. Pure tone bone and air conduction audiometry were performed on all subjects at 250, 500, 1,000, 2,000, 4,000, and 6,000 Hz. Hearing loss as determined by PTA was measured by calculating the average hearing threshold at 0.5-4 kHz. High frequency of PTA was measured by calculating the average hearing threshold at 4,000 and 6,000 Hz. Data were processed and evaluated with OAE software (Otoscreen OAE Screening and NOAH based Software, Assens, Denmark). The signal-to-noise ratio (SNR) for each frequency was calculated. All patient and control subjects had normal peak compliances, peak pressures, gradients, ear canal volumes, and acoustic reflexes as defined by the American Speech Language and Hearing Association [14]. Informed consent was obtained from all study participants, and the research protocol was approved by the Ethics Committee (2012/456).
Data analysis was performed using SPSS ver. 15.0. (SPSS Inc., Chicago, IL, USA). According to result of central limit theorem 30 subjects are needed in each of the subgroupings if parametric tests are going to be used. The Kolmogorov-Smirnov test was used to determine whether continuous variables conformed to a normal distribution. Overall group comparisons between PsA patients and controls for data that were not normally distributed were performed with the Mann-Whitney U-test. If the distribution was normal in this case independent samples t-test was used. Categorical variables was analysed by using chi-square test. Non parametric Spearman bivariate correlation test was used to calculate the correlation between duration of disease versus hearing loss, PASI versus hearing loss and age versus hearing loss. Data were expressed as the mean±1SD for continuous or median and the range for categorical variables. A P-value less or equal to 0.05 was considered statistically significant.
On average the PsA patients were 36.1±8.5 years old and their ages ranged from 14 to 62 years. Overall there were 19 female and 12 male patients. The mean age of the control group was 37.9±8.1 years and their ages ranged from 16 to 62 years. There were 16 female and 15 male subjects in the control group. The age ranges according to the number of patients in study and control groups are shown in Table 1. Otoscopic examinations were normal for all study participants. There was no statistically significant difference between the age and gender distribution between the patient and control groups (P>0.05). Study subject demographics are shown in Table 2. PsA severity was evaluated by the PASI, and the mean PASI score was 16.3± 9.4 with scores ranging from 2 to 36. The mean duration of having a diagnosis of PsA was 5.5±3.8 years ranging from 1 to 15 years.
Normal peak compliance, gradient, peak pressure, ear canal volume, and acoustic reflexes were obtained with a 226-Hz probe. Discrepancies in SDSs were not observed between patient and control groups (P>0.05). The mean PTA for patients with PsA was 15.2±5.7 dB, and the mean PTA for the control group was 8.5±4.4 dB. Also the mean high frequency PTA as 26.7±6.8 for patients with PsA, and the mean PTA for the control group was 8.8±3 dB. The bone conduction threshold was taken into account for patients with PsA since an air-bone gap was not present. Comparing PTA between control and PsA subjects, statistically significant differences were observed over all frequencies (P<0.05). This difference was evident, especially at high frequencies for right and left ear (Figs. 1, 2). There was no statistically significant differences in SDS between PsA groups and control groups (P>0.05). In addition, age and PTA values were not correlated (P>0.05).
Differences in TEOAE were statistically significant at certain frequencies (right 4.0 and left emission at the 1.0) when contrasting the control and PsA groups (P<0.05). SNR values with frequency of TEOAE findings for all study subjects are summarized in Figs. 3, 4. There wasn't a significant association between the length of time having PsA and the severity of hearing loss (P>0.05). No correlation was identified between the PASI measurement of PsA severity and hearing loss (P>0.05) except at right ear audio at the 2,000 Hz. Also both audiological and OAEs were not significant difference between right and left ear (P>0.05).
As seen in these results, there were changes at the mean hearing threshold and OAEs in PsA compared to the controls. These results show important features in PsA besides other aortic incompetence. The exact mechanism of hearing changes hearing changes is not known in PsA because of the absence of any investigation up to the present. Also although the precise pathogenesis of PsA is unknown, there is evidence that immunologic, vascular, environmental, and genetic factors contribute to the persistence and predisposition of patients to PsA [15].
The inner ear can be a target of systemic or local autoimmune attack leading to SHL. Immune inner-ear disease causes those cases of cochleovestibular dysfunction related to other systemic immune-mediated illnesses, such as the collagen vascular diseases. In these cases, the inner ear may not be the primary target of an immune attack but is injured indirectly by immune complex deposition or other mechanisms. There were several studies about immune-mediated hearing loss. These have demonstrated that the inner ear was the source of the antigen, with the endolymphatic sac (ES) being the location of antigen processing that produced the local antibody response [1617]. Immunocompetent cells have been identified in the normal murine ES via immunohistochemistry, and it was demonstrated that CD4 cells were specifically localized in the epithelial perisaccular region and CD8 cells were rarely identified. Another immunohistochemical study of the extraosseous part of the ES revealed that T-helper cells (CD4 cells) predominate in the ES. There were B cells as well as macrophages in the lumen and perisaccular region as synovial fluid and synovial cultures [18]. Kumar et al. [19] reported that if peripheral T lymphocytes remain activated over long periods of time, as in chronic inflammatory diseases, they continue to produce high levels of proinflammatory cytokines such as interleukin (IL) 1, IL-6, and TNF-α that mediate cochlear degeneration. Studies have demonstrated that TNF-α blockers aid in the treatment of SHL. Specifically, it has been demonstrated that etanercept is effective in reducing cochlear inflammation and hearing loss. Probable immunological mechanisms that may play a role role in PsA and formation of hearing loss. Garcia Berrocal et al. [20] reported in two studies that a local immune response within the inner ear likely originates and propagates in the ES as immunocompetent cells are recruited from the bloodstream. It was reported that vascular disorders, viral infection, and autoimmune-mediated processes all contribute to the pathogenesis of labyrinthitis in animal models.
As a conclusion all of these studies, there seems to exist some relationship between physiological, biochemical, and morphological changes in the inner ear in various immunologic and pathologic conditions.
Diagnosing AIED is difficult that there is the absence of a 'gold standard' test. Thus, the diagnosis is based upon clinical evaluation and experience. To date, specific diagnostic tests are not available in diagnosis [21].
In our study, bilateral changes in hearing thresholds was more common in middle-aged females, which are both support diagnostic criteria for type 2 AIED. Lockshin [22] reported that the definition for type 2 AIED is hearing loss in the setting of a coexisting systemic autoimmune disease, and females are more commonly affected. Consistent with this definition, females made up the majority of the PsA patient group in our study. Karabulut et al. [23] investigated hearing and cochlear outer hair cell function in patients with Ps. They found that the symptoms of Ps occurred severely in middle-aged women, suggesting that age and gender may contribute to disease expression. Also our PsA patients were frequently middle aged female subjects but this result wasn't significantly different when we performed correlation with statistical method in our study (P>0.05). In addition in contrast to our results, Karabulut et al. [23] found no cochlear outer hair cell damage in psoriatic patients. PsA and Ps may be different entities. Although there is still some controversy about the existence of PsA as a specific form of inflammatory arthritis associated with Ps, epidemiological and clinical studies support the matchless features of PsA. Because of this difference difference, Ps and PsA may be associated with different disease courses [15].
There was no significant association found between PASI scores and the degree of changes in hearing. This data indicates that the degree of changes in hearing did not correlate with the severity of PsA symptoms. Also having a diagnosis of PsA over longer durations was not associated with increased changes in hearing thresholds. This result suggested that inner-ear injury depends on chronic damage to the cochlea due to impairment of the inner-ear microcirculation rather than on an acute inflammatory reactivation of the disease [24].
Bilateral high frequency SHL changes and some OAEs changes were seen with audiological tests in PsA patients. These resulsts suggest the presence of subtle deterioration in cochlear functions at the high frequency regions, indicating the basal turn of cochlea. Also SDS was not different between the patient and control group. This result supported changes of inner ear. This result was caused by cochlear outer hair cell involvement. Although audiologic and certain OAEs frequency changes were observed in PsA patients, we did not identify abnormalities in SDS, which suggests cochlear pathology. In general, PsA is a condition that promotes the formation of additional bone and it is possible that the autoimmune-mediated formation of fibro-osseous deposits in the cochlea might compromise outer hair cell integrity [25]. Early damage of outer hair cell integrity cannot be revealed unless OAEs [26]. In order to determine the health of these specialized cells, we measured OAEs that arise from outer hair cell vibratory motion. The basilar membrane contacts the outer hair cells in cochlea that permit sound wave transmission toward the stapedial footplate and eventually into the external auditory canal [27]. PsA patients demonstrated impaired OAEs and abnormal PTA results, which suggests that outer hair cell function is compromised in our study. Overall, our data reveal that PsA patients have impaired average hearing thresholds and OAEs.
A limitation of the study was that our PsA patients were on treatment at the time of measurements. Both methotrexate and biological agents are not definitive ototoxic drugs though. Since PsA is a relatively rare diagnosis, it was not possible to recruit patients who had not received any treatment.
In conclusion, as PsA patients demonstrated hearing impairments in our study, we recommend that these patients be routinely assessed for the hearing loss with OAEs and audiologic testing. Our study is the first study to find audiological differences in PsA, compared to healthy controls. Further long-term and multicentric studies are required to learn the exact nature of these changes.
ACKNOWLEDGMENTS
The authors thank Yusuf Celik, professor, from the Department of Bioistatistics, Dicle University Faculty of Medicine, for his valuable statistical assistance and Assist. Professor Yılmaz Palancı, from Department of Public Health, Dicle University Faculty of Medicine.
References
1. Wright V, Moll JM. Psoriatric arthritis. In : Wright V, Moll JM, editors. Seronegative polyarthritis. Amsterdam: North Holland Publishing Co.;1976. p. 169–235.
2. Alamanos Y, Voulgari PV, Drosos AA. Incidence and prevalence of psoriatic arthritis: a systematic review. J Rheumatol. 2008; 7. 35(7):1354–1358. PMID: 18464305.
3. Amherd-Hoekstra A, Naher H, Lorenz HM, Enk AH. Psoriatic arthritis: a review. J Dtsch Dermatol Ges. 2010; 5. 8(5):332–339. PMID: 20015187.

4. McCabe BF. Autoimmune sensorineural hearing loss. Ann Otol Rhinol Laryngol. 1979; Sep-Oct. 88(5 Pt 1):585–589. PMID: 496191.

5. Halligan CS, Bauch CD, Brey RH, Achenbach SJ, Bamlet WR, McDonald TJ, et al. Hearing loss in rheumatoid arthritis. Laryngoscope. 2006; 11. 116(11):2044–2049. PMID: 17075400.

6. Dagli M, Sivas Acar F, Karabulut H, Eryilmaz A, Erkol Inal E. Evaluation of hearing and cochlear function by DPOAE and audiometric tests in patients with ankylosing spondilitis. Rheumatol Int. 2007; 4. 27(6):511–516. PMID: 17094003.

7. Dagli M, Eryilmaz A, Tanrikulu S, Aydin A, Gonul M, Gul U, et al. Evaluation of cochlear involvement by distortion product otoacoustic emission in Behcet's disease. Auris Nasus Larynx. 2008; 9. 35(3):333–337. PMID: 17996415.
8. Hatzopoulos S, Amoroso C, Aimoni C, Lo Monaco A, Govoni M, Martini A. Hearing loss evaluation of Sjogren's syndrome using distortion product otoacoustic emissions. Acta Otolaryngol Suppl. 2002; (548):20–25. PMID: 12211351.

9. Karatas E, Onat AM, Durucu C, Baglam T, Kanlikama M, Altunoren O, et al. Audiovestibular disturbance in patients with systemic lupus erythematosus. Otolaryngol Head Neck Surg. 2007; 1. 136(1):82–86. PMID: 17210339.

10. Srikumar S, Deepak MK, Basu S, Kumar BN. Sensorineural hearing loss associated with psoriatic arthritis. J Laryngol Otol. 2004; 11. 118(11):909–911. PMID: 15638985.

11. Giani T, Simonini G, Lunardi C, Puccetti A, De Martino M, Falcini F. Juvenile psoriatic arthritis and acquired sensorineural hearing loss in a teenager: is there an association? Clin Exp Rheumatol. 2006; May-Jun. 24(3):344–346. PMID: 16870107.
12. Taylor W, Gladman D, Helliwell P, Marchesoni A, Mease P, Mielants H, et al. Classification criteria for psoriatic arthritis: development of new criteria from a large international study. Arthritis Rheum. 2006; 8. 54(8):2665–2673. PMID: 16871531.

13. van de Kerkhof PC. The Psoriasis Area and Severity Index and alternative approaches for the assessment of severity: persisting areas of confusion. Br J Dermatol. 1997; 10. 137(4):661–662. PMID: 9390363.
14. American Speech-Language-Hearing Association. Guidelines for screening for hearing impairment and middle-ear disorders. Working Group on Acoustic Immittance Measurements and the Committee on Audiologic Evaluation. ASHA Suppl. 1990; 4. (2):17–24. PMID: 1970249.
15. Gladman DD. Current concepts in psoriatic arthritis. Curr Opin Rheumatol. 2002; 7. 14(4):361–366. PMID: 12118168.

16. Harris JP. Immunology of the inner ear: evidence of local antibody production. Ann Otol Rhinol Laryngol. 1984; Mar-Apr. 93(2 Pt 1):157–162. PMID: 6712089.

17. Mogi G, Kawauchi H, Suzuki M, Sato N. Inner ear immunology. Am J Otolaryngol. 1985; May-Jun. 6(3):142–147. PMID: 4014576.
18. Yoo TJ, Yazawa Y. Immunology of cochlear and vestibular disorders. In : Luxon LM, Furman JM, Martini A, Stephens D, editors. Textbook of audiological medicine: clinical aspects of hearing and balance. London: Taylor & Francis Group;2003. p. 61–88.
19. Kumar BN, Walsh RM, Wilson PS, Carlin WV. Sensorineural hearing loss and ulcerative colitis. J Laryngol Otol. 1997; 3. 111(3):277–278. PMID: 9156068.

20. Garcia Berrocal JR, Ramirez-Camacho R, Vargas JA, Millan I. Does the serological testing really play a role in the diagnosis immune-mediated inner ear disease? Acta Otolaryngol. 2002; 4. 122(3):243–248. PMID: 12030569.
21. Bovo R, Aimoni C, Martini A. Immune-mediated inner ear disease. Acta Otolaryngol. 2006; 10. 126(10):1012–1021. PMID: 16923703.

22. Lockshin MD. Sex differences in autoimmune disease. Lupus. 2006; 11. 15(11):753–756. PMID: 17153846.

23. Karabulut H, Karadag AS, Dagli M, Acar B, Babademez MA, Sahin Y, et al. Investigation of hearing and outer hair cell function of cochlea in patients with psoriasis. Int Adv Otol. 2010; 1. 6(2):239–244.
24. Salvinelli F, D'Ascanio L, Casale M. Staging rheumatoid arthritis: what about otoacoustic emissions? Acta Otolaryngol. 2004; 9. 124(7):874–875. PMID: 15370576.

25. Harris JP, Keithley EM, Gopen Q. Autoimmune ear disease and other autoimmune diseases with inner ear involvement. In : Snow JB, editor. Ballenger's manual of otorhinolaryngology head and neck surgery. Hamilton: BC Decker;2002. p. 396–407.
26. Bayindir T, Filiz A, Iraz M, Kaya S, Tan M, Kalcioglu MT. Evaluation of the protective effect of Beta glucan on amikacin ototoxicity using distortion product otoacoustic emission measurements in rats. Clin Exp Otorhinolaryngol. 2013; 3. 6(1):1–6. PMID: 23525870.

27. Prieve BA, Fitzgerald TS. Otoacoustic emissions. In : Katz J, Burkard R, Medwetsky L, editors. Handbook of clinical audiology. 5th ed. Philadelphia: Lippincott Williams & Wilkins;2002. p. 440–466.
Fig. 1
Right audiometric graphic of frequency and pure tone thresholds of patient and control groups. Frequency of right audiometry: 250 Hz, P<0.001; 500 Hz, P<0.001; 1,000 Hz, P=0.011; 2,000 Hz, P<0.001; 4,000 Hz, P=0.001; 6,000 Hz, P<0.001.
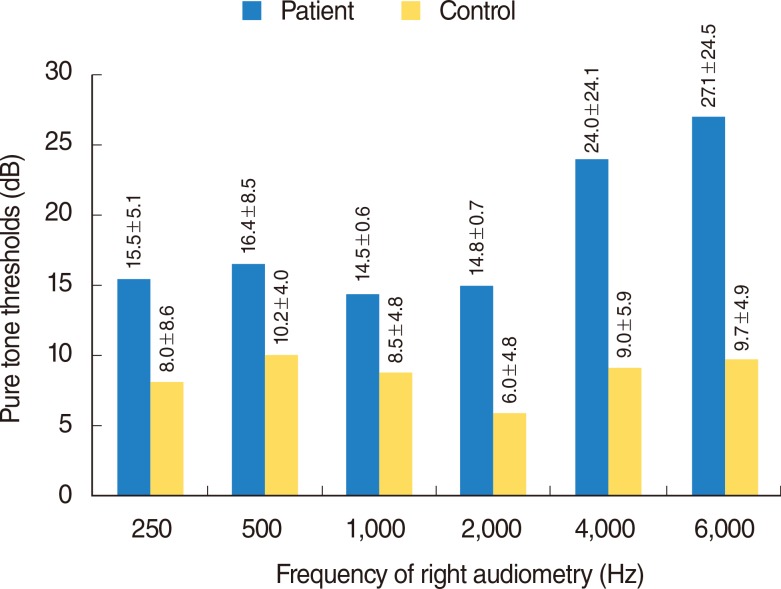
Fig. 2
Left audiometric graphic of frequency and pure tone thresholds of patient and control groups. Frequency of left audiometry: 250 Hz, P=0.013; 500 Hz, P=0.023; 1,000 Hz, P=0.007; 2,000 Hz, P=0.002; 4,000 Hz, P=0.002; 6,000 Hz, P<0.001.

Fig. 3
Right otoacoustic emission graphic of frequency and sound noise ratio changes of patient and control groups. Frequency of right otoacoustic emission: 1.0 Hz, P=0.053; 1.4 Hz, P=0.683; 2.0 Hz, P=0.989; 2.8 Hz, P=0.714; 4.0 Hz, P=0.024. Mann-Whitney U-test.
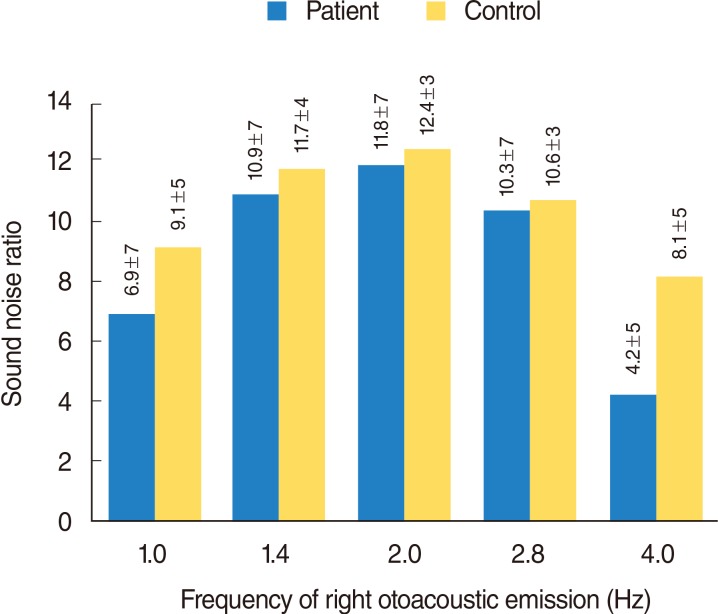
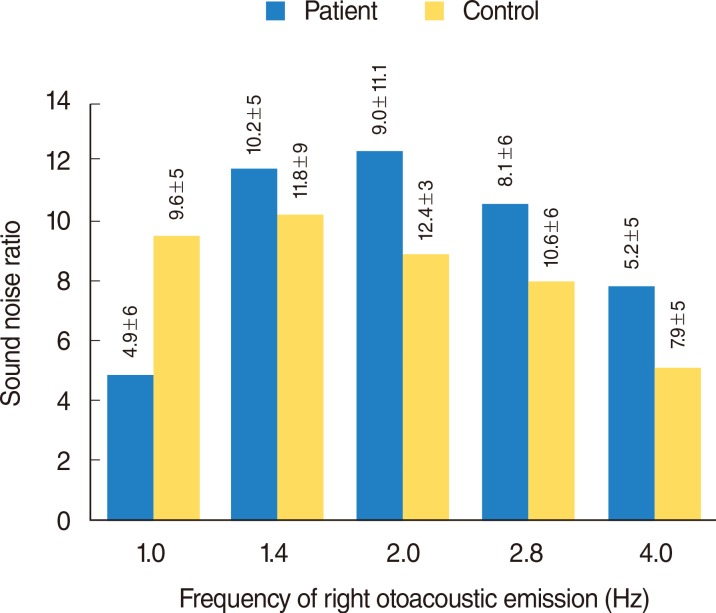
Fig. 4
Left otoacoustic emission graphic of frequency and sound noise ratio changes of patient and control groups. Frequency of left otoacoustic emission: 1.0 Hz, P=0.005; 1.4 Hz, P=0.213; 2.0 Hz, P=0.357; 2.8 Hz, P=0.212; 4.0 Hz, P=0.076. Mann-Whitney U-test.




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download