Abstract
Objectives
The microRNAs have been implicated in the development and function of the inner ear, especially in contribution to hearing. However, the impact of idiopathic sudden sensorineural hearing loss (SSNHL) on expression of miRNA biogenesis-related components has not been established. To investigate the regulations of microRNA (miRNA) biogenesis-related components, argonaute 2 (AGO2) and DiGeorge syndrome critical region gene 8 (DGCR8) mRNA expression in SSNHL and to evaluate the value of clinical parameters on their expression.
Methods
Thirty-seven patients diagnosed with SSNHL and fifty-one healthy volunteers were included in this study. We measured mRNA expression levels of AGO2 and DGCR8 in whole blood cells but erythrocytes of patients with SSNHL and controls, using reverse transcription and real-time polymerase chain reaction analysis.
Idiopathic sudden sensorineural hearing loss (SSNHL), also known as sudden deafness, is defined as more than 30 dB sensorineural hearing loss over three continuous test frequencies occurring within a 72 hours period [1]. SSNHL has been reported to have an incidence of between 5 to 20 cases per 100,000 populations per year [2]. Although spontaneous recovery in 45% to 65% of patients, permanent hearing loss remains in an important clinical issue [3]. Several suggested theories attempt to elucidate the aetiopathogenesis of idiopathic SSNHL, including viral infection, vascular impairment, autoimmune diseases, and cochlear membrane ruptures [3,4,5], however, which mechanism is involved in developing SSNHL is not yet to be adequately explained. Among the theories being investigated were the possibilities that genetic risk factors made a significant contribution to SSNHL by our study group [6,7].
MicroRNAs (miRNAs) are new class of highly conserved and small noncoding RNAs that can negatively regulate gene expression by degradation and translational inhibition of their target mRNAs [8]. The biogenesis of miRNA is a well-organized process and accomplished by several important components including the DiGeorge syndrome critical region gene 8 (DGCR8 or pasha) and argonaute 2 (AGO2) [8,9]. DGCR8 is a factor of the microprocessor complex and has been shown to be necessary for miRNAs maturation [9]. Within the cytoplasm, the pre-miRNAs are further processed by multidomain. The gene expression regulating effects of miRNAs are accomplished by the RNA-induced silencing complex (RISC), multiprotein effector complex with endonuclease activity, which integrates mature miRNA strands [8]. The RISC is the main element of the RNA silencing process and consist of several different proteins that comprise multiprotein complex, including AGO1, AGO2, and the double-stranded RNA-binding protein [10].
In the recent studies, miRNA have received increasing attention because of their implication in hair cell development and regeneration and in degeneration of the organ of corti during age-related hearing loss [11,12]. The expression of miRNA biogenesis pathway proteins, DGCR8 and AGO2 directly influence the biosynthesis of all miRNAs, severely affecting their target the gene expression pattern. If any miRNA biogenesis-related component is dysregulated, miRNA maturation could be significantly altered. The dysregulated expression of important individual miRNA biogenesis-related component has recently been shown in various human disease, including respiratory syncytial virus disease [13], neurological diseases [14], cancer [10], and immunologic diseases [15]. As a result, it was hypothesized that the miRNA biogenesis pathway may be involved in the idiopathic SSNHL.
In the present study, we aimed to investigate the mRNA expression levels of DGCR8 and AGO2 in whole blood cells but erythrocytes of patients with SSNHL and healthy controls, and examined the correlation of the mRNA levels of these miRNA machinery components with various clinical parameters.
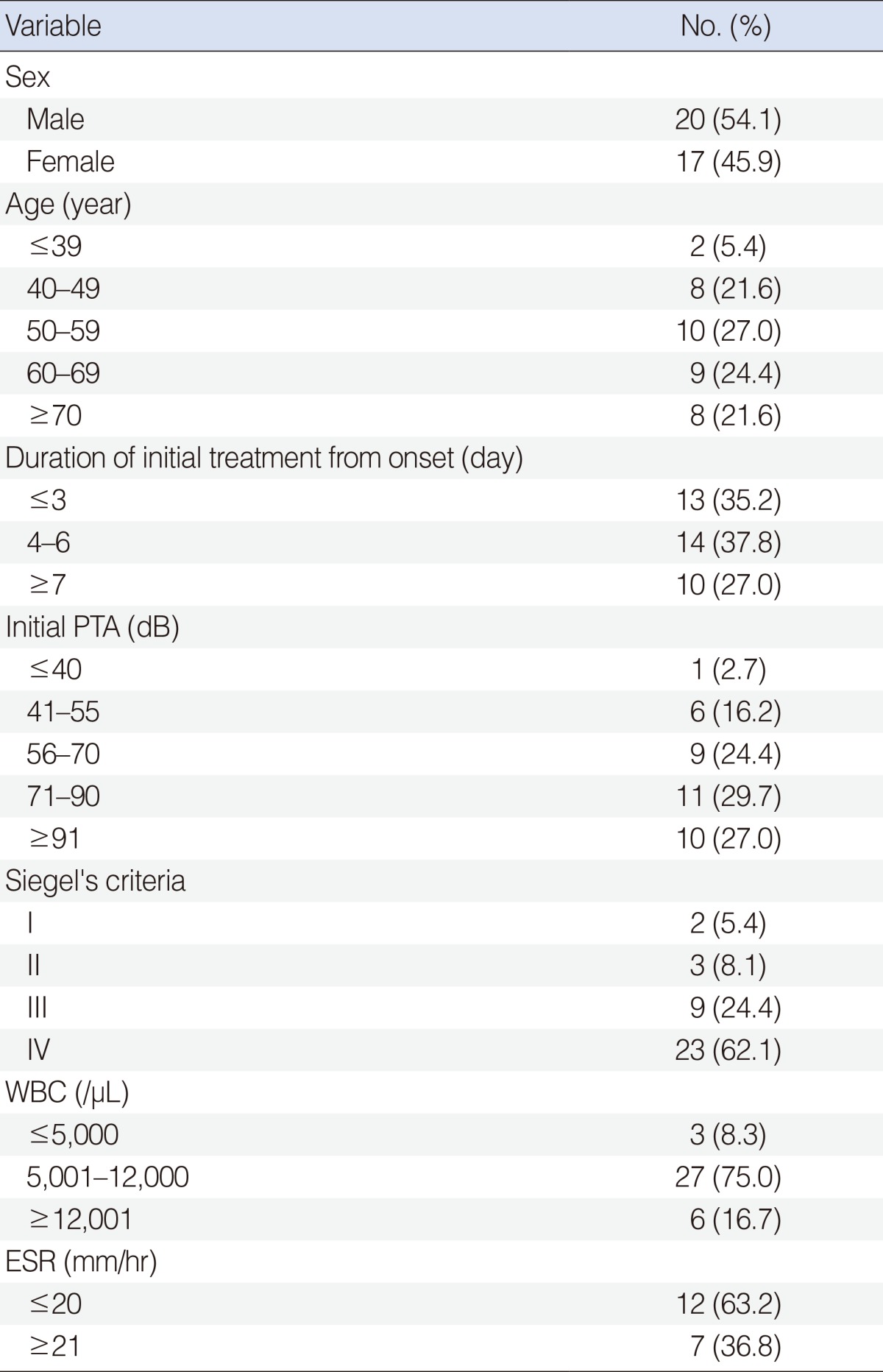
Thirty-seven patients diagnosed with SSNHL between April 2012 and September 2012 at Dongsan Medical Center by well-trained doctors specialized in ear, nose and throat. Patients were included according to the following diagnostic criteria: >30 dB sensorineural hearing loss in more than 3 consecutive frequencies by pure tone audiogram with onset within 3 days, and no involvement of cranial nerves other than the eighth cranial nerve. SSHNL patients were grouped into several clinical characteristics (Table 1). Fifty-one healthy volunteers, Korean who underwent physical examinations and with no clinical evidence of SSNHL or any other disorders were collected as controls. Blood samples were taken from SSHNL patients in the acute phase of hearing loss. All patients and healthy volunteers were explained the study purpose, and informed consent was obtained from each study participant. The protocols were approved by the Institutional Review Board of Keimyung University Dongsan Medical Center (approval #12-119).
Peripheral blood was obtained from healthy volunteers and patients with SSNHL. Blood specimens were collected into ethylenediaminetetraacetic acid (EDTA) tubes to avoid clots. EDTA tubes were centrifuged and then phosphate buffered saline (PBS) was added to wash. The erythrocytes were removed twice with ACK lysis buffer (Gibco, Grand Island, NY, USA).
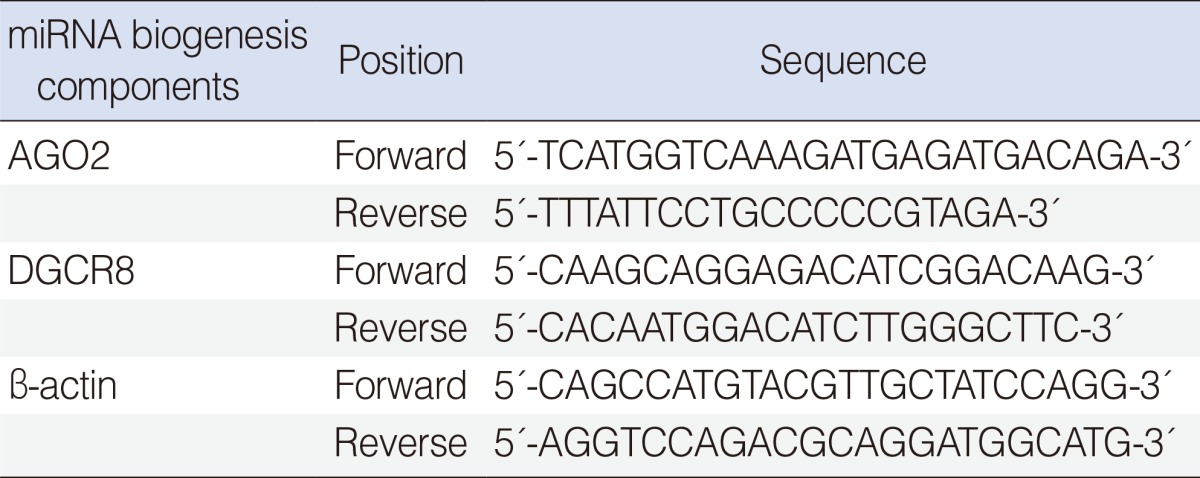
The mRNA expression of components of the miRNA biogenesis including DGCR8 and AGO2 was detected by means of quantitative reverse transcription polymerase chain reaction (RT-PCR). Total cellular RNA was extracted from whole blood cells but erythrocytes using TRIzol (Invitrogen, Calsbad, CA, USA) and treated with a DNAse to remove DNA. Each cDNA was synthesized form 2 g of total RNA using M-MLV reverse transcriptase (Promega, Madison, WI, USA). By using the specific primer pairs described in Table 2 and SYBR GREEN Premix (TOYOBO, Osaka, Japan). Quantitative real-time PCR was performed on the LightCycler 480 real-time PCR system (Roche, Mannheim, Germany). Relative gene expression was calculated by using the comparative CT (ΔCt) method (ΔCt=specific target gene Ct-β-actin Ct) [16]. β-actin was used as a housekeeping gene for normalization, and a no template sample was used as a negative control.
Statistical analysis was performed with SPSS ver. 18.0 (SPSS Inc., Chicago, IL, USA). In order to compare the mRNA expression levels of miRNA biogenesis-related components between patients with SSNHL and healthy volunteers, the data were analyzed using the Mann-Whitney U-test of variance for continuous variables. Correlations between mRNA expressions of interindividual miRNA biogenesis-related components were analyzed by the Pearson correlation coefficient analysis. Statistical analyses between each miRNA biogenesis-related component and each clinical characteristic were performed by using the Kruskal-Wallis test for categorical variables. Generally, P<0.05 was established to denote significance in all statistical analyses performed in the study.
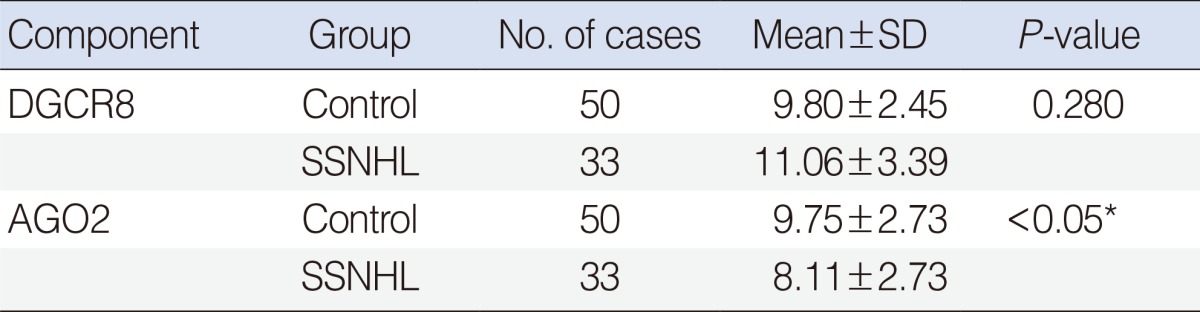
To investigate whether the significant changes on the mRNA expression levels of miRNA biogenesis-related components in the specimens of patients with SSNHL, we evaluated the mRNA expression levels of DGCR8 and AGO2 in patients with SSNHL and healthy controls. Prior to statistical analysis, raw qPCR data of DGCR8 and AGO2 mRNA expression were normalized to reference gene, β-actin. The AGO2 expression level was significantly higher in patients with SSNHL (P<0.05) (Table 3, Fig. 1). However, in the expression level of DGCR8, there were no significant differences between patients with SSNHL and healthy controls (Table 3, Fig. 1).
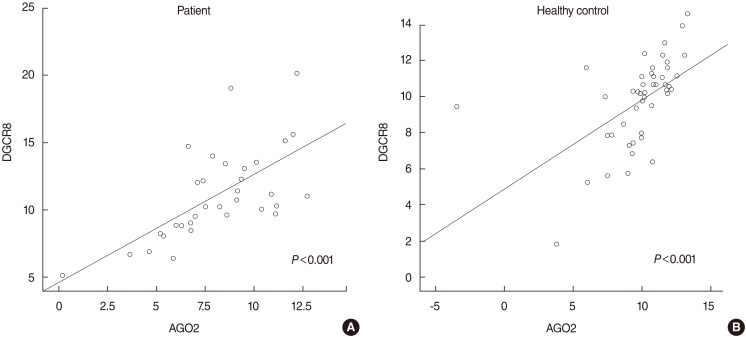
There was a significant correlation between AGO2 and DGCR8 with Pearson correlation coefficient value of 0.650 and 0.551 was found in patients with SSNHL (P<0.001) and healthy control (P<0.001), respectively (Fig. 2).
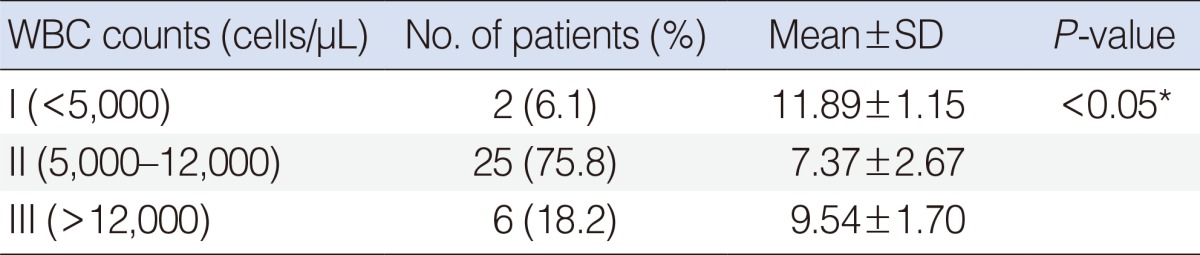
To evaluate the influence of the clinical characteristics on the mRNA expression levels of AGO2 and DGCR8, the patients were classified according to each clinical characteristic. The clinical characteristics of all 37 patients (mean age, 57.51±12.56 years) with SSNHL were presented in Table 1. As shown in Table 4, the AGO2 mRNA expression level was significantly associated with WBC count in SSNHL patients (P<0.05). This statistical analysis was performed by using the Kruskal-Wallis test for categorical variables. There were no significant associations between expression levels of the DGCR8 and each clinical characteristic after the same analytic method (data not shown).
The discovery that gene expression can be altered through RNA interference [17] has stimulated research on the role of RNA interference in the development of various human diseases. RNA interference such as miRNA or siRNA could influence expression patterns of various genes by inhibiting mRNA expression. The maturation of miRNA or siRNA is highly intricate and tightly controlled processes, regulated by miRNA biogenesis-related components. Dysregulation of miRNA biogenesis frequently causes diseases in humans. Alteration of miRNA biogenesis-related components in various human diseases has been reported. For example, it has been revealed that DGCR8 is upregulated in colorectal carcinoma [18], and protein activator of the interferon-induced protein kinase (PACT) mRNA expression was found to be altered in chronic rhinosinusitis [19]. In addition, reduced Dicer expression in the cord blood of infants admitted with severe respiratory syncytial virus disease [13]. But, the regulation of miRNA biogenesis-related components has not been evaluated in SSNHL.
In this study, we investigated the mRNA expression levels of miRNA biogenesis-related components such as DGCR8 and AGO2 in SSNHL. Using a quantitative real-time PCR approach, as shown in Fig. 1, we found that the AGO2 mRNA expression level is significantly upregulated in patients with SSNHL in comparison to healthy controls. These results may not reflect what's going on in the inner ear because we evaluated only through transcripts from the peripheral blood cells not the inner ear cells. Although, more research is needed on the regulations of the AGO2 in many other disease, this result suggest that the upregulated mRNA expression level of AGO2 is involved in SSNHL pathogenesis. In a recent study, mRNA levels of DGCR8 and AGO2 have been appeared positively correlated to each other in colorectal carcinoma [18]. So, in the present study, the correlation between expression levels of interindividual miRNA biogenesis-related components was evaluated by using the Pearson correlation coefficient analysis. In both SSNHLs and controls, a significant correlation between DGCR8 and AGO2 was found (Fig. 2). Based on the finding that significant correlations between DGCR8 and AGO2 obtained from statistical analyses in SSNHLs or controls, unlike reported results [18], the correlations between DGCR8 and AGO2 are not a specific epiphenomenon or a pathogenesis of SSNHL.
Altered expressions of miRNA biogenesis-related components are associated with various human diseases. Although it is difficult to know whether the dysregulated component is cause or outcome in a pathogenesis of the diseases, it has been reported that there were significant correlations between the dysregulated components and clinical characteristics [20]. It has been reported that various prognostic factors are significantly associated with clinical characteristics of SSNHL. Known several prognostic factors associated with SSNHL are initial hearing loss level, audiogram patterns, age, presence or absence of vertigo and tinnitus, duration of initial treatment from onset, underlying disease [2,21,22]. Enache and Sarafoleanu [23] described good prognosis in the hearing recovery of SSNHL was associated with absence of vertigo, early treatment (the first 7 days), and initial hearing loss less than 50 dB. Wei and He [24] described the factor of age, the trouble time before treatment, vertigo with (or) tinnitus, degree of hearing loss, the patterns of audiogram are correlated with prognosis of SSNHL. Moreover, to the best of our knowledge, this is the first study to investigate correlations between expressions of miRNA biogenesis-related components and clinical characteristics in SSNHL. We analyzed the influence of clinical characteristics, including sex, age, the duration of initial treatment from onset, initial pure tone average, white blood cell (WBC) count, Siegel's criteria, and erythrocyte sedimentation rate in SSNHL by using the Kruskal-Wallis for categorical variables. The WBC counts were divided into three groups depending upon WBC numbers per microliter blood in an attempt to find out association with mRNA expression levels of AGO2 (Table 4). The AGO2 mRNA expression level was significantly associated with the groups of WBC counts in SSNHL (Table 4). It has been reported that WBC counts are a biomarker related to systemic stress and cochlear inflammation [25]. Additionally, several systemic inflammatory processes can be detected by examination of the blood, which may also be a very promising approach for monitoring possible pathogenetic mechanisms associated with SSNHL [26]. In this study, downregulated AGO2 mRNA expression level (I, 11.89±1.15; III, 9.54±1.7) was found in abnormal WBC counts group I and III in comparison with normal WBC counts group II (7.37±2.67). An elevated WBC count does not indicate a specific disease by itself, however, is correlated with infection, systemic illness, inflammation, allergy, leukemia, and tissue injury caused due to burns, etc. Although, more research is needed on the relationship between altered AGO2 mRNA expression level and aberrant WBC counts as a inflammatory marker, this finding suggested that downregulated AGO2 mRNA expression level in the patient with SSNHL may be associated with cochlear inflammation or systemic inflammatory condition in the disease.
The current study showed that the expression of important miRNA biogenesis-related components is disturbed in SSNHL patients' whole blood cells except erythrocyte. Although, based on the data, it doesn't provide clear aetiopathogenesis of SSNHL, this study would be expected to help investigating SSNHL as a good research starting point.
In this study, we investigated the mRNA expression levels of two selected miRNA machinery components, AGO2, and DGCR8, and their clinical association in SSNHL for the first time. Our data demonstrated that AGO2, a RISC component, is significantly up-regulated in SSNHL, suggesting that reduced expression of AGO2 may play an important role in the development of SSNHL.
References
1. Stachler RJ, Chandrasekhar SS, Archer SM, Rosenfeld RM, Schwartz SR, Barrs DM, et al. Clinical practice guideline: sudden hearing loss. Otolaryngol Head Neck Surg. 2012; 3. 146(3 Suppl):S1–S35. PMID: 22383545.

2. Byl FM Jr. Sudden hearing loss: eight years' experience and suggested prognostic table. Laryngoscope. 1984; 5. 94(5 Pt 1):647–661. PMID: 6325838.
3. Zadeh MH, Storper IS, Spitzer JB. Diagnosis and treatment of sudden-onset sensorineural hearing loss: a study of 51 patients. Otolaryngol Head Neck Surg. 2003; 1. 128(1):92–98. PMID: 12574765.

4. Yossepowitch O, Lossos A, Lossos IS. Sudden hearing loss following acute hepatitis. Postgrad Med J. 1999; 5. 75(883):309–312. PMID: 10533644.

5. Suckfull M. Perspectives on the pathophysiology and treatment of sudden idiopathic sensorineural hearing loss. Dtsch Arztebl Int. 2009; 10. 106(41):669–675. PMID: 19946432.
6. Nam SI, Yu GI, Kim HJ, Park KO, Chung JH, Ha E, et al. A polymorphism at -1607 2G in the matrix metalloproteinase-1 (MMP-1) increased risk of sudden deafness in Korean population but not at -519A/G in MMP-1. Laryngoscope. 2011; 1. 121(1):171–175. PMID: 21154774.

7. Nam SI, Ha E, Jung KH, Baik HH, Yoon SH, Park HJ, et al. IL4 receptor polymorphism is associated with increased risk of sudden deafness in Korean population. Life Sci. 2006; 1. 78(6):664–667. PMID: 16280132.
8. Bartel DP. MicroRNAs: genomics, biogenesis, mechanism, and function. Cell. 2004; 1. 116(2):281–297. PMID: 14744438.
9. Gregory RI, Yan KP, Amuthan G, Chendrimada T, Doratotaj B, Cooch N, et al. The Microprocessor complex mediates the genesis of microRNAs. Nature. 2004; 11. 432(7014):235–240. PMID: 15531877.

10. Sand M, Skrygan M, Georgas D, Arenz C, Gambichler T, Sand D, et al. Expression levels of the microRNA maturing microprocessor complex component DGCR8 and the RNA-induced silencing complex (RISC) components argonaute-1, argonaute-2, PACT, TARBP1, and TARBP2 in epithelial skin cancer. Mol Carcinog. 2012; 11. 51(11):916–922. PMID: 22025453.

11. Groves AK, Zhang KD, Fekete DM. The genetics of hair cell development and regeneration. Annu Rev Neurosci. 2013; 7. 36:361–381. PMID: 23724999.

12. Zhang Q, Liu H, McGee J, Walsh EJ, Soukup GA, He DZ. Identifying microRNAs involved in degeneration of the organ of corti during age-related hearing loss. PLoS One. 2013; 4. 8(4):e62786. PMID: 23646144.

13. Inchley CS, Sonerud T, Fjærli HO, Nakstad B. Reduced Dicer expression in the cord blood of infants admitted with severe respiratory syncytial virus disease. BMC Infect Dis. 2011; 3. 11:59. PMID: 21385408.

14. Haussecker D, Huang Y, Lau A, Parameswaran P, Fire AZ, Kay MA. Human tRNA-derived small RNAs in the global regulation of RNA silencing. RNA. 2010; 4. 16(4):673–695. PMID: 20181738.

15. Smith KM, Guerau-de-Arellano M, Costinean S, Williams JL, Bottoni A, Mavrikis Cox G, et al. miR-29ab1 deficiency identifies a negative feedback loop controlling Th1 bias that is dysregulated in multiple sclerosis. J Immunol. 2012; 8. 189(4):1567–1576. PMID: 22772450.

16. Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc. 2008; 3(6):1101–1108. PMID: 18546601.

17. Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature. 1998; 2. 391(6669):806–811. PMID: 9486653.

18. Kim B, Lee JH, Park JW, Kwon TK, Baek SK, Hwang I, et al. An essential microRNA maturing microprocessor complex component DGCR8 is up-regulated in colorectal carcinomas. Clin Exp Med. 2014; 8. 14(3):331–336. PMID: 23775303.

19. Zhang YN, Cao PP, Zhang XH, Lu X, Liu Z. Expression of microRNA machinery proteins in different types of chronic rhinosinusitis. Laryngoscope. 2012; 12. 122(12):2621–2627. PMID: 22961479.

20. Mockenhaupt S, Schurmann N, Grimm D. When cellular networks run out of control: global dysregulation of the RNAi machinery in human pathology and therapy. Prog Mol Biol Transl Sci. 2011; 102:165–242. PMID: 21846572.
21. Jaffe BF. Clinical studies in sudden deafness. Adv Otorhinolaryngol. 1973; 20:221–228. PMID: 4351035.

22. Saeki N, Kitahara M. Assessment of prognosis in sudden deafness. Acta Otolaryngol Suppl. 1994; 510:56–61. PMID: 8128875.

23. Enache R, Sarafoleanu C. Prognostic factors in sudden hearing loss. J Med Life. 2008; Jul-Sep. 1(3):343–347. PMID: 20108511.
24. Wei X, He J. Analysis of prognostic factors for sudden sensorineural hearing loss. Lin Chung Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2011; 7. 25(13):599–601. PMID: 21949992.
25. Masuda M, Kanzaki S, Minami S, Kikuchi J, Kanzaki J, Sato H, et al. Correlations of inflammatory biomarkers with the onset and prognosis of idiopathic sudden sensorineural hearing loss. Otol Neurotol. 2012; 9. 33(7):1142–1150. PMID: 22872174.

26. Kassner SS, Schottler S, Bonaterra GA, Stern-Strater J, Sommer U, Hormann K, et al. Proinflammatory and proadhesive activation of lymphocytes and macrophages in sudden sensorineural hearing loss. Audiol Neurootol. 2011; 16(4):254–262. PMID: 20980746.

Fig. 1
The relative mRNA expression levels of AGO2 and DGCR8 in patients with sudden sensorineural hearing loss and healthy controls (mean±SD). *P<0.05. AGO2, argonaute-2; DGCR8, DiGeorge syndrome critical region gene 8.

Fig. 2
Correlation between AGO2 and DGCR8 mRNA expression levels in patients with sudden sensorineural hearing loss and healthy controls. AGO2, argonaute-2; DGCR8, DiGeorge syndrome critical region gene 8.




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download