Abstract
Objectives
Methods
Results
ACKNOWLEDGMENTS
REFERENCES
Fig. 1.
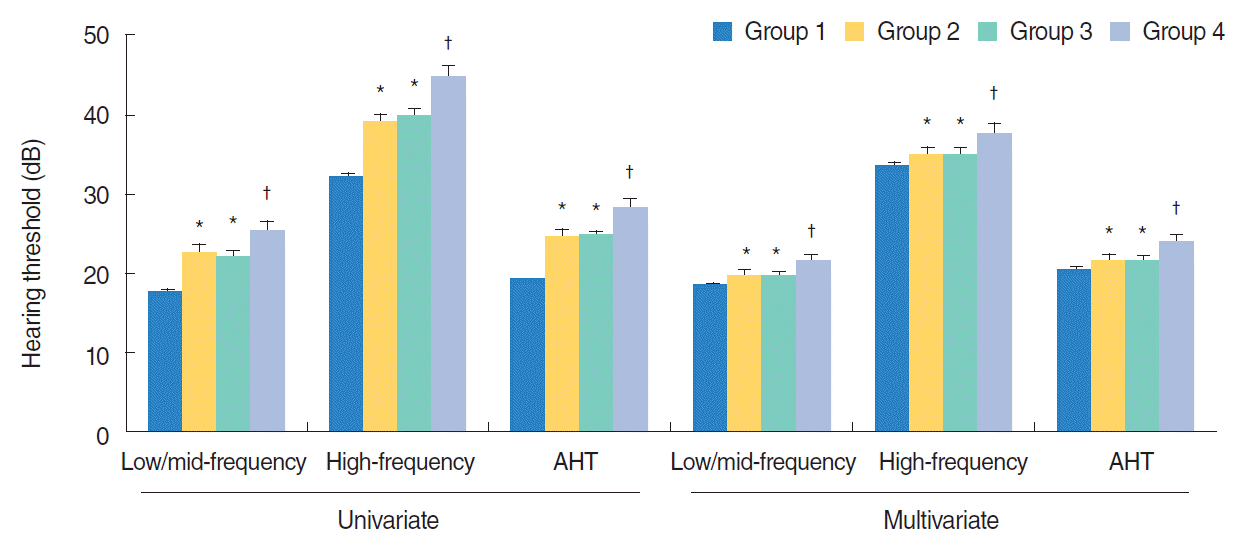
Table 1.
Values are presented as number (%) or mean±standard deviation.
The P-values were tested using one-way analysis of variance for continuous variables and Pearson chi-square test or Fisher exact test for categorical variables.
BMI, body mass index; eGFR, estimated glomerular filtration rate; HbA1c, glycated hemoglobin; SBP, systolic blood pressure; DBP, diastolic blood pressure; UACR, urine albumin/creatinine ratio; HDL, high-density lipoprotein.
Table 2.
| Variable |
Univariatea) |
Multivariatea) |
||
|---|---|---|---|---|
| OR (95% CI) | P-value | OR (95% CI) | P-value | |
| Group 1 | 1.00 | 1.00 | ||
| Group 1 vs. group 2 | 1.762 (1.471–2.110) | <0.001 | 1.043 (0.840–1.295) | 0.700 |
| Group 1 vs. group 3 | 1.859 (1.646–2.099) | <0.001 | 1.155 (0.961–1.387) | 0.125 |
| Group 1 vs. group 4 | 2.803 (2.280–3.446) | <0.001 | 1.557 (1.168–2.075) | 0.003 |
| Group 2 | 1.00 | 1.00 | ||
| Group 2 vs. group 3 | 1.055 (0.860–1.295) | 0.608 | 1.077 (0.819–1.416) | 0.597 |
| Group 2 vs. group 4 | 1.591 (1.222–2.072) | 0.001 | 1.456 (1.023–2.072) | 0.037 |
| Group 3 | 1.00 | 1.00 | ||
| Group 3 vs. group 4 | 1.508 (1.200–1.895) | <0.001 | 1.321 (1.005–1.738) | 0.046 |
a) Hearing loss (average hearing threshold>25 dB), and the multivariate analysis was adjusted for age, sex, high-density lipoprotein cholesterol levels, triglyceride levels, estimated glomerular filtration rate, systolic blood pressure, diastolic blood pressure, body mass index, alcohol consumption, smoking behavior, glycated hemoglobin levels, and exposure to explosives or occupational noise.
Table 3.
| Variable |
Mild hearing loss |
Moderate to severe hearing loss |
||||||
|---|---|---|---|---|---|---|---|---|
|
Univariatea) |
Multivariatea) |
Univariatea) |
Multivariatea) |
|||||
| OR (95% CI) | P-value | OR (95% CI) | P-value | OR (95% CI) | P-value | OR (95% CI) | P-value | |
| Group 1 | 1.00 | 1.00 | 1.00 | 1.00 | ||||
| Group 1 vs. group 2 | 1.563 (1.256–1.944) | <0.001 | 0.982 (0.786–1.255) | 0.885 | 2.111 (1.653–2.695) | <0.001 | 1.147 (0.861–1.529) | 0.349 |
| Group 1 vs. group 3 | 1.353 (1.260–1.453) | <0.001 | 1.069 (0.958–1.194) | 0.233 | 1.382 (1.266–1.508) | <0.001 | 1.098 (0.957–1.259) | 0.182 |
| Group 1 vs. group 4 | 1.388 (1.282–1.503) | <0.001 | 1.172 (1.037–1.324) | 0.011 | 1.477 (1.319–1.587) | <0.001 | 1.146 (0.987–1.330) | 0.075 |
| Group 2 | 1.00 | 1.00 | 1.00 | 1.00 | ||||
| Group 2 vs. group 3 | 1.171 (0.916–1.497) | 0.208 | 1.143 (0.826–1.583) | 0.419 | 0.905 (0.684–1.196) | 0.482 | 1.011 (0.688–1.485) | 0.956 |
| Group 2 vs. group 4 | 1.308 (1.120–1.529) | 0.001 | 1.292 (1.021–1.635) | 0.033 | 1.198 (1.004–1.429) | 0.045 | 1.142 (0.864–1.508) | 0.351 |
| Group 3 | 1.00 | 1.00 | 1.00 | 1.00 | ||||
| Group 3 vs. group 4 | 1.461 (1.123–1.902) | 0.005 | 1.229 (0.963–1.752) | 0.087 | 1.586 (1.164–2.161) | 0.003 | 1.354 (0.936–1.958) | 0.108 |
a) Mild hearing loss or moderate to severe hearing loss and the multivariate analysis was adjusted for age, sex, high-density lipoprotein cholesterol levels, triglyceride levels, estimated glomerular filtration rate, systolic blood pressure, diastolic blood pressure, body mass index, alcohol consumption, smoking behavior, glycated hemoglobin levels, and exposure to explosives or occupational noise.




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download