Introduction
Intraoperative hypothermia commonly develops due to redistribution of core body heat to the skin surface through anesthesia-associated vasodilation and depression of hypothalamic thermoregulatory centers, and heat loss after exposure to cold environment [
1]. This can be aggravated by massive volume resuscitation with room-temperature crystalloid fluid and refrigerated blood transfusions [
2]. Many laboratory and clinical studies at moderate and high flow rates (above 1,000 ml/h) have shown that fluid warmers can effectively heat intravenous fluids, and prevent hypothermia [
3456].
However, intraoperative hypothermia with risks of cardiovascular, hemorrhagic, and infectious complications can develop when cold fluid is infused at low flow rates for intraoperative basal fluid replacement [
78]. Infusion of cold intravenous fluids is known to be one of the potential risk factors for the development of intraoperative hypothermia [
9]. Therefore, it has been recommended that intravenous fluid infusion be performed after warming the fluids to 37℃ using a fluid warmer, if the fluid requirement exceeds 500 ml; this can prevent and treat inadvertent perioperative hypothermia in adults [
10]. Studies at low and moderate infusion rates showed that fluid warming systems had different heating capabilities according to the degree of flow rates [
611]. Nevertheless, at low and moderate flow rates, most anesthesiologists do not use any fluid warmer or use them without considering their heating capabilities.
Ranger™, ThermoSens®, and Mega Acer Kit® are commonly used in our hospital. Ranger™ warms the intravenous fluids with dry heat technology, using a flat plastic sheet in contact with a counter-current metal heating plate. ThermoSens® also warms fluids with dry heat technology, using plastic cassettes in contact with a heating plate [
51213]. Mega Acer Kit® has been recently developed as a device with a convective warming system, which can warm the fluid directly by using heated convective air currents [
37]. Mega Acer Kit® consists of a fluid line (length: 100 cm, volume: 5 ml) that is placed along a heating wire wrapped in cotton within a humidified and heated circuit [
37].
Therefore, we hypothesized that Mega Acer Kit®, Ranger™, and ThermoSens® have different heating capabilities at low flow rates as their mechanisms of heating fluids are different. In the present study, we tested whether Mega Acer Kit®, Ranger™, and ThermoSens® can be effectively warm infusion fluids to 37℃, at a flow rate of 440 ml/h to compensate for the fluid deficit and maintenance fluid for a 70 kg patient. We measured the fluid temperatures at the inlet and outlet (76 cm distance) points of each warmer after 60 min of infusion, and calculated the expected decrease in mean body temperature after 5 h of infusion for a 70 kg patient (ΔMBT5).
Go to :

Materials and Methods
The Institutional Review Board waived ethical approval for this laboratory investigation. The investigation room temperature was automatically controlled, and maintained at 22 ± 2℃. The intravenous fluids were kept in this room for at least 24 h before testing to ensure calibration with the ambient temperature.
All devices were set-up with a warming temperature of 41℃ according to the manufacturers' instructions, and they were preheated for 10 min to calibrate each device's condition. The infusion set was primed with normal saline hung at a height of 1 m from the warming device and attached to a roller pump (TE-171, Terumo Corp., Tokyo, Japan). The following three warming devices were tested: (1) Mega Acer Kit® (Group M, Ace Medical, Seoul, Korea), (2) Ranger™ (Group R, Arizant Healthcare, Inc., MN, USA), and (3) ThermoSens® (Group T, Sewoon Medical Company, Seoul, Korea). Equal distances from each device to the outlet point (P
out, 76 cm) were achieved using a 55-cm fluid extension line, three-way connectors, and 18-gauge intravenous catheters connected to the outlet of the fluid warmers in series (
Fig. 1). These extended lines were exposed to ambient room temperature. Two PT 100 temperature probes (KRGA-50, Kimo Instruments, Edenbridge, UK) were connected to a Kistock Datalogger (KTH350, Kimo Instruments, Edenbridge, UK). The probes were inserted at the inlet point (P
in) and at P
out. An artificial lung was ventilated with 500 ml of tidal volume, at 10 breaths/min, and 6 L/min of oxygen and medical air using Mega Acer Kit® without humidification in all groups. Normal saline was then delivered at flow rates of 440 ml/h, which was sum of the one third (300 ml/h) of maintenance fluid, simply calculated with “4-2-1” formula for a 70 kg patient with 8 h NPO, and the required deficit replacement [140 ml/h (2 ml/kg/h)] due to losses by losses third spacing and evaporation during surgery with anticipated minimal to moderate tissue trauma [
1415].
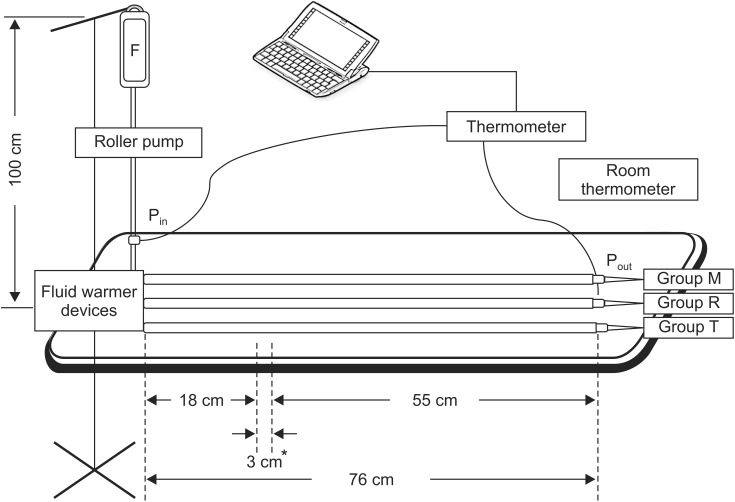 | Fig. 1Illustration of laboratory settings. Intravenous fluid was warmed with Mega Acer Kit® (Group M), Ranger™ (Group R), and ThermoSens®(Group T). F: normal saline. Pin and Pout are the inlet point and the outlet point (76 cm from device), respectively. *Length after applying three-way connectors to obtain the same distance (76 cm) from each device.
|
The fluid temperatures at two recoding points (TPin and TPout) were recorded automatically using the Kistock Datalogger at 5-min intervals for 60 min. After downloading each result at the aforementioned time points to our computer using a software (Kilog; Kimo Instruments, Edenbridge, UK), we calculated the mean values of TPin and TPout recorded at 5-min intervals for 60 min after infusion in each group for the expected decrease in mean body temperature (ΔMBT). Each test was repeated ten times.
ΔMBT was calculated for a 70 kg patient for 5 hours, in intervals of 1 h. For calculating ΔMBT when the patients received cold intravenous fluid, we recollected all the values of mean TP
in in groups M, R, and T for each test, and we defined it as a group UWF. For calculating ΔMBT, the mean TP
out of each group (group M, group R, and group T) and the mean fluid temperature of group UWF were used. This was done using Horowitz's formula, as given below [
16]:
where ΔMBT is the expected decrease in mean body temperature; TF, the temperature of the infused fluid; TPt, the patient's baseline core temperature (37℃); SF, the specific heat of the infused fluid (1.0 1 kcal/L/℃ for saline); Vol, the volume of the infused fluid (L); SPt, the specific heat of human tissue (0.83 kcal/L/℃); and Wt, the weight of the patient (kg).
The primary endpoint was the fluid temperature at 76 cm from the device, 60 min after infusion. The secondary endpoint was the expected decrease in mean body temperature for a 70 kg patient, 5 h after intravenous infusion of the warmed fluid (ΔMBT5).
Statistical analysis
Statistical analysis was performed using SPSS (Windows ver. 21.0, IBM Corp., Armonk, NY, USA). All measured values were presented as mean (95% CI). The temperatures at 76 cm from device, 60 min after infusion, and ΔMBT were analyzed using one-way analysis of variance (ANOVA). If the test revealed significant differences among the fluid warmers, Scheffé's test was used for post-hoc multiple comparisons to estimate the pairwise differences among the groups. Statistical significance was defined as P values < 0.05.
Go to :

Results
The TP
out 60 min after infusion was highest in group M (36.01 [35.73–36.29]℃), followed by groups T (29.81 [29.38–30.24]℃, P < 0.001) and R (29.12 [28.52–29.72]℃, P < 0.001), respectively (
Table 1,
Fig. 2). The TP
out at 5-min intervals for 60 min was significantly higher in group M than in groups R and T at all time points (
Fig. 2, P < 0.001). However, it was not significantly higher in group T than group R except at some time points (
Fig. 2).
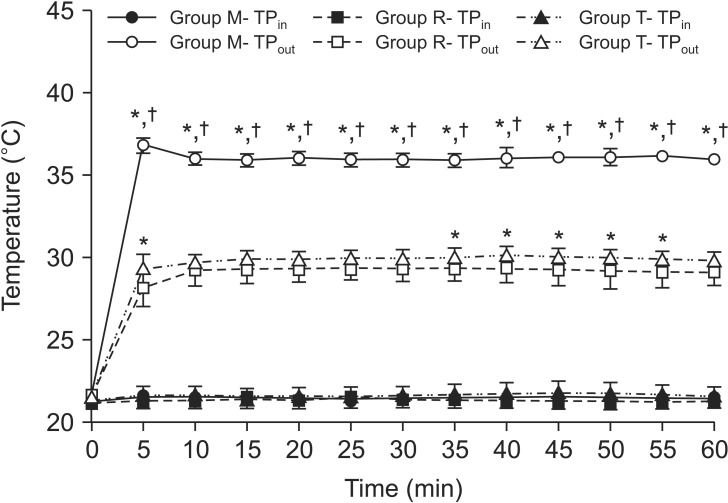 | Fig. 2Fluid temperatures in chronological order. Intravenous fluid was warmed with Mega Acer Kit® (Group M), Ranger™ (Group R), and ThermoSens® (Group T). TPout was significantly higher in group M than in groups R and T, at all time points. None of the investigated fluid warmers provided a constant normothermic temperature (above 36.5℃). TPin and TPout are the fluid temperatures at the inlet point and the outlet point (76 cm from device), respectively. Values are expressed as mean (95% CI). *,†P < 0.05 compared with groups R and T, respectively.
|
Table 1
Fluid Temperature (℃) and Expected Decrease in Mean Body Temperature (ΔMBT, ℃/h)
|
Group M (n = 10) |
Group R (n = 10) |
Group T (n = 10) |
Group UWF (n = 30) |
P value §
|
|
Fluid temperature 60 min after infusion |
|
|
|
|
|
|
At Pin
|
21.46
(21.37, 21.54) |
21.41
(21.05, 21.76) |
21.60
(21.22, 21.97) |
|
0.586 |
|
At Pout
|
36.01*,†
(35.73, 36.29) |
29.12
(28.52, 29.72) |
29.81
(29.38, 30.24) |
|
< 0.001 |
|
Mean fluid temperature during 60 min |
|
|
|
|
|
|
At Pin
|
21.46
(21.39, 21.54) |
21.41
(21.05, 21.75) |
21.64
(21.23, 22.05) |
|
0.468 |
|
At Pout
|
36.07 *,†
(35.84, 36.30) |
29.17
(28.56, 29.78) |
29.93*
(29.59, 30.27) |
|
< 0.001 |
|
ΔMBT after 5 h infusion |
−0.04 *,†,‡
(−0.04, −0.03) |
−0.30
(−0.32, −0.27) |
−0.27*
(−0.28, −0.29) |
−0.59*,†
(−0.59, −0.58) |
< 0.001 |

The mean TP
out was significantly highest in group M (36.07 [35.84–36.30]℃), compared to groups T (29.93 [29.59–30.27]℃, P < 0.001) and R (29.17 [28.56–29.78]℃, P < 0.001) (
Table 1). It was significantly higher in group T than in group R (
Table 1, P = 0.028). The mean TP
in showed no significant differences among the three groups. None of the investigated fluid warmers provided a constant normothermic temperature (above 36.5℃) (
Fig. 2).
The ΔMBT at 5 h after infusion was significantly smallest in group M (−0.04 [−0.04 to −0.03]℃), compared to groups T (−0.27 [−0.28 to −0.29]℃, P < 0.001) and R (−0.30 [−0.32 to −0.27]℃, P < 0.001) (
Table 1,
Fig. 3). It was also significant higher in group T than in group R (
Table 1, P = 0.019). However, if an unwarmed fluid without warmers was infused with TP
in in all groups (group UWF), the ΔMBT at 5 h after infusion was significantly higher [−0.59 (−0.59 to −0.58)℃] (
Table 1,
Fig. 3, P < 0.001). ΔMBT at 1 h intervals for 5 h after fluid infusion using each warmer was significantly lower in group M than in groups T, R, and UWF for a 70 kg patient (
Fig. 3, P < 0.001).
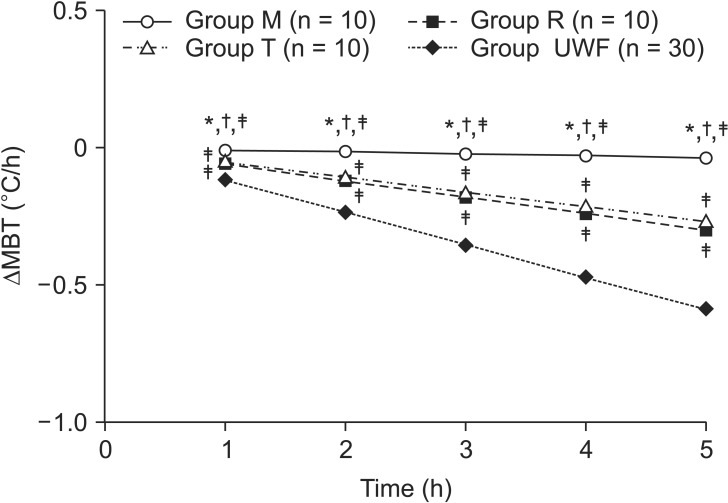 | Fig. 3The expected decrease in mean body temperature (ΔMBT) during 5 hours with 1 h intervals. Intravenous fluid was warmed with Mega Acer Kit® (Group M), Ranger™ (Group R), and ThermoSens® (Group T). Group UWF: group was tested with TPin in group M, R, and T for calculating ΔMBT. ΔMBT was significantly lower in group M than in groups T, R, and UWF, followed by groups T, R, and UWF at all time points. ΔMBT above 0.5℃ in group UWF was expected after 5 hours infusion with a significantly higher decrease compared to groups M, R, and T. For calculating ΔMBT, the mean TPout of each group was used. Values are expressed as mean (95% CI). *,†,‡P < 0.05 compared with groups R, T, and UWF, respectively.
|
Go to :

Discussion
The results this study show that Mega Acer Kit® warms fluids more effectively than Ranger™ and ThermoSens® at a flow rate of 440 ml/h. However, none of the fluid warmers investigated in our study achieved a constant normothermic temperature (above 36.5℃), even though Mega Acer Kit® could warm the fluid above 35.5℃, with a lower 95% CI.
The results of studies using Mega Acer Kit® to record the fluid temperatures at the distal recorded point are discrepant [
37]. Kim et al. [
3] experimentally showed that Mega Acer Kit® warmed the fluid (33.6 ± 1.4℃) at its outlet site (18 cm) at flow rates of 400 ml/h with a device temperature of 38℃ under humidification. They also found that, in a clinical situation, the warming fluid temperature was 31.0 ± 1.0℃ at a distance of 118 cm from the device at a mean flow rate of 442 ml/h. However, our present study revealed that Mega Acer Kit® warmed the fluid to 36.07 (35.84–36.30)℃ at 440 ml/h. In addition, Jung et al. [
7] have documented higher warmed fluid temperature (37.4 ± 1.7℃) compared to our current study at a similar flow rate.
This discrepancy in delivered fluid temperatures may be explained by several reasons. First, there may be an influence of the humidity and temperature of the inspired gas on the performance of the warming fluid during ventilation [
3]. The humidity of inspired gas was the highest 15 min post-induction and it decreased gradually, while the temperature of the inspired gas was stable even 75 min post-induction. The temperature of the delivered fluid seemed to have a negative correlation with the humidity of the inspired gas and a positive correlation with the temperature of the inspired gas, even though the results of the correlation analysis were not mentioned. In contrast, we did not operate the humidification system as we could not confirm this interaction before starting the study. Second, we may attribute this discrepancy to the use of unwarmed fluid at a higher temperature and a cover application on the extended line to eliminate the indirect effect of room temperature. Jung et al. [
7] applied a cover on the extension line and used the unwarmed fluid at a higher temperature (~2℃) than that of this study. They recorded a higher fluid temperature (37.4 ± 1.7℃) than that (36.07 [35.84–36.30]℃) of our study, even though we applied a shorter extended line and a higher device temperature (41℃) without humidification.
The effectiveness of ThermoSens® and Ranger™ have been studied at different flow rates [
571316]. However, there were no studies investigating whether ThermoSens® and Ranger™ can effectively deliver warm fluid at low and moderate flow rates (below 1000 ml/h) in clinical and laboratory conditions, except for Jung's report using Ranger™ [
7]. ThermoSens® warms the fluid using a heating tube containing a heating plate, in direct contact with the fluid [
13]. FloTem and JOYOTHER BM-1 (Joyother, Seoul, Korea) also employ the dry heat technology for warming the fluid with a tube heated by electric heater elements, similar to technology of ThermoSens® [
1117]. WarmFlo® heats the fluid using a dry cassette heat exchange technology, similar to Ranger™, which employs dry heat technology using a counter-current metal heating plate [
18].
Therefore, we expect that the effects of ThermoSens® and Ranger™ are similar to those of FloTem, JOYOTHER BM-1, and WarmFlo®, which operate using similar warming methods. FloTem, with a disposable length of 108 cm, requires a flow rate of at least 300 ml/h to deliver fluid at temperatures above 32℃; however, it could not deliver fluid above 35℃, even when the flow rate was increased to 1,000 ml/h [
6]. JOYOTHER BM-1 was not effective in delivering the warmed fluid at a distance greater than 75 cm from the outlet of the device at low and moderate flow rates [
17]. Patel et al. [
11] also showed that FloTem delivered warmed fluid with a temperature of 29.5℃ at 390 ml/h and 30.8℃ at 780 ml/h, similar to the result of this study (29.93 [29.59–30.27]℃) using ThermoSens®. Ranger™ could also not achieve temperatures greater than 35℃ (32.8 ± 0.6℃), at 108-cm distance from the device, at a flow rate of 400 ml/h [
7]. Warmblood® could not deliver warmed fluid at temperatures greater than 35℃ at a flow rate of below 600 ml/h, at the distal site with a device temperature of 42℃ [
18], even though it was possible to warm the fluid at flow rates of 600 ml/h or more. This result also showed that fluid temperatures greater than 35℃ (29.17 [28.56–29.78]℃) cannot be obtained with Ranger™ at a 78-cm distance and a flow rate of 440 ml/h [
7].
The anticipated decrease in intraoperative body temperature when using a fluid warmer can be predicted by ΔMBT, as calculated using Horowitz's equation [
16]. The work suggested that a warming device should be used only if ΔMBT is expected to decrease by more than 0.5℃, because this degree of hypothermia can be clinically tolerated or reversed using a forced-air heater alone. Generally, most anesthesiologists do not use a fluid warmer at lower flow rates because of the minimal effect on the MBT. However, our results showed that the ΔMBT was −0.59℃ if an unwarmed fluid was infused at a flow rate of 440 ml/h for 5 h, and fluid warmers can be helpful to decrease the ΔMBT as much as possible. Mega Acer Kit® could especially decrease ΔMBT to −0.04℃, which is significantly less than that of ThermoSens® and Ranger™. Even though the anticipated decrease in MBT was below 0.32℃ with the 3 h infusion of normal saline at 23℃ without fluid warmer, the intraoperative core temperature could decrease to below 35℃ after 3 h at a flow rate of 400 ml/h [
7]. Actually, intraoperative hypothermia below 35℃ commonly develops due to the impairment of central thermoregulation and heat loss through the exposed body surface during anesthesia and surgery [
1]. This means that a fluid warmer should be used for preventing and treating intraoperative hypothermia even if the anticipated decrease in MBT is below 0.5℃ and the infusion flow rate is low.
There are some limitations associated with the present study. First, we did not show the clinical results on the degree of warmth produced by warming fluids at 440 ml/h using each device. Second, this study was performed at a single, fixed infusion rate. Therefore, further studies will be required to determine whether intravenous fluid warmers such as Mega Acer Kit®, Ranger™, and ThermoSens® are effective in warming fluids to 37℃ at several different low and moderate flow rates.
In conclusion, in this study, Mega Acer Kit®, ThermoSens®, and Ranger™ showed different fluid heating capabilities at low flow rates, and Mega Acer Kit® was demonstrated to warm the fluid more effectively compared to ThermoSens® and Ranger™. The lowest anticipated ΔMBT was obtained at a flow rate of 440 ml/h, although all fluid warming devices cannot deliver fluids with temperatures greater than 36.5℃ at such infusion rates.
Go to :








 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download