1. Henzi I, Walder B, Tramèr MR. Dexamethasone for the prevention of postoperative nausea and vomiting: a quantitative systematic review. Anesth Analg. 2000; 90:186–94.

2. Lee SH, Lee YC, Lee JH, Choi SR, Lee SC, Lee JH, et al. The prophylactic effect of dexamethasone on postoperative sore throat in prone position surgery. Korean J Anesthesiol. 2016; 69:255–61.

3. Dieleman JM, Nierich AP, Rosseel PM, van der Maaten JM, Hofland J, Diephuis JC, et al. Intraoperative high-dose dexamethasone for cardiac surgery: a randomized controlled trial. JAMA. 2012; 308:1761–7.
4. Coursin DB, Wood KE. Corticosteroid supplementation for adrenal insufficiency. JAMA. 2002; 287:236–40.

5. Dalkara T, Onur R. Facilitatory effects of dexamethasone on neuromuscular transmission. Exp Neurol. 1987; 95:116–25.

6. Oliveira L, Costa AC, Noronha-Matos JB, Silva I, Cavalcante WL, Timóteo MA, et al. Amplification of neuromuscular transmission by methylprednisolone involves activation of presynaptic facilitatory adenosine A2A receptors and redistribution of synaptic vesicles. Neuropharmacology. 2015; 89:64–76.

7. Dal Belo CA, Leite GB, Fontana MD, Corrado AP, Zanandréa Baso AC, Moreno Serra CS, et al. New evidence for a presynaptic action of prednisolone at neuromuscular junctions. Muscle Nerve. 2002; 26:37–43.

8. Maes K, Testelmans D, Thomas D, Decramer M, Gayan-Ramirez G. High dose methylprednisolone counteracts the negative effects of rocuronium on diaphragm function. Intensive Care Med. 2011; 37:1865–72.

9. Robinson BJ, Lee E, Rees D, Purdie GL, Galletly DC. Betamethasone-induced resistance to neuromuscular blockade: a comparison of atracurium and vecuronium in vitro. Anesth Analg. 1992; 74:762–5.
10. Soltész S, Fraisl P, Noé KG, Hinkelbein J, Mellinghoff H, Mencke T. Dexamethasone decreases the duration of rocuronium-induced neuromuscular block: a randomised controlled study. Eur J Anaesthesiol. 2014; 31:417–22.
11. Bom A, Bradley M, Cameron K, Clark JK, Van Egmond J, Feilden H, et al. A novel concept of reversing neuromuscular block: chemical encapsulation of rocuronium bromide by a cyclodextrin-based synthetic host. Angew Chem Int Ed Engl. 2002; 41:266–70.

12. Itoh H, Shibata K, Matsumoto T, Nitta S, Nishi M, Kobayashi T, et al. Effects of neuromuscular-blocking drugs in rats in vivo: direct measurements in the diaphragm and tibialis anterior muscle. Acta Anaesthesiol Scand. 2004; 48:903–8.

13. Rezonja K, Lorenzon P, Mars T. Opposing effects of dexamethasone, agrin and sugammadex on functional innervation and constitutive secretion of IL-6 in in vitro innervated primary human muscle cells. Neurosci Lett. 2013; 549:186–90.

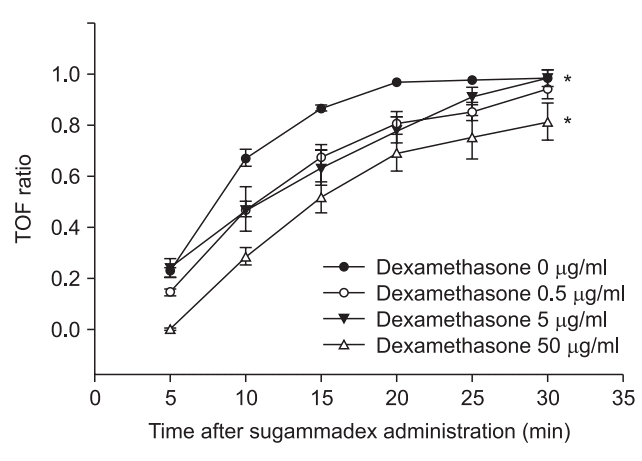
14. Rezonja K, Sostaric M, Vidmar G, Mars T. Dexamethasone produces dose-dependent inhibition of sugammadex reversal in in vitro innervated primary human muscle cells. Anesth Analg. 2014; 118:755–63.

15. Buonanno P, Laiola A, Palumbo C, Spinelli G, Servillo G, Di Minno RM, et al. Dexamethasone does not inhibit sugammadex reversal after rocuronium-induced neuromuscular block. Anesth Analg. 2016; 122:1826–30.

16. Gulec E, Biricik E, Turktan M, Hatipoglu Z, Unlugenc H. The effect of intravenous dexamethasone on sugammadex reversaltime in children undergoing adenotonsillectomy. Anesth Analg. 2016; 122:1147–52.
17. Rezonja K, Mars T, Jerin A, Kozelj G, Pozar-Lukanovic N, Sostaric M. Dexamethasone does not diminish sugammadex reversal of neuromuscular block - clinical study in surgical patients undergoing general anesthesia. BMC Anesthesiol. 2016; 16:101.

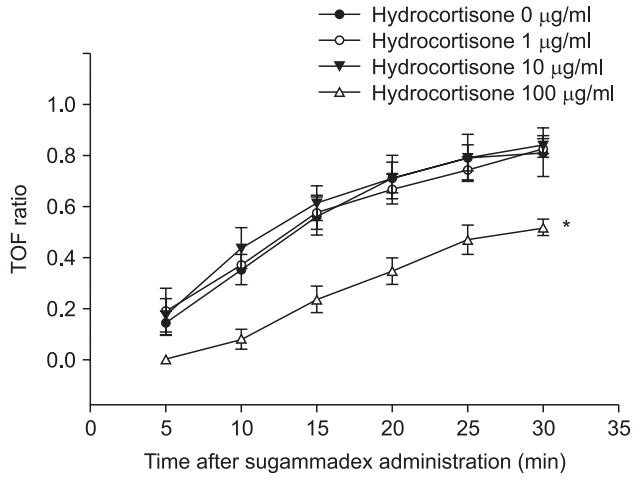
18. Durant NN, Briscoe JR, Katz RL. The effects of acute and chronic hydrocortisone treatment on neuromuscular blockade in the anesthetized cat. Anesthesiology. 1984; 61:144–50.
19. Kilkenny C, Browne W, Cuthill IC, Emerson M, Altman DG. Animal research: reporting in vivo experiments: the ARRIVE guidelines. Br J Pharmacol. 2010; 160:1577–9.

20. Zwiers A, van den Heuvel M, Smeets J, Rutherford S. Assessment of the potential for displacement interactions with sugammadex: a pharmacokinetic-pharmacodynamic modelling approach. Clin Drug Investig. 2011; 31:101–11.
21. Tsuei SE, Moore RG, Ashley JJ, McBride WG. Disposition of synethetic glucocorticoids. I. Pharmacokinetics of dexamethasone in healthy adults. J Pharmacokinet Biopharm. 1979; 7:249–64.
22. Morariu AM, Loef BG, Aarts LP, Rietman GW, Rakhorst G, van Oeveren W, et al. Dexamethasone: benefit and prejudice for patients undergoing on-pump coronary artery bypass grafting: a study on myocardial, pulmonary, renal, intestinal, and hepatic injury. Chest. 2005; 128:2677–87.
23. Yared JP, Bakri MH, Erzurum SC, Moravec CS, Laskowski DM, Van Wagoner DR, et al. Effect of dexamethasone on atrial fibrillation after cardiac surgery: prospective, randomized, double-blind, placebo-controlled trial. J Cardiothorac Vasc Anesth. 2007; 21:68–75.

24. Brady ME, Sartiano GP, Rosenblum SL, Zaglama NE, Bauguess CT. The pharmacokinetics of single high doses of dexamethasone in cancer patients. Eur J Clin Pharmacol. 1987; 32:593–6.

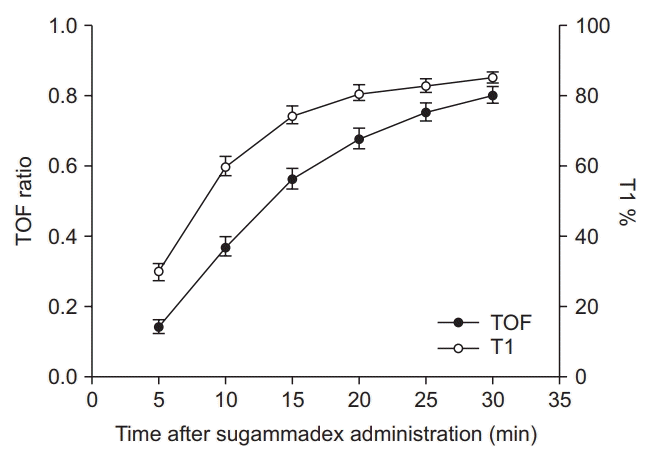
25. Kopman AF, Klewicka MM, Neuman GG. The relationship between acceleromyographic train-of-four fade and single twitch depression. Anesthesiology. 2002; 96:583–7.

26. de Kam PJ, Hou J, Wang Z, Lin WH, van den Heuvel M. Pharmacokinetics of sugammadex 16 mg/kg in healthy Chinese volunteers. Int J Clin Pharmacol Ther. 2015; 53:456–61.
27. Lamberts SW, Bruining HA, de Jong FH. Corticosteroid therapy in severe illness. N Engl J Med. 1997; 337:1285–92.

28. Hamitouche N, Comets E, Ribot M, Alvarez JC, Bellissant E, Laviolle B. Population pharmacokinetic-pharmacodynamic model of oralfludrocortisone and intravenous hydrocortisone in healthy volunteers. AAPS J. 2017; 19:727–35.
29. Melin J, Parra-Guillen ZP, Hartung N, Huisinga W, Ross RJ, Whitaker MJ, et al. Predicting cortisol exposure from paediatric hydrocortisoneformulation using a semi-mechanistic pharmacokinetic modelestablished in healthy adults. Clin Pharmacokinet. 2018; 57:515–27.
30. Derendorf H, Möllmann H, Barth J, Möllmann C, Tunn S, Krieg M. Pharmacokinetics and oral bioavailability of hydrocortisone. J Clin Pharmacol. 1991; 31:473–6.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download