Abstract
Anesthetic management of pediatric liver transplantation in a patient with osteogenesis imperfecta (OI) requires tough decisions and comprehensive considerations of the cascade of effects that may arise and the required monitoring. Total intravenous anesthesia (TIVA) with propofol and remifentanil was chosen as the main anesthetic strategy. Malignant hyperthermia (MH), skeletal fragility, anhepatic phase during liver transplantation, uncertainties of TIVA in children, and propofol infusion syndrome were considered and monitored. There were no adverse events during the operation. Despite meticulous precautions with regard to the risk of MH, there was an episode of high fever (40℃) in the ICU a few hours after the operation, which was initially feared as MH. Fortunately, MH was ruled out as the fever subsided soon after hydration and antipyretics were given. Although the delivery of supportive care and the administration of dantrolene are the core principles in the management of MH, perioperative fever does not always mean a MH in patients at risk for MH, and other common causes of fever should also be considered.
Osteogenesis imperfecta (OI) is an autosomal dominant genetic disorder that manifests with skeletal fragility. Patients with this disorder are susceptible to malignant hyperthermia (MH) during anesthesia and therefore, close monitoring of signs of MH such as acidosis, tachycardia, tachypnea, hyperthermia, electrolyte imbalance, and rhabdomyolysis is required [1].
For children with end-stage liver disease, liver transplantation is accepted as the treatment of choice. Five-year graft and patient survival rates after pediatric liver transplantations are reported to be around 70% [2].
We report a case of an 8-month-old OI patient who underwent liver transplantation due to progressive familial intrahepatic cholestasis. The combination of OI and liver transplantation required special attention towards skeletal fragility and MH during the operation. Additional attention was paid to the possibility of the patient developing propofol infusion syndrome, taking into consideration that high doses of propofol may be required when the duration of surgery is long [3].
An 8-month-old male infant weighing 7.9 kg was scheduled for living donor liver transplantation due to progressive familial intrahepatic cholestasis. He was previously diagnosed with type 3 or 4 OI (paternal COL1A2 gene). The patient had suffered from fractures of the femur and humerus and had undergone four cycles of pamidronate treatment. Preoperative echocardiography showed a small patent foramen ovale with a left to right shunt. Apart from the elevated liver function tests (alanine transaminase: 165 IU/L, aspartate transaminase: 211 IU/L and total Bilirubin: 19.3 mg/dl) due to cholestasis, other preoperative lab results were within the normal range.
To minimize the risk of MH, after installation of a pediatric circuit and new CO2 absorbent, ventilator washing (Ohmeda S/5 Avance, Madison, WI, USA, flow 10 L/min, TV 600 ml/fx, 10 /min, for 30 minutes) was thoroughly performed and total intravenous anesthesia using propofol and remifentanil was planned and prepared [4]. Dantrolene was readily available for use in case of a malignant hyperthermia crisis. The patient was monitored using electrocardiogram, pulse oximetry, noninvasive blood pressure measurement, and BIS. Anesthesia was induced with 0.15 mg of atropine, 20 mg of propofol, and 5 mg of rocuronium. After intubation, the right radial artery and the right femoral artery were cannulated. A 5-Fr double lumen central venous catheter was placed in the right internal jugular vein under ultrasound guidance. When performing endotracheal intubation and central venous catheter insertion, we avoided excessive neck extension and rotation. A temperature probe was placed in the oropharynx. Anesthesia was maintained with total intravenous anesthesia using 2% propofol and remifentanil. During the operation, the average infusion rates of propofol and remifentanil were 100 µg/kg/min and 0.1 µg/kg/min respectively. Body temperature and end-tidal CO2 were carefully monitored and were within the normal range throughout the operation. Arterial blood gas analyses were performed periodically to check for pH, PaO2, and PaCO2.
The patient received the lateral section of the left lobe of the liver from his mother. Donor liver graft was preserved with the histidine-tryptophan-ketoglutarate (HTK) solution (Custodiol®, Zenith pharm Inc., Germany). Just before anastomosis between the hepatic vein of the graft and reconstructed common orifice of the right, middle, and left hepatic vein, and the inferior vena cava of the recipient, the graft was perfused with 5% albumin through the portal vein. After portal vein anastomosis, the graft was reperfused by releasing the hepatic vein and portal vein clamps.
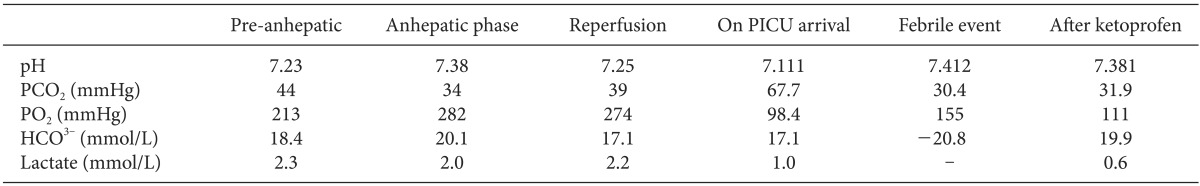
During the reperfusion period, 50 mg of calcium chloride and 3 µg of epinephrine were administered to treat the hypotension caused by the postreperfusion syndrome. End to end anastomosis of the hepatic artery and biliary reconstruction were done successfully. Apart from the reperfusion period, the patient's vital signs were stable throughout the operation and the BIS score was maintained between 45 and 65. Intraoperative laboratory data did not show features suggestive of propofol infusion syndrome (Table 1). Anesthesia time was 8 hours and 20 minutes. The anesthetic team worked in cooperation with the surgeon throughout the whole operation, and the surgery was cautiously carried out without a retractor.
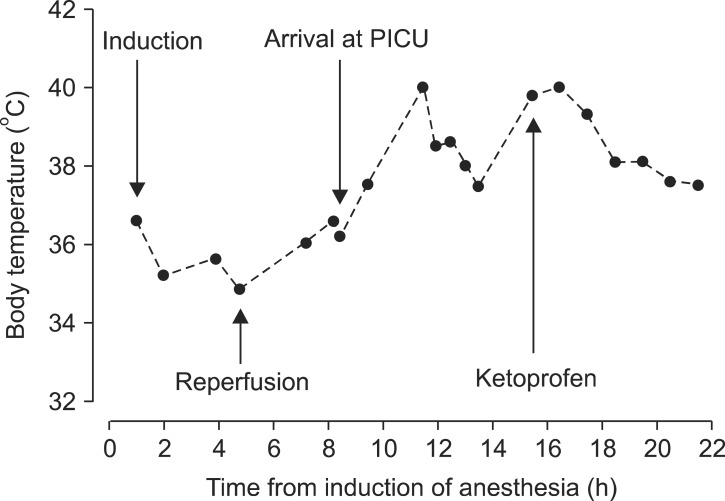
After the operation, the patient was transferred to the pediatric intensive care unit (PICU). Vital signs were stable when the patient arrived at the PICU and the axillary temperature was 36.2℃. Three hours later, the patient had a sudden spiking fever of 40℃ (Fig. 1) and an increase in the abdominal circumference was observed. For assessment of the fever, blood and sputum cultures were performed. Clinically, no focus of infection was detected. Liver ultrasound was performed to rule out bowel congestion that can be associated with portal vein obstruction. However, the hepatic artery and portal vein flows were normal. Electrolytes and arterial blood gas analysis were checked to investigate the possibility or MH but were normal (Table 1). The fever subsided after hydration and injection of 8 mg of ketoprofen (Fig. 1). The patient was discharged on postoperative day 15 without any adverse events.
Liver transplantation in this 8 month-old was performed using TIVA because of the underlying disease of OI.
OI is a group of inherited diseases responsible for varying degrees of skeletal fragility. The reported incidence range is 5-10 cases per 100,000 births and the disorder is commonly classified into 5 types [5]. Clinical manifestations of OI are brittle teeth, scoliosis, and bone fragility with fractures occurring with minimal trauma. Patients suffering from OI often undergo orthopedic surgery due to their skeletal problems. MH is a serious problem that can occur during anesthesia in these patients. However, MH can be prevented if TIVA is used instead of inhaled anesthetics and triggering factors such as succinylcholine are avoided.
TIVA is still controversial in pediatric anesthesia and the main concern is intraoperative awareness [6]. Methods to assess the depth of anesthesia in pediatric patients are not reliable. It is not clear whether the adult BIS algorithm and equipment can be applied to pediatric patients. The EEG of newborns is different from that of adults because brain maturation and synapse formation continues until the age of 5 [7,8,9]. However, some have suggested the possibility of using BIS in pediatric patients by showing the correlation of BIS with end-tidal sevoflurane concentration in infants and children [10]. Therefore, although not optimal, we monitored the BIS and maintained the BIS score between 45 and 65.
What complicates TIVA in pediatric liver transplantation even further are the pharmacokinetic changes that occur during the anhepatic phase. Hepatic metabolism of propofol contributes about 60% of the total propofol clearance [11]. In adult liver transplantation, apparent systemic clearance of propofol was decreased by up to 42% during the anhepatic phase compared with the liver dissection phase [12]. In our case, the infusion rate of propofol was carefully titrated with the aid of the BIS monitor so that it was not low enough to cause intraoperative awareness, but not high enough to trigger the side effects of propofol, especially the propofol infusion syndrome. Intraoperative laboratory data may not be able to discriminate between the metabolic acidosis produced in the anhepatic period during liver transplantation and that caused by the propofol infusion syndrome. However, the degree of metabolic acidosis is expected to be much more severe when the propofol infusion syndrome coincides with the lactic acidosis that usually develops during liver transplantation. The average infusion rates of propofol and remifentanil were 100-150 µg/kg/min and 0.1-0.15 µg/kg/min respectively throughout the operation. The total amount of infused propofol was 540 mg.
Volatile anesthetics and succinylcholine are the most commonly known triggers of MH in susceptible patients. There are reports of other MH triggering drugs in the literature and these include serotonergic drugs, phosphodiesterase type III inhibitors, statins, tetracaine, methylene blue and ondansetron [13,14,15]. We were unable to find reports of MH triggered by drugs or solutions used in liver transplantation such as HTK solution for liver graft preservation, nafamostat mesilate (Futhan®, SK chemicals, Japan) and basiliximab (Simulect®, Novartis, Switzerland); this lack of reports was yet another challenge when managing our case. Given that most triggers of MH are related to anesthesia and that liver transplantation is the only viable option, we performed TIVA to manage the OI patient receiving pediatric liver transplantation and we report this case.
Although cautious anesthesia was perforrmed with regard to MH, there was an episode of fever in the ICU. However, the physicians considered that giving conservative treatment and performing a work-up for other surgical complications were more appropriate prior to using dantrolene. We closely monitored the patient, and as the fever subsided after hydration and antipyretics, the possibility of MH was considered insignificant. It is difficult to form conclusions about what caused the febrile event. However, dehydration atelectasis and excessive temperature preservation are thought to be likely causes of the postoperative fever.
In pediatric liver transplantation of an OI patient, anesthesiologists need to consider some issues. First, when monitoring the sedation depth using BIS in pediatric patients, the difference in BIS between pediatric patients and adults owing to EEG has to be taken into account. Second, for the maintenance of TIVA during the anhepatic phase, it should be kept in mind that changes in drug metabolism can occur due to the absence of the liver. Third, close monitoring and frequent measurements of laboratory data such as arterial blood gas are required. If metabolic acidosis is more severe than usually occurs during the anhepatic phase, propofol infusion syndrome should be considered and ruled out. Fourth, the potential for drugs used during organ transplantation to trigger MH should always be considered. Lastly, when patients with a high risk of developing MH develop fever, conservative treatment such as hydration and antipyretics should be a priority.
Based on our experience, we believe that propofol based TIVA is a safe alternative anesthetic strategy in OI patients undergoing liver transplantation. We would also like to stress that when managing a febrile event in a patient with a high risk of developing MH, other causes should be ruled out and conservative supportive management such as hydration and antipyretics should not be overlooked.
References
1. Litman RS, Rosenberg H. Malignant hyperthermia: update on susceptibility testing. JAMA. 2005; 293:2918–2924. PMID: 15956637.
2. LaRosa C, Baluarte HJ, Meyers KE. Outcomes in pediatric solid-organ transplantation. Pediatr Transplant. 2011; 15:128–141. PMID: 21309962.

3. Laquay N, Pouard P, Silicani MA, Vaccaroni L, Orliaguet G. Early stages of propofol infusion syndrome in paediatric cardiac surgery: two cases in adolescent girls. Br J Anaesth. 2008; 101:880–881. PMID: 19004919.

4. Brünner HW, Pohl S, Grond S. Washout of sevoflurane from the GE Avance and Amingo Carestation anesthetic machines. Acta Anaesthesiol Scand. 2011; 55:1118–1123. PMID: 22092210.
6. Lerman J, Jöhr M. Inhalational anesthesia vs total intravenous anesthesia (TIVA) for pediatric anesthesia. Paediatr Anaesth. 2009; 19:521–534. PMID: 19453585.

7. Torres F, Anderson C. The normal EEG of the human newborn. J Clin Neurophysiol. 1985; 2:89–103. PMID: 3916842.

8. Scher MS, Sun M, Hatzilabrou GM, Greenberg NL, Cebulka G, Krieger D, et al. Computer analyses of EEG-sleep in the neonate: methodological considerations. J Clin Neurophysiol. 1990; 7:417–441. PMID: 2211996.

9. Holmes GL, Lombroso CT. Prognostic value of background patterns in the neonatal EEG. J Clin Neurophysiol. 1993; 10:323–352. PMID: 8408599.

10. Denman WT, Swanson EL, Rosow D, Ezbicki K, Connors PD, Rosow CE. Pediatric evaluation of the bispectral index (BIS) monitor and correlation of BIS with end-tidal sevoflurane concentration in infants and children. Anesth Analg. 2000; 90:872–877. PMID: 10735791.

11. Veroli P, Okelly B, Bertrand F, Trouvin JH, Farinotti R, Ecoffey C. Extrahepatic metabolism of propofol in man during the anhepatic phase of orthotopic liver-transplantation. Br J Anaesth. 1992; 68:183–186. PMID: 1540462.
12. Takizawa D, Sato E, Hiraoka H, Tomioka A, Yamamoto K, Horiuchi R, et al. Changes in apparent systemic clearance of propofol during transplantation of living related donor liver. Br J Anaesth. 2005; 95:643–647. PMID: 16169890.

13. Fiege M, Wappler F, Scholz J, Weisshorn R, von Richthofen V, Schulte am Esch J. Effects of the phosphodiesterase-III inhibitor enoximone on skeletal muscle specimens from malignant hyperthermia susceptible patients. J Clin Anesth. 2000; 12:123–128. PMID: 10818326.

14. Wappler F, Fiege M, Schulte am. Pathophysiological role of the serotonin system in malignant hyperthermia. Br J Anaesth. 2001; 87:794–798. PMID: 11878537.

15. Hopkins PM. Malignant hyperthermia: pharmacology of triggering. Br J Anaesth. 2011; 107:48–56. PMID: 21624965.





 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download