Abstract
Background
Ulinastatin is a glycoprotein derived from human urine and a serine protease inhibitor found in human urine and blood. Ulinastatin increases both liver blood flow and urine output. Rocuronium is eliminated mainly through the liver and partly through the kidney, hepatic elimination of rocuronium might be enhanced by ulinastatin. We examined the effect of ulinastatin on the neuromuscular block caused by rocuronium.
Methods
Forty four adult patients were randomly divided into two groups of 22 patients each, i.e. the study group and the control group. In the study group, a bolus dose of ulinastatin 5,000 U/kg was administered 2 min before the injection of rocuronium 0.6 mg/kg. In the control group, normal saline was administered instead of ulinastatin. For the monitoring of both onset and recovery from neuromuscular blockade, train-of-four (TOF) and post-tetanic count were used with TOF-Watch Sx. All patients underwent general anesthesia with total intravenous anesthesia (TIVA) of remifentanil and propofol, using the effect site target infusion system.
Results
In the study group, the onset of neuromuscular block was significantly slower than in the control group (P < 0.05). The recovery time from the rocuronium injection to the return of PTC was also significantly shorter in the study group than in the control group (P < 0.05). Similarly, times to the return of T1, T2, T3, and T4 (i.e. the first, second, third, and fourth response of TOF) were significantly shorter in the study group than in the control group (P < 0.05).
Rocuronium, the 2-morpholino, 3-desacetyl, 16-N-allylpyrrolidino derivative of vecuronium, is an aminosteroid, non-depolarizing neuromuscular blocking agent [1]. It is widely used during anesthesia for its short onset time and intermediate duration of action, similar to that of vecuronium [1-3]. The pharmacokinetics of rocuronium also resembles those of vecuronium. Thus, rocuronium has relatively rapid onset of neuromuscular block and an intermediate duration of action. In constrast to vecuronium, however, rocuronium has no metabolite [3]. Although hepatic uptake and biliary excretion have been suggested to be the main mechanisms of rocuronium metabolism, a previous study in humans has shown that a 33% of a rocuronium dose of 1 mg/kg was recovered from urine within 24 h [4].
Urinary trypsin inhibitor (ulinastatin, UTI, Ulistin®, Han Lim Pharmaceutical, Seoul, Korea) is an intrinstic serine-protease inhibitor, which is extracted and purified from human urine [5,6]. Because ulinastatin increases both liver blood flow [7] and urine output [8], and rocuronium is eliminated mainly through the liver and partly through the kidney [4], hepatic elimination of rocuronium might be enhanced by ulinastatin. A previous study on protease inhibitor homologues showed that ulinastatin releases acetylcholine at the neuromuscular junction, and this may cause resistance to rocuronium [9]. Saitoh et al. [10] reported that, in anaesthetized healthy patients, the administration of ulinastatin delayed the onset of neuromuscular block and accelerated the recovery from vecuronium induced neruomuscular block.
In this study, we hypothesized that ulinastatin will also accelerate the recovery of rocuronium-induced neuromuscular block. To the best of our knowledge, the neuromuscular effect of rocuronium after the administration of ulinastatin has not been investigated previously.
Therefore, the aim of the present study was to investigate the onset of neuromuscular block and recovery of post-tetanic count (PTC) and train-of-four (TOF) responses, after administration of ulinastatin, in anesthetized patients receiving rocuronium.
After approval and, This study was approved by the Institutional Review Board and informed consent was obtained from all participants. In total, 44 patients with American Society of Anesthesiologists physical status I or II and undergoing elective surgery with a general anesthesia technique were studied. Exclusion criteria included body weight 30% greater than the ideal values, chronic antibiotic therapy, acute and chronic renal or hepatic disease, neuromuscular disorders, and previous history of an unusual reaction to neuromuscular blocking drugs. The patients were randomly divided into two groups of 22 patients each, i.e. the ulinastatin group and the control group. Glycopyrrolate 0.2 mg/kg IM was given 30 min before the induction of anesthesia, as premedication. The skin overlying the ulnar nerve at each wrist was lightly abraded with the plastic backing of an ECG electrode and then cleansed with isopropyl alcohol. Two electrodes were placed over the prepared skin just lateral to the flexor carpi ulnaris tendon. A force displacement transducer was attached to the thumb of the investigated arm. All patients were fasted for at least 6 hours before surgery. Anesthesia was performed by the same experienced anesthesiologist, assisted by an appropriately trained anesthesia nurse. Standard intraoperative monitoring included electrocardiograph, non-invasive measurement of blood pressure, pulse oximetry, capnography and body temperature (Datex-Ohmeda, Helsinki, Finland). The non-invasive blood pressure cuff was placed on the opposite side of the arm in which drug was given intravenously, to minimize the influence of BP cuff inflation during initial drug distribution.
After placement of the non-invasive monitors and during pre-oxygenation, the TOF-Watch SX (Organon Ireland Limited, Dublin, Ireland) peripheral nerve stimulator was calibrated by pressing the CAL button for one second. If the initial calibration procedure resulted in maximal stimulating current of 60 mA, the device was recalibrated, with a maximum of three total calibrations, in an effort to reach a lower stimulating current. In the ulinastatin group, bolus dose of ulinastatin 5,000 U/kg was administered intravenously before induction of anesthesia. In the control group, 0.1 ml/kg of normal saline was given instead of ulinastatin. Immediately after the injection of either ulinastatin or normal saline, target of propofol (Fresofol 2%, Fresenius, Germany) was started at 4 µg/ml and that of remifentanil was initially started at 2 ng/ml, for adequate induction by an infusion pump (Fresenius Infusomat CP®; Bad Homburg, Germany). After the loss of eyelash reflex was confirmed, the ulnar nerve was trans-cutaneously stimulated supramaximally at the wrist, with square pulses of 0.2 milliseconds duration, delivered in a TOF sequence at 2 Hz and repeated every 12 seconds. The PTC with the TOF-Watch peripheral nerve stimulator was obtained by pressing the PTC button and recording the result displayed on the monitor. The post-tetanic count was repeated at 3 minute intervals, until the TOF-Watch stimulator detected the presence of at least one twitch prior to PTC. Thereafter, TOF measurements were repeated at 12 seconds interval. Before tracheal intubation, rocuronium 0.6 mg/kg was administered intravenously. The time interval between the administration of either ulinastatin or normal saline and that of rocuronium was 2 min. After the rocuronium injection, the disappearance of the TOF response was regarded as the onset of neuromuscular block. The time to the onset of neuromuscular block was compared between the ulinastatin and the control groups. In addition, in both groups, time courses of recovery of PTC were compared. The times from the injection of rocuronium to the return of T1, T2, T3, and T4 (i.e. the first, second, third, and fourth response in TOF) were compared between the two groups.
All patients received total intravenous anesthesia (TIVA) of remifentanil and propofol using the effect site target infusion system. In each group, patients received a mixture of air and 30% oxygen.
The initial target of propofol was set at 4 µg/ml; after induction, the target was maintained at 4 µg/ml until the end of surgery. The target of remifentanil was initially set at 2 ng/ml and was later adjusted between 2 and 4 ng/ml, according to clinical needs. Either the decrease in systolic blood pressure below 85 mmHg or the decrease in mean blood pressure below 55 mmHg was treated by decreasing the target of remifentanil to a minimum level of 2 ng/ml. In addition, the patients received rapid infusion of Ringer's acetate solution and/or 5-10 mg of intravenous ephedrine, when considered necessary. The fresh gas flow was kept at 6 L/min until tracheal intubation, using 100% oxygen. During maintenance of anesthesia, the gas flow was set at 4 L/min, using the above mentioned gas mixture, i.e. air plus 30% oxygen. Ventilation was controlled sufficiently to maintain normocarbia. The concentrations of anesthetics and PET CO2 were measured, using a multiple gas monitor (Capnomac Ultima; Datex Inc., Helsinki, Finland).
All results were expressed as either number of patients or mean ± SD. Patient data were compared between the two groups, using the analysis of variance (ANOVA) and the unpaired t-test. The time to the onset of neuromuscular block and the times to the return of PTC were compared between the study and control groups, using the unpaired t-test. Similarly, times for the return of T1, T2, T3, or T4 responses were compared between the study group and control groups, using the unpaired t-test. A P value < 0.05 was considered as statistically significant. Statistical analyses were performed with the statistical package R 2.11.1 (R Foundation for Statistical Computing, Vienna, Austria).
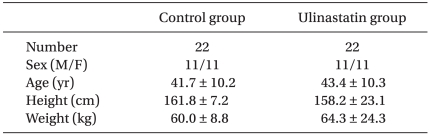
There were no differences between the two groups in sex, age, body height, and body weight, as presented in Table 1.
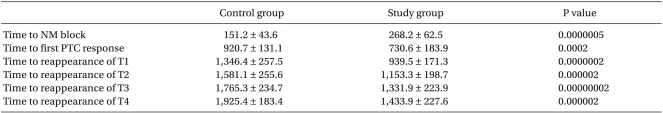
The time to the onset of neuromuscular block was significantly longer in the study group than in the control group (268 ± 62 vs. 151 ± 43s, P < 0.05) (Table 2).
The time from the rocuronium injection to the return of the first PTC response was significantly shorter in the study group than in the control group (920 ± 131 vs 730 ± 183s, P < 0.05) (Table 2). Also, the times to the return of the T1, T2, T3, and T4 responses were significantly shorter in the study group than in the control group (1,346 ± 257 vs 939 ± 171s, 1,581 ± 255 vs 1,153 ± 198s, 1,765 ± 234 vs 1,331 ± 223s, and 1,925 ± 183 vs 1,433 ± 227s, respectively, P < 0.05) (Table 2).
After the administration of either ulinastatin or normal saline, no patient showed severe hypertension (i.e. systolic arterial pressure > 200 mmHg) or hypotension (i.e. systolic arterial pressure < 80 mmHg), severe tachycardia (heart rate > 120 bpm), bradycardia (heart rate < 50 bpm), or arrhythmia.
We investigated the neuromuscular block characteristics of rocuronium affected by bolus administration of ulinastatin. We hypothesized that bolus administration of ulinastatin would delay the onset of the rocuronium-induced neuromuscular block and would accelerate the recovery from the neuromuscular block. The times to the return of PTC, T1, T2, T3 and T4 responses were shortened because the protease inhibitor homolgues increase the release of acetylcholine (Ach), in response to motor nerve stimulation [10]. It has been demonstrated that ulinastatin increases both the liver blood flow [7] and the urine output [8]. Rocuronium is eliminated mainly through the liver and partly through the kidney [4]; therefore, the hepatic elimination of rocuronium might be enhanced by ulinastatin, which accelerates the recovery from neuromuscular block. We consider that the onset of neuromuscular blockade was delayed and the recovery from neuromuscular block was accelerated after the administration of ulinastatin.
Ulinastatin, a urinary trypsin inhibitor, is a glycoprotein derived from human urine, and is a serine protease inhibitor found in human urine and blood [11]. Ulinastatin is secreted when inter-α-trypsin inhibitors are degraded by the neutrophilic enzyme elastase [11]. It also has been known as mingin, human inhibitor 30, miraclid, and bikunin [12]. Ulinastatin is indicated in acute inflammatory disorders, including acute pancreatitis, systemic inflammatory reaction syndrome (SIRS), circulatory insufficiency, disseminated intravascular coagulation (DIC) and multiple organ failure [13]. The urinary trypsin inhibitor exists naturally in the human body and is secreted in increased quantities during stressful conditions, such as inflammation, cancer, infection, and tissue damage [14]. It is composed of 143 amino acid residues and two Kunitz-type protease inhibitor domains [15]. Both Kunitz domains inhibit serine proteases, especially elastase. The levels of several proteases, such as elastase, cathepsin G, and collagenase, increase during both infection and inflammation; therefore, protease inhibitors have anti-inflammatory activity [15].
To date, knowledge of the routes of elimination of rocuronium has been limited to laboratory animal data and to urinary excretion data in man. In animals, 9-25% of radiolabelled rocuronium was found in urine and 65-75% in feces [3]. In the isolated perfused rat, rocuronium was rapidly taken up, with a high extraction ratio, and was rapidly excreted into the bile [16]. In cats, 54% of the intravenous dose was found in the bile and 21% in liver homogenate [17]. In addition, when the liver was excluded from the circulation (via a portal vein-to-inferior vena cava shunt), the clinical duration of action of rocuronium increased almost threefold [17]. These findings suggest that both the pharmacokinetics and the time course of neuromuscular effect of rocuronium may be altered by liver disease [18]. Since ulinastatin increases the blood flow in the liver [7], the hepatic elimination of rocuronium would be increased after administration of ulinastatin.
A previous study in humans has shown that 33% of the rocuronium dose of 1 mg/kg was recovered from urine within the first 24 hours [4]. Another study showed that about 10% of the IV dose of rocuronium was excreted in urine [17,19]. Kocabas et al. [20] reported that the clearance of rocuronium was reduced by 39% in patients suffering from renal failure compared with controls, with 84% increase in the mean residence time, whereas the volume of distribution was unaffected by renal failure. Therefore, the duration of action of rocuronium can be expected to be prolonged, as it undergoes organ-dependent elimination [20]. Proost et al. [3] demonstrated that rocuronium is taken up by the liver and excreted into the bile in very high concentration [3]. The proportion of rocuronium excreted into urine within 7 days was 26%. Since ulinastatin increases the urine volume [8], rocuronium would be rapidly eliminated in the kidney after ulinastatin injection.
In this study, the time interval from the administration of ulinastatin to that of rocuronium was 2 min. Sugiki et al. [21] reported that the distribution half-life of the protease inhibitor was 3.9 min. However, ulinastatin given only 2 mininutes before the rocuronium injection significantly delayed the onset of the neuromuscular block. There were no previous studies investigating the onset of the ulinastatin-induced effect on neuromuscular transmission. However, Saitoh et al. [10] reported that, after bolus administration of ulinastatin, the ulinastatin-induced effect on the skeletal muscles may become apparent within 2 minutes.
Previous studies done by Saitoh et al. on the effect of ulinastatin on vecuronium included isoflurane as the main anesthetic agent; isoflurane is known to potentiate non-depolarizing neuromuscular blockade [10]. We wanted to exclude any factors that could have had any effect on the potency of neuromuscular blockade of rocuronium. Therefore, in our study, patients received total intravenous anesthesia (TIVA), using target controlled infusion of propofol and remifentanil. The bispectral index (BIS) was not monitored in our study. The initial target of propofol was set at 4 µg/ml by an infusion pump and the target of remifentanil was initially set at 2 ng/ml, and later adjusted between 2 and 4 ng/ml, according to clinical needs. Kil et al. [22] proved that BIS shows a significant correlation with propofol and the effect site concentration of 43.5 µg/kg propofol was reached at a BIS of 41.1 ± 2.5. Another study showed that, during TIVA, the effect site concentration for blunting the hemodynamic response to endotracheal intubation was 3.5 ng/mL, using the dose of 4.0 µg/ml propofol with remifentanil [23].
It has been shown that the onset time of the neuromuscular block is partially determined by the cardiac output [24,25]. No previous studies examined the ulinastatin-induced changes in either cardiac output or blood pressure. However, Ohnishi et al. [24] showed that ulinastatin did not significantly alter the heart rate after hemorrhagic shock. In this study, there was no significant change in blood pressure and heart rate after administration of ulinastatin, which may affect the onset time of neuromuscular block.
In conclusion, in this study, we demonstrated that, in anesthetized patients, ulinastatin delays the onset of the rocuronium-induced neuromuscular block and accelerates the recovery from the neuromuscular block. This is probably because ulinastatin increases the release of acetylcholine (Ach), the blood flow in the liver, and the urine volume.
References
1. Sparr HJ, Beaufort TM, Fuchs-Buder T. Newer neuromuscular blocking agents: how do they compare with established agents? Drugs. 2001; 61:919–942. PMID: 11434449.
2. McCourt KC, Salmela L, Mirakhur RK, Carroll M, Mäkinen MT, Kansanaho M, et al. Comparison of rocuronium and sexamethonium for use during rapid sequence induction of anaesthesia. Anaesthesia. 1998; 53:867–871. PMID: 9849280.
3. Proost JH, Eriksson LI, Mirakhur RK, Roest G, Wierda JM. Urinary, biliary and faecal excretion of rocuronium in humans. Br J Anaesth. 2000; 85:717–723. PMID: 11094587.

4. Wierda JM, Kleef UW, Lambalk LM, Kloppenburg WD, Agoston S. The pharmaodynamics and pharmacokinetics of Org 9426, a new non-depolarizing neuromuscular blocking agent, in patients anesthetized with nitrous oxide, halothane and fentanyl. Can J Anaesth. 1991; 38:430–435. PMID: 1829656.
5. Tanaka Y, Maehara S, Sumi H, Toki N, Moriyama S, Sasaki K. Purification and partial characterization of two forms of urinary trypsin inhibitor. Biochim Biophys Acta. 1982; 705:192–199. PMID: 6810937.

6. Bromke BJ, Kueppers F. The major urinary protease inhibitor: simplified purification and characterization. Biochem Med. 1982; 27:56–67. PMID: 6921994.

7. Matsumoto N, Ohara K, Yoshida N, Nakamura S, Nagasaka H, Aikawa K, et al. Protective effects of ulinastatin on hepatic oxygen metabolism during halothane anesthesia in the presence of graded hypoxic hypoxemia. Masui. 1989; 38:531–539. PMID: 2724518.
8. Aoike I, Takano Y, Gejyo F, Arakawa M. Ulinastatin gives rise to an effectual diuresis in oliguric acute renal failure. Nephron. 1989; 52:368–369. PMID: 2770956.

9. Harvey AL, Karlsson E. Protease inhibitor homologues from mamba venoms: facilitation of acetylcholine release and interactions with prejunctional blocking toxins. Br J Pharmacol. 1982; 77:153–161. PMID: 6751453.

10. Saitoh Y, Fujii Y, Oshima T. The ulinastatin-induced effect on neuromuscular block caused by vecuronium. Anesth Analg. 1999; 89:1565–1569. PMID: 10589650.

11. Hirose J, Ozawa T, Miura T, Isaji M, Nagao Y, Yamashiro K, et al. Human neutrophil elastase degrades inter-alpha-trypsin inhibitor to liberate urinary trypsin inhibitor related proteins. Biol Pharm Bull. 1998; 21:651–656. PMID: 9703243.

12. Pugia MJ, Lott JA. Pathophysiology and diagnostic value of urinary trypsin inhibitors. Clin Chem Lab Med. 2005; 43:1–16. PMID: 15653436.

13. Inoue K, Takano H, Yanagisawa R, Yoshikawa T. Protective effects of urinary trypsin inhibitor on systemic inflammatory response induced by lipopolysaccharide. J Clin Biochem Nutr. 2008; 43:139–142. PMID: 19015747.

14. Pugia MJ, Valdes R Jr, Jortani SA. Bikunin (urinary trypsin inhibitor): structure, biological relevance, and measurement. Adv Clin Chem. 2007; 44:223–245. PMID: 17682344.

15. Kobayashi H, Suzuki M, Hirashima Y, Terao T. The protease inhibitor bikunin, a novel anti-metastatic agent. Biol Chem. 2003; 384:749–754. PMID: 12817471.

16. Proost JH, Roggeveld J, Weirda JM, Meijer DK. Relationship between chemical structure and physiochemical properties of series of bulky organic cations and their hepatic uptake and biliary excretion rates. J Pharmacol Exp Ther. 1997; 282:715–726. PMID: 9262335.
17. Khuenl-Brady K, Castagnoli KP, Canfell PC, Caldwell JE, Agoston S, Miller RD. The neuromuscular blocking effects and pharmacokinetics of ORG 9426 and ORG 9616 in the cat. Anesthesiology. 1990; 72:669–674. PMID: 1969717.

18. Magorian T, Wood P, Caldwell J, Fisher D, Segredo V, Szenohradszky J, et al. The pharmacokinetics and neuromuscular effects of rocuronium bromide in patients with liver disease. Anesth Analg. 1995; 80:754–759. PMID: 7893030.

19. Szenohradszky J, Fisher DM, Segredo V, Caldwell JE, Bragg P, Sharma ML, et al. Pharmacokinetics of rocuronium bromide (ORG 9426) in patients with normal renal function or patients undergoing cadaver renal transplantation. Anesthesiology. 1992; 77:899–904. PMID: 1443743.

20. Kocabas S, Yedicocuklu D, Askar FZ. The neuromuscular effects of 0.6mgkg-1 rocuronium in elderly and young adults with or without renal failure. Eur J Anaesthesiol. 2008; 25:940–946. PMID: 18538050.
21. Sugiki M, Maruyama M, Yoshida E, Sumi H, Mihara H. Acid-stable protease inhibitor in chronic phase of carrageenin-induced inflammation in rats. Inflammation. 1991; 15:281–289. PMID: 1769732.

22. Kil HY, Lee SI, Lee SJ, Lee SW, Lee DH. The bispectral index and modified observer's assessment of alertness/sedation scale comparable to effect site concentration of propofol in Koreans. Korean J Anesthesiol. 2000; 38:251–257.

23. Heo BH, Yoon MH, Lee HG, Kim WM. Effect-site concentration of remifentanil for blunting hemodynamic response to double lumen endobronchial intubation during target controlled infusion-total intravenous anesthesia using propofol with remifentanil. Korean J Anesthesiol. 2009; 57:8–12.

24. Ohnishi H, Suzuki K, Niho T, Ito C, Yamaguchi K. Protective effects of urinary trypsin inhibitor in experimental shock. Jpn J Pharmacol. 1985; 39:137–144. PMID: 2418241.

25. Muñoz HR, González AG, Dagnino JA, González JA, Pérez AE. The effect of ephedrine on the onset of rocuronium. Anesth Analg. 1997; 85:437–440. PMID: 9249127.




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download