Abstract
In the spring of 2011, a cluster of lung injuries caused by humidifier disinfectant (HD) usage were reported in Korea. Many patients required mechanical ventilation, extracorporeal membrane oxygenation, and even lung transplantation (LTPL). However, the long-term course of HD-associated lung injury remains unclear because the majority of survivors recovered normal lung function. Here we report a 33-year-old woman who underwent LTPL approximately four years after severe HD-associated lung injury. The patient was initially admitted to the intensive care unit and was supported by a high-flow nasal cannula. Although she had been discharged, she was recurrently admitted to our hospital due to progressive lung fibrosis and a persistent decline in lung function. Finally, sequential double LTPL was successfully performed, and the patient’s clinical and radiological findings showed significant improvement. Therefore, we conclude that LTPL can be a therapeutic option for patients with chronic inhalation injury.
Inhalation injury increases acute morbidity and mortality.[1,2] Commencing in February 2011, a cluster of young adults who were previously healthy and without a history of lung disease were admitted to the intensive care unit (ICU) of a tertiary hospital in Seoul with severe respiratory distress of an unknown origin.[3] The findings of these patients were reported to the Korean Centers for Disease Control and Prevention. Moreover, subsequent epidemiological studies conducted in Korea concluded that exposure to nebulized humidifier disinfectants (HDs) in small water tanks can cause lung disease characterized by diffuse interstitial fibrosis.[4,5] During the affected period, many patients required mechanical ventilation, extracorporeal membrane oxygenation, and even lung transplantation (LTPL) for survival.[3] However, information regarding the chronic course of the disease is lacking. In this report, we describe the case a patient with severe HD-associated lung injury who survived for approximately four years with conservative treatment before receiving LTPL.
A 33-year-old woman was referred to our hospital for the evaluation of nonproductive cough, dyspnea, and progressive bilateral infiltration on chest computed tomography (CT). Two months prior, the patient was pregnant and had symptoms of an upper respiratory infection. She was admitted to the local hospital. However, the symptoms persisted despite antibiotic treatment. The patient was referred to another hospital for further evaluation, and chest CT revealed diffuse centrilobular ground-glass opacity (GGO) nodules with patchy multifocal opacities (Fig. 1A.). An emergency cesarean section was performed, followed by a surgical lung biopsy exhibiting early stages of diffuse alveolar damage. Under the diagnosis of atypical pneumonia, broad-spectrum antibiotics were used. Neither clinical nor radiological improvement was observed, and the patient was transferred to our hospital. The patient had previously been healthy, without a history of lung disease, and was a never smoker. Her initial vital signs upon ICU admission were as follows: blood pressure, 131/77 mmHg; heart rate, 95 beats/min; respiratory rate, 32 breaths/min; and body temperature, 36.3°C. Despite the application of a partial rebreather mask with a flow rate of 15 L/min and FiO2 of 0.7, her PaO2/FiO2 (PF) ratio was 124 mmHg, showing moderate hypoxemia. The chest CT revealed an increase in the extent of GGOs in both upper lobes with newly appeared pneumomediastinum and interstitial emphysema (Fig. 1B). There was no evidence of left ventricular dysfunction or increased left atrial pressure, as assessed via echocardiography. Findings of laboratory studies demonstrated a white blood cell count of 26,900/mm3 with 92.0% neutrophils, hemoglobin 9.9 g/dL, and a platelet count of 344,000/mm3. Electrolytes, measures of renal function, and liver enzymes were within normal limits. Her C-reactive protein levels were elevated to 7.96 mg/dL.
By the time of her admission, we were aware that several other patients in other regions of Korea were identified as having HD-associated lung injury, including pregnant women, infants, and preschoolers. The patient’s clinical, radiological, and pathological findings as well as the previous history of HD use enabled the probable diagnosis of HD-associated lung injury. She used a disinfectant containing polyhexamethylene guanidine phosphate (PHMG-phosphate) every other day for about 18 months, including eight months of pregnancy. After admission, her hypoxemia worsened, and a high-flow nasal cannula (HFNC) (Optiflow, Fisher & Paykel Healthcare, Auckland, New Zealand) was used. The patient was treated with high-dose steroid therapy and empirical antibiotics (meropenem and vancomycin). During the eight days of her ICU stay, her PF ratio consistently improved to 255 mmHg with FiO2 of 0.4. Eventually, the patient was discharged with home oxygen therapy on hospital day 31. Before discharge, a pulmonary function test (PFT) and chest CT were performed. The forced vital capacity (FVC) was 0.92 L (28% of predicted), the forced expiratory volume in 1 s (FEV1) was 0.88 L (31% of predicted), FEV1/FVC was 0.96, and the diffusing capacity of the lung for carbon monoxide (DLCO) was 25% of predicted, showing a severe restrictive pattern. The chest CT revealed improved GGOs in both upper lobes, but diffuse centrilobular GGO nodules and pneumomediastinum remained, and a newly appeared left pneumothorax was also observed (Fig. 1C). We discussed with the patient and her family members about her anticipated clinical course and possibility of LTPL, but they refused, primarily due to economic reasons.
After discharge, the patient’s disease course was followed by both chest CTs and PFTs. Follow-up CT exams revealed residual diffuse centrilobular GGO nodules and multiple air cysts with parenchymal distortion and fibrosis in both lungs (Fig. 2). The FVC, FEV1, and DLCO exhibited a decline that persisted during the following years (Fig. 3). She was recurrently admitted to our hospital because of pneumonia and combined hypercapnia. The patient finally agreed to receive LTPL when the government decided to compensate victims for medical expenses related to HD-associated lung injury. Accordingly, she was listed on the Korean Network for Organ Sharing. During the waiting period for an appropriate donor, she developed profound dyspnea caused by left pneumothorax, and was admitted for a chest tube insertion. Fortunately, an appropriate donor was identified, and the patient underwent sequential double LTPL. There were no postoperative complications, and her chest CT and PFT findings revealed a significant improvement (Fig. 2 and Fig. 3, respectively). The pathology of her explanted lung exhibited end-stage lung pathology with patchy fibrosis, honeycomb changes, and bronchiolar fibrous obliteration that were consistent with HD-associated lung injury (Fig. 4).
HDs were originally introduced in the market as cleansing detergents for the water tanks of humidifiers. However, in Korea, people started diluting detergents in water to prevent the growth of molds or bacteria in the tanks. Most HDs in the Korean market contained the guanidine chemical PHMG-phosphate.[6] The disinfectant dissolved in water was dispersed into the air as nanosized particles (30–80 nm) by the humidifier’s aerosolizer, enabling these particles to easily reach the distal airways.[7] HD-associated lung injury is biologically plausible, based on a toxicological study demonstrating that the histopathological findings in rats that inhaled PHMG were identical to those of patients with lung injury.[8] We previously reported 17 cases of HD-associated lung injury. Five of the ten patients in the ICU died, and four received LTPL during the affected period. Three of the four patients who received LTPL remain active without any limitations. To date, the follow-up time is more than four years, although one patient died of acute rejection and combined pneumonia eight months after LTPL. The unique radiological transition from consolidation to centrilobular GGO nodules, and the histological features of diffuse alveolar damage, obliterative bronchiolitis, and organizing pneumonia without granulomas were observed.[3] However, the long-term course of HD-associated lung injury has not been evaluated because the majority of survivors had recovered normal lung function over the following years. Further, during the follow-up of 40 confirmed patients (median: 48 months), the median FVC (2.38 L to 3.12 L; p < 0.001) and the median FEV1 (1.98 L to 2.64 L; p < 0.001) significantly increased (unpublished data). Here, we describe a 33-year-old female patient who underwent LTPL as a final therapeutic option for chronic HD-associated lung injury. Her clinical, radiological, and pathological findings were similar to those who received LTPL during the affected period. This case suggests that cases of refractory inhalation lung injury are possible candidates for LTPL. LTPL is typically conducted in patients with irreversible and end-stage lung disease,[9] although several cases of LTPL to treat refractory respiratory failure with inhalation injury have been reported. [10-12] In previous reports, the two out of three patients were in good condition with a follow-up time of two-and-half years and over four years, respectively. However, one patient died of respiratory failure due to chronic rejection approximately two years after a bilateral LTPL.[12] In our case, due to the progressive fibrotic changes with air leaks on the CT exams and the persistent decline in lung function, we considered the condition as irreversible and bilateral LTPL to be the only option. End-stage fibrosis with honeycomb changes in the patient’s explanted lung (not observed in the subacute stage of HD-associated lung injury) supported our clinical judgement.
Our case is also an example of the successful use of HFNC in acute respiratory failure due to HD-associated lung injury. HFNC is a novel oxygen device that can deliver up to 100% heated and humidified oxygen via a wide-bore nasal cannula at a maximum flow rate of 60 L/min. HFNC has been found to be effective for both hypercapnic[13,14] and mild to moderate hypoxemic[15] respiratory failure. In a recent multicenter trial, compared with standard oxygen therapy and noninvasive ventilation, the use of HFNC in acute hypoxemic respiratory failure significantly decreased the total ICU and 90-day mortality, and the intubation rate in patients with a PF ratio of ≤200 mmHg.[16] On the other hand, HFNC failure with late intubation (>48 h after HFNC initiation) was associated with a higher overall ICU mortality and poorer extubation success.[17] In our case, the patient initially presented with moderate hypoxemia (PF ratio, 124 mmHg) and hypercapnia (PaCO2, 55 mmHg). With an oxygen flow of 35–40 L/min via HFNC, her dyspnea was alleviated and oxygenation improved preventing intubation.
In conclusion, we report the case of successful LTPL in a patient with a long-term course of HD-associated lung injury. This encourages the consideration of cases of chronic inhalation injury as possible candidates for LTPL.
References
1. Tredget EE, Shankowsky HA, Taerum TV, Moysa GL, Alton JD. The role of inhalation injury in burn trauma. A Canadian experience. Ann Surg. 1990; 212:720–7.

2. Suzuki M, Aikawa N, Kobayashi K, Higuchi R. Prognostic implications of inhalation injury in burn patients in Tokyo. Burns. 2005; 31:331–6.

3. Hong SB, Kim HJ, Huh JW, Do KH, Jang SJ, Song JS, et al. A cluster of lung injury associated with home humidifier use: clinical, radiological and pathological description of a new syndrome. Thorax. 2014; 69:694–702.

4. Kim HJ, Lee MS, Hong SB, Huh JW, Do KH, Jang SJ, et al. A cluster of lung injury cases associated with home humidifier use: an epidemiological investigation. Thorax. 2014; 69:703–8.

5. Kim KW, Ahn K, Yang HJ, Lee S, Park JD, Kim WK, et al. Humidifier disinfectant-associated children’s interstitial lung disease. Am J Respir Crit Care Med. 2014; 189:48–56.
6. Lee JH, Kim YH, Kwon JH. Fatal misuse of humidifier disinfectants in Korea: importance of screening risk assessment and implications for management of chemicals in consumer products. Environ Sci Technol. 2012; 46:2498–500.

7. Suda T, Sato A, Ida M, Gemma H, Hayakawa H, Chida K. Hypersensitivity pneumonitis associated with home ultrasonic humidifiers. Chest. 1995; 107:711–7.

8. Kim YB, Choi SJ, Yang YS, Kim YH, Song CW, Cho JW, et al. Comparative pulmonary response to aerosolized humidifier disinfectants by intratracheal instillation and inhalation exposure. In : 2013 Annual Meeting Poster #207; 2013.
9. Christie JD, Edwards LB, Aurora P, Dobbels F, Kirk R, Rahmel AO, et al. The registry of the international society for heart and lung transplantation: twenty-sixth official adult lung and heart-lung transplantation report-2009. J Heart Lung Transplant. 2009; 28:1031–49.

10. Barrio J, Sánchez C, Vicente R, Ramos F, Montero R, Morales P, et al. Successful sequential double-lung transplantation for adult respiratory distress syndrome after long-term mechanical ventilation. Eur J Anaesthesiol. 2004; 21:326–7.

11. Hayes D Jr. Lung transplantation for bronchiectasis due to smoke inhalation. Burns. 2011; 37:e24–8.

12. Pirjavec A, Kovic I, Lulic I, Zupan Z. Massive anhydrous ammonia injury leading to lung transplantation. J Trauma. 2009; 67:E93–7.

13. Millar J, Lutton S, O’Connor P. The use of high-flow nasal oxygen therapy in the management of hypercarbic respiratory failure. Ther Adv Respir Dis. 2014; 8:63–4.

14. Nilius G, Franke KJ, Domanski U, Rühle KH, Kirkness JP, Schneider H. Effects of nasal insufflation on arterial gas exchange and breathing pattern in patients with chronic obstructive pulmonary disease and hypercapnic respiratory failure. Adv Exp Med Biol. 2013; 755:27–34.

15. Sztrymf B, Messika J, Bertrand F, Hurel D, Leon R, Dreyfuss D, et al. Beneficial effects of humidified high flow nasal oxygen in critical care patients: a prospective pilot study. Intensive Care Med. 2011; 37:1780–6.

Fig. 1.
The three stages of humidifier disinfectant-associated lung injury in a 33-year-old woman. (A) Initial computed tomography (CT) scan revealing diffuse, centrilobular ground-glass opacity (GGO) nodules with patchy multifocal consolidations. (B) On the CT obtained on the day of admission to our hospital, the consolidations disappeared, but diffuse GGO nodules became more distinct (lower), with newly appeared GGOs in both upper lobes (upper). (C) Follow-up CT scan obtained two weeks after exhibited improved GGOs in both upper lobes (upper), but diffuse GGO nodules and pneumomediastinum remained (lower). A small amount of left pneumothorax is also visible (upper).
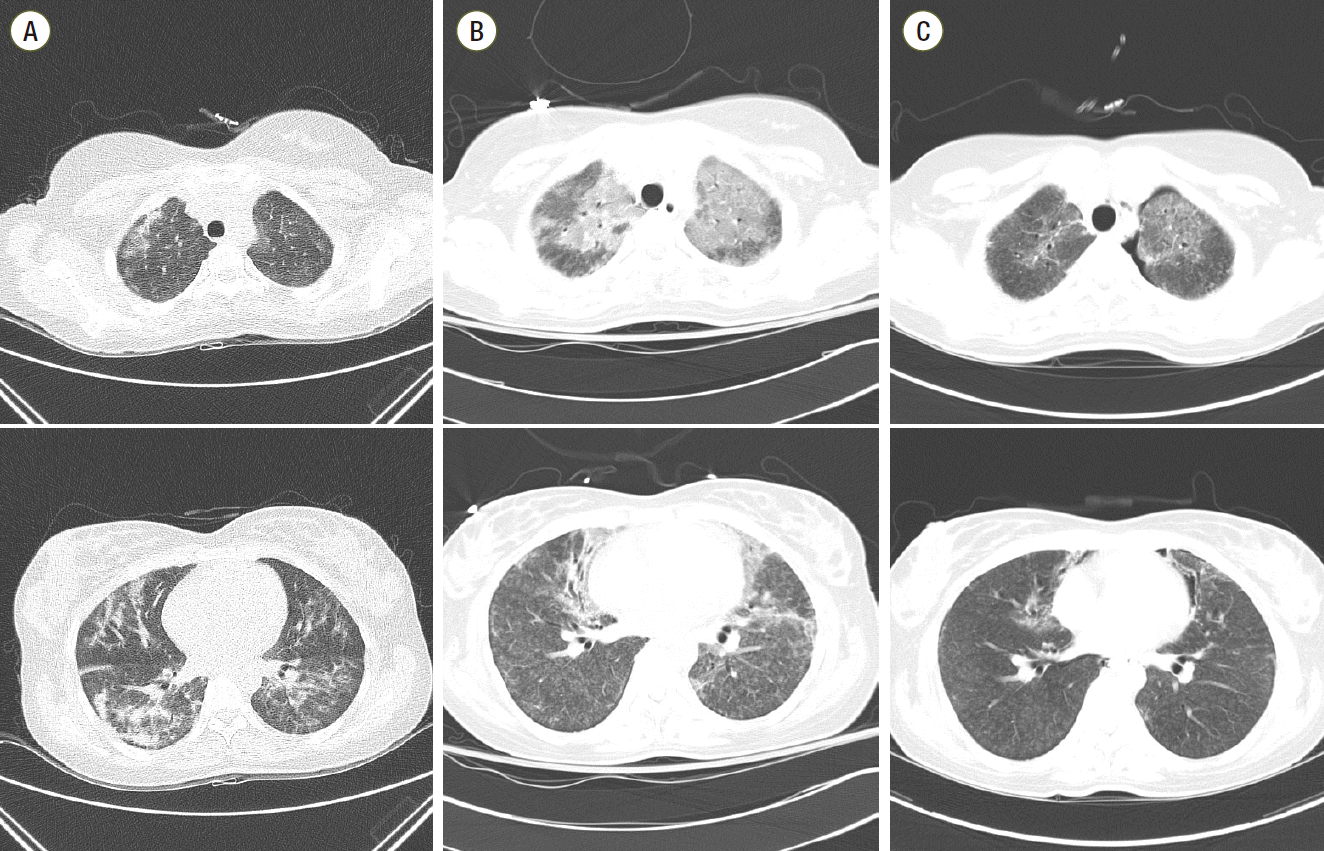
Fig. 2.
The patient’s disease course after discharge. (A-C) Follow-up computed tomography (CT) scans exhibited residual diffuse centrilobular ground-glass opacity nodules and multiple air cysts with parenchymal distortion and fibrosis. (D) Normal CT scans two months after lung transplantation.
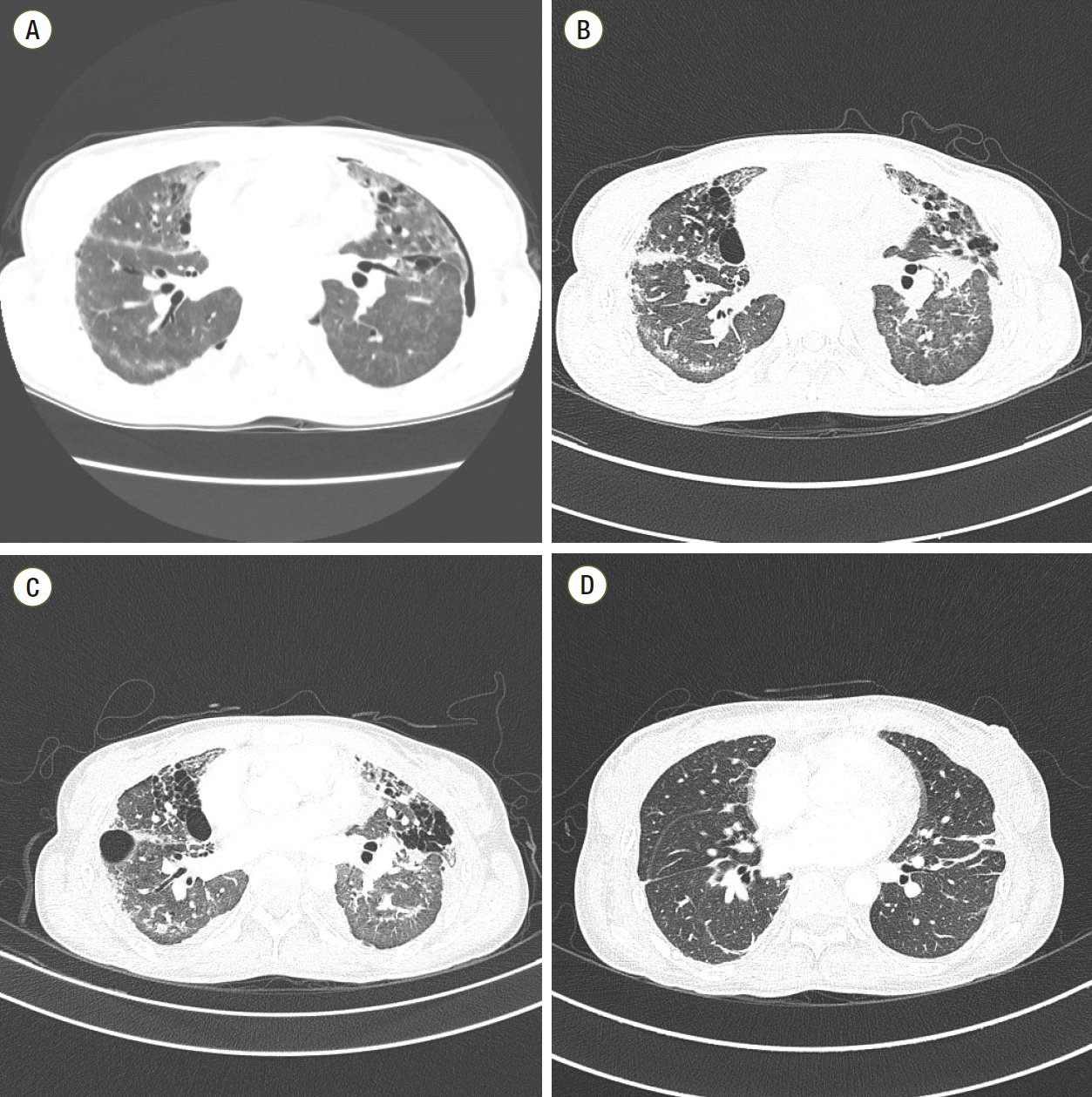
Fig. 3.
Serial pulmonary function test findings showing a decline that persisted during follow-up. After the lung transplantation, these findings were significantly improved. LTPL: lung transplantation; FVC: forced vital capacity; FEV1: forced expiratory volume in 1 s; DLCO: diffusing capacity of lung for carbon monoxide.
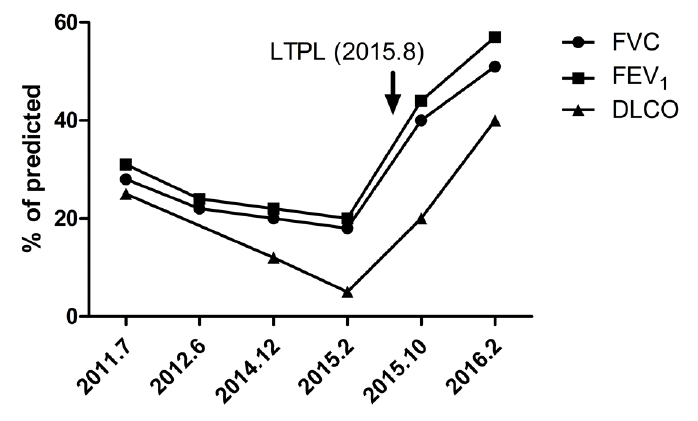
Fig. 4.
Pathology of the explanted lungs. (A) Centrilobular and patchy fibrosis with honeycomb changes (H&E stain, ×10). Characteristically, the lung architecture was destroyed and replaced by irregular cystic sacs with dense fibrotic walls. (B) Patchy, interstitial lymphoid cellular infiltration with fibrosis of the bronchioles (H&E stain, ×40).





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download