Abstract
Background:
Colistin (polymyxin E) is active against multidrug-resistant Gram-negative bacteria (MDR-GNB). However, the effectiveness of inhaled colistin is unclear. This study was designed to assess the effectiveness and safety of aerosolized colistin for the treatment of ventilator-associated pneumonia (VAP) caused by MDR-GNB.
Methods:
In this retrospective longitudinal study, we evaluated the medical records of 63 patients who received aerosolized colistin treatment for VAP caused by MDR-GNB in the medical intensive care unit (MICU) from February 2012 to March 2014.
Results:
A total of 25 patients with VAP caused by MDR-GNB were included in this study. The negative conversion rate was 84.6% after treatment, and acute kidney injury (AKI) occurred in 11 patients (44%, AKI group). The average length of MICU stay and colistin treatment-related factors, such as daily and total cumulative doses and administration period, were not significantly different between groups. In-hospital mortality tended to be higher in the AKI group (p = 0.07). Multivariate analysis showed that a body mass index less than 18 was an independent risk factor of mortality (odds ratio [OR] = 21.95, 95% confidence interval [CI] 1.59-302.23; p = 0.02). Notably, AKI occurrence was closely related to the administration of more than two nephrotoxic drugs combined with aerosolized colistin (OR = 15.03, 95% CI 1.40-161.76; p = 0.025) and septic shock (OR = 8.10, 95% CI 1.40-161.76; p = 0.04).
Conclusions:
The use of adjunctive aerosolized colistin treatment appears to be a relatively safe and effective option for the treatment of VAP caused by MDR-GNB. However, more research on the concomitant use of nephrotoxic drugs with aerosolized colistin will be necessary, as this can be an important risk factor of development of AKI.
Colistin (Polymyxin E) is an antibiotic whose mode-of-action changes the structure of phospholipids in bacterial cell membranes.[1] After its discovery in the 1940s from Bacillus polymyxa, colistin was commonly used to treat Gram-negative bacterial infections up until the early 1980s.[2] In spite of its excellent bactericidal efficacy, colistin has been replaced by new antibiotics, and its use has largely decreased due to a 50% rate of serious nephrotoxicity and neurotoxicity associated with intravenous administration.[2-4] Recently, however, colistin has been re-introduced in clinical practice due to increasing rates of nosocomial infection by multidrug-resistant Gram-negative bacteria (MDR-GNB), particularly Pseudomonas aeruginosa and Acinetobacter baumannii. Because these bacteria are highly susceptible to colistin, its use has again increased.[2]
A number of recent studies on colistin indicate that its neurotoxicity has decreased and have failed to identify a clear association between colistin treatment and neurotoxic events. However, the incidence of nephrotoxicity is still high, ranging from 8-45%.[2-4] Additionally, its low penetration into infected tissues is a potential obstacle for intravenous injection.[3-6] A previous study demonstrated that aerosolized colistin treatment was effective for treating cystic fibrosis patients with chronic P. aeruginosa lung infections.[7] Furthermore, favorable results for aerosolized colistin treatment have been reported in patients with ventilator-associated pneumonia (VAP),[8] including a lower incidence of nephrotoxicity with aerosol treatment than with intravenous injection. Nonetheless, in these studies, nephrotoxicity was still frequent (15-32%).[9,10] Critically, research on the relationship between daily and cumulative colistin doses and the incidence of nephrotoxicity with aerosolized colistin treatment has been limited,[11,12] The purpose of this study was to clarify the effectiveness, safety profiles, and the incidence and related risk factors of inhaled colistin-associated nephrotoxicity in patients with VAP caused by MDR-GNB.
The data were obtained retrospectively from medical records of patients who received aerosolized colistin treatment for VAP caused by MDR-GNB in the medical intensive care unit (MICU) at Chonbuk National University Hospital (CNUH) between February 2012 and March 2014. Exclusion criteria were: (i) patients who received colistin for < 72 hours; (ii) patients who were diagnosed with colonization or contamination; (iii) patients who received renal replacement therapy before colistin therapy; and (iv) patients who were < 18 years of age. If a patient received multiple courses of colistin, only the first one was included in the analysis. This study was approved by the Research Ethics Committee of the CNUH under number 2014-10-024.
Demographic data on age, sex, length of MICU stay, medical history, and comorbidities were obtained from electronic medical records. The severity of disease was measured according to the Acute Physiological and Chronic Health Evaluation II (APACHE II) score. The results of microbiological culture and antibiogram, daily and total cumulative colistin doses, aerosol treatment period, and any concomitant medications with nephrotoxic potential were reviewed. We designated any drug that was used for more than 72 hours in addition to aerosolized colistin as a concomitant medication. In addition, laboratory data on complete blood counts, blood chemistry (including renal and liver function markers), and the levels of high-sensitivity C-reactive protein (hs-CRP) and procalcitonin (PCT) were obtained just before and at the completion of treatment. These measures were used to assess any adverse drug reactions, as well as to determine the efficacy of aerosolized colistin treatment.
Pneumonia was considered to be ventilator-associated if the onset occurred after 48 hours of intubation and if the infection was determined to be absent before the initiation of mechanical ventilation. Pneumonia was diagnosed based on radiographic findings of new and progressive pulmonary infiltrates and at least two of the following clinical criteria: body temperature > 38°C or < 35.5°C, leukocytosis (leukocyte count > 12,000 cells/mm3) or leukopenia (leukocyte count < 4,000 cells/mm3), and clinical evidence suggestive of pneumonia, such as purulent bronchial secretions and a decrease in oxygenation.[13]
MDR-GNB was defined as an extreme drug resistance state in which bacterial strains demonstrated in vitro resistance to penicillins, cephalosporins, carbapenems, aminoglycosides, fluoroquinolones, and monobactams.
Microbiological diagnosis of VAP was established by mini-bronchoalveolar lavage (mini-BAL) or BAL cultures positive for MDR-GNB, with a concentration of > 104 colony-forming units (CFU)/mL.[14] Bacteriological sampling was performed for all patients on the day that VAP was suspected (day 0) and before new antimicrobials were started. Bacterial colonization was defined as persistent growth of the pathogen without symptoms or signs of infection.
The severity of the clinical condition at the time of VAP diagnosis was assessed according to APACHE II[15] and clinical pulmonary infection scores.[16]
Response to treatment was assessed at the end of antimicrobial therapy. Negative conversion was defined “no growth of the pathogen,” and persistence was defined as continuous growth of the responsible pathogen regardless of the clinical outcome of the infection.[17]
In patients with normal renal function, nephrotoxicity was defined as serum creatinine > 2 mg/dL or a 50% reduction in the automatically estimated glomerular filtration rate (eGFR) using the Chronic Kidney Disease Epidemiology Collaboration equation formula (CKD-EPI)[18] compared to the value at the start of treatment or a decline in renal function that prompted renal replacement therapy. In patients with preexisting renal dysfunction, nephrotoxicity was defined as an increase > 50% of the baseline creatinine level or as a 50% reduction in the eGFR by CKD-EPI relative to the value at therapy initiation, according to risk, injury, failure, loss of kidney function, and end-stage renal failure (RIFLE) criteria.[19]
The aerosolized colistin used in this study was Coly–Mycin® M Parenteral (SteriMax Pharmaceuticals, Mississauga, ON, Canada), supplied as 400 mg of Colistimethate sodium (150 mg of colistin base activity) per vial. For patients with normal renal function, a total daily dose of 300 mg colistin base activity was inhaled as an aerosol, using a Servo Ultra Nebulizer 345 (Maquet, Sweden) 2-4 times a day.[20] Each dose was mixed with 2 mL of normal saline prior to inhalation. During nebulization, the mechanical ventilator setting was kept in pressure-controlled mode, with a pressure trigger of 2.0 cm H2O, a tidal volume of 8 mL/kg of predicted body weight, and an I:E (inspiration:expiration) ratio of 1:2. The frequency and adjusted aerosolized colistin doses were calculated using recommendations from the package insert,[21,22] with the eGFR based on CKD-EPI twice daily (morning [5:00 am] and evening [5:00 pm]) and hourly urine output. A bronchodilator (β2 adrenergic agonist and/or anticholinergic agent) was administered 20 minutes before the nebulization of aerosolized colistin to prevent bronchoconstriction. The duration of aerosolized colistin therapy was based upon the clinical response.
To determine the effectiveness of aerosolized colistin treatment for MDR-GNB pneumonia, we evaluated the length of MICU stay, mortality, mechanical ventilation day, and the microbiological outcome. The incidence and potential risk factors of nephrotoxicity and the occurrence of adverse events related to aerosolized colistin treatment were also evaluated. All potential adverse effects related to aerosolized colistin use, such as bronchoconstriction, cough, apnea, chest tightness, and arterial hypoxemia were reviewed.
Analyses were performed with IBM SPSS Statistics software, version 18.0 (IBM Co., Armonk, NY, USA). Categorical variables are presented as frequency and percentage, and these were verified by the Chi-square test. Continuous variables are expressed as mean ± standard deviation (SD) and verified by either the paired or unpaired Student’s t-test. To identify the risk factors of nephrotoxicity in aerosolized colistin treatment, the relationship between concomitant nephrotoxic drug use and the occurrence of acute kidney injury (AKI) was verified using Fisher’s exact probability test. A p-value < 0.05 was considered to be significant. Univariate and multivariate analyses of risk factors associated with aerosolized colistin-induced nephrotoxicity and mortality during treatment were performed using logistic regression models. Variables that emerged from the univariate analysis with significant p-values were candidates for inclusion in the multivariate analysis.
A total 25 patients were underwent aerosolized colistin treatment for MDR-GNB VAP in the study period and finally analyzed (Fig. 1). The mean age of the 25 patients was 66.6 ± 14.5 years. Sixteen patients (64.0%) were male, and the average body mass index (BMI) was 20.4 ± 4.1. Thirteen patients (52.0%) had cardiovascular disease, such as hypertension, and four of the remaining patients (16.0%) had cerebrovascular disease. Three additional patients (12.0%) had diabetes and chronic obstructive pulmonary disease (COPD). The mean APACHE II score for all 25 subjects was 23.0 ± 6.8 at MICU admission.
The microbiological etiologies of VAP were predominantly found to be A. baumannii (22 patients, 88.0%; single infection in 18 patients, 72.0%), followed by P. aeruginosa (six patients, 24.0%; single infection in two patients, 8.0%), and Klebsiella pneumoniae (two patients, 8.0%; all mixed infections). All three organisms were cultured from two subjects (8.0%), and two others had both A. baumannii and P. aeruginosa. One patient was treated without microorganism identification (Table 1). The mean culture days of MDR pathogens was 7.56 ± 6.3 days, and the aerosolized colistin treatment was started at 15.64 ± 9.8 days after intensive care unit admission.
Of 25 patients, 13 underwent follow-up cultures for bacteria at the end of treatment with aerosolized colistin; of these, 11 patients (84.6%) showed negative conversion, or “no growth,” of bacteria. Notably, 11 of 12 (91.7%) A. baumannii cases showed negative conversion, and one of two (50.0%) P. aeruginosa cases showed negative conversion. However, P. aeruginosa and K. pneumonia were continuously detected in one patient with negative conversion of a simultaneous A. baumannii infection. Among the 13 patients in whom follow-up cultures were performed, five of seven (71.4%) patients with normal renal function showed negative conversion of bacteria, and all six patients (100%) with AKI showed negative conversion. There were no significant differences in the mean daily colistin dose (218.2 ± 30.0 mg vs. 225.0 ± 56.0 mg, respectively p = 0.933), cumulative colistin dose (3063.6 ± 516.1 mg vs. 4,350.0 ± 2,631.5 mg, p = 0.466), or aerosol treatment period (14.4 ± 2.2 day vs. 16.0 ± 7.5 day, p = 0.805) between negative conversion and non-conversion patients.
The mean length of MICU stay was 30.5 ± 21.9 days, and the mean length of mechanical ventilation was 21.8 ± 14.8 days. Mortality during MICU stay was 24.0% (6/25), and the total in-hospital mortality was 40.0% (10/25). A number of factors, such as BMI < 18, concomitant use of aminoglycosides, and administration of amphotericin B with aerosolized colistin, were significantly different between survivors and non-survivors (Table 2). However, while aminoglycoside use was significantly related to mortality in univariate analysis, multivariate analysis revealed that only a BMI < 18 was a unique independent risk factor of mortality (odds ratio [OR] = 21.95, 95% confidence interval [CI] 1.59-302.23; p = 0.020) (Fig. 2). Notably, aerosolized colistin-associated AKI does not seem to have an effect on the mortality (OR = 0.33, 95% CI 0.06-1.75; p = 0.19) (Table 3).
Both the leukocyte count and PCT level decreased after treatment with aerosolized colistin, although these decreases were not significant. However, the level of hsCRP was significantly decreased after aerosolized colistin treatment (Table 4).
Eleven of the 25 patients (44.0%) who underwent aerosolized colistin treatment exhibited AKI according to the RIFLE criteria; one patient (9.1%) was in the risk group, six patients (54.5%) were in the injury group, and four patients (36.4%) were in the failure group. The eGFR based on CKD-EPI of the AKI group was nearly half that of the non-AKI group (45.9 ± 24.2 vs. 86.3 ± 45.3, respectively p = 0.01) (Table 5).
The cumulative dose and time interval from aerosolized colistin administration to the onset of AKI were 1,140.55 ± 722.57 mg and 4.72 ± 2.41 days, respectively. All patients with AKI received 24 hour continuous renal replacement therapy and continued aerosolized colistin treatment because there were no susceptible antibiotics in the sensitivity test. The surviving patients of 11 AKI cases demonstrated slowly normalized renal function over four weeks.
The demographics, comorbidities, daily and total cumulative doses of colistin, and concomitantly administered drugs with potential nephrotoxicity were compared between the AKI group and the non-AKI group. We found no significant differences in age, sex, body weight, or comorbidities (e.g., cardiovascular diseases, cerebrovascular diseases, diabetes, and COPD) between the groups. The length of MICU stay and APACHE II score also did not differ between the groups, but mortality in the MICU (14.3% vs. 36.3%, respectively p = 0.350) and total in-hospital stay (28.6% vs. 54.5%, respectively p = 0.074) tended to be higher in the AKI group. In addition, septic shock with vasopressor use (35.7% vs. 81.8%, respectively p = 0.02) and treatment with aminoglycosides concomitant with aerosolized colistin use (7.1% vs. 45.5%, respectively p = 0.03) were significantly higher in the AKI group. Conversely, vancomycin, amphotericin B, and diuretic use were not significantly different. Furthermore, there were no significant differences in the results of the daily mean dose of aerosolized colistin, mean total cumulative dose, or mean administration period between the AKI and non-AKI groups.
Multivariate analysis showed that septic shock was an independent risk factor of AKI (OR = 8.10, 95% CI 1.23-53.20; p = 0.04). The concomitant use of more than two nephrotoxic drugs administered during septic shock, excluding vasopressors, was also a significant predictor of AKI (p < 0.01) (Figs. 3, 4); this result was verified by the multivariate analysis (OR = 15.03, 95% CI 1.40-161.76; p = 0.025) (Table 6).
The highlight of the presents study was that negative conversion rate of inhaled colistin was more than 80% in patients with MDR-GNB VAP. And the incidence of AKI defined by RIFLE criteria was 44% of inhaled colistin treatment patients. Finally, septic shock and more than two nephrotoxic drugs combined with aerosolized colistin were independent risk factors of AKI.
The low tissue penetration rate and high nephrotoxicity of intravenous colistin treatment has led to its use as an aerosol treatment in patients with cystic fibrosis and VAP.[23] In this study, we observed an 84.6% bacterial negative conversion rate after aerosolized colistin treatment. Leukocyte count and PCT level decreased after treatment, but not significantly so. However, the level of hs-CRP measured after treatment was significantly lower than those observed before treatment.
Our study also offers insight into the tissue penetration and optimal dosage for aerosolized colistin. It is estimated that only 60% of each dose is inhaled into the bronchi, as some of the drug remains in the nebulizer. Therefore, a previous study recommended a dose of 150 mg colistin base activity three times per day (450 mg daily) for aerosol treatment.[24] However, nebulizer types and techniques, including ventilator settings, also affect aerosol distribution to the lungs.[24] In our study, a daily dosage of 300 mg colistin base activity was administered in 2-4 doses daily using an ultrasonic nebulizer with ventilator settings.[25] This was found to provide effective aerosol therapy, as evidenced by our high negative conversion rate.[17]
A previous study on aerosolized colistin reported a reduced incidence of nephrotoxicity for this mode of treatment compared to intravenous injection.[6] However, based on the RIFLE criteria, we observed AKI in 44% of our subjects (11 patients). This dissimilar incidence of colistin-related nephrotoxicity in the two studies could be resulted by the differences in the definitions of nephrotoxicity, dosing schedules, or duration of treatment. In other studies that have used the RIFLE criteria, colistin-related nephrotoxicity was observed in up to 49% of patients;[26] in another study, it was closely associated with the total dose and treatment duration of colistin.[11] However, in our study, the daily dose, cumulative dose, and period of administration did not differ between the AKI and non-AKI groups, and none of these were predictors of AKI. The dose and administration schedules of aerosolized colistin were repeatedly adjusted by renal function based on the drug package insert,[19,21] using eGFR[22] in the morning and evening and hourly urine output. As a result, colistin dose and administration intervals were adjusted very strictly. In addition, the administration route itself could be an important predictive factor of AKI. Inhalation is theoretically appealing, as aerosolized local delivery to the lungs should avoid, or at least minimize, potential renal toxicity compared to systemic administration (~45% vs. ~32%, respectively).[3,10]
Most patients in the MICU are treated with multiple medications at the same time. In our study, all patients in the AKI group were concomitantly administered more than one potentially nephrotoxic medication, and receiving more than two nephrotoxic medications was predictive of renal toxicity. Thus, the concomitant use of nephrotoxic medications seems to be more influential to the development of nephrotoxicity than monotherapy with aerosolized colistin. Therefore, renal function such as eGFR must be monitored regularly when colistin is administered with other drugs that have nephrotoxic potential.
Several factors such as BMI < 18 kg/m2 and concomitant administration of aminoglycosides or amphotericin B were correlated with significantly higher mortality (Table 2). The obesity is closely associated with many medical problems such as diabetes, cardiovascular and respiratory diseases, and cancers.[27] The impact of obesity on outcome in critically ill patients has not been conclusive yet, the mortality in obese and very obese patients with septic shock seems to be lower than normal weight patients.[28-30] This absurd phenomenon is called the “obesity paradox”.[30] In our study, only BMI < 18 kg/m2 was a unique independent risk factor of mortality in multivariate analysis (OR = 21.95, 95% CI 1.59-302.23; p = 0.020).
The use of aerosolized antibiotics can be accompanied by other adverse effects, including chest symptoms (e.g., cough, wheezing, and dyspnea) and pharyngolaryngitis. Because the aerosolized formulation of colistin has not yet been developed, the intravenous vial formulation of colistin is converted for aerosol use in clinical practice. Therefore, these intravenous vials contain preservatives, and some patients with asthma or sensitive bronchi have reported decreased forced expiratory volume and bronchospasm when this formulation was used for aerosol treatment.[31] In this study, a bronchodilator, such as a β2 adrenergic agonist and/or anticholinergic agent, was used 20 minutes before treatment to prevent bronchospasm. Nevertheless, one patient showed severe bronchospasm, followed by decreased blood pressure. This patient was thereafter given intravenous colistin instead of aerosolized treatment. No other colistin-related toxicities, such as neurotoxicity or muscular weakness, were observed.
Despite its merit and the plausible research question, this paper is flawed by its retrospective design and the small number of patients enrolled. Furthermore, the lack of a control group who did not receive aerosolized colistin limited the evaluation of the true effect. Finally, the concentrations of colistin in the bronchial and alveolar tissues and blood were not measured after treatment. In this study, 84.6% of MDR-GNB pneumonia patients showed negative conversion after aerosolized colistin treatment, and inflammation-associated hs-CRP level improved. Although 44% of patients exhibited signs of nephrotoxicity, this effect was likely due to concomitant treatment with other nephrotoxic medications, rather than colistin monotherapy. These limits clearly jeopardize the solidity of the final conclusions. Therefore, further studies on the optimal dose of colistin should be conducted to minimize nephrotoxicity and effectively treat patients with pulmonary infection.
In conclusion, adjunctive aerosolized colistin treatment might be an effective and relatively safe option to eradicate MDR-GNB in patients with pneumonia. However, renal function should be monitored regularly to reduce the incidence of aerosolized colistin-induced nephrotoxicity when colistin is administered concomitantly with potentially nephrotoxic drugs.
ACKNOWLEDGMENTS
This manuscript was supported by the Biomedical Research Institute Fund, Chonbuk National University Hospital. The authors would like to thank Amanda Weiskoff, Ph.D. for providing medical writing services.
References
2. Falagas ME, Kasiakou SK. Colistin: the revival of polymyxins for the management of multidrug-resistant gram-negative bacterial infections. Clin Infect Dis. 2005; 40:1333–41.

3. Falagas ME, Kasiakou SK. Toxicity of polymyxins: a systematic review of the evidence from old and recent studies. Crit Care. 2006; 10:R27.
4. Pogue JM, Lee J, Marchaim D, Yee V, Zhao JJ, Chopra T, et al. Incidence of and risk factors for colistinassociated nephrotoxicity in a large academic health system. Clin Infect Dis. 2011; 53:879–84.

5. Hartzell JD, Neff R, Ake J, Howard R, Olson S, Paolino K, et al. Nephrotoxicity associated with intravenous colistin (colistimethate sodium) treatment at a tertiary care medical center. Clin Infect Dis. 2009; 48:1724–8.

6. Yapa SWS, Li J, Patel K, Wilson JW, Dooley MJ, George J, et al. Pulmonary and systemic pharmacokinetics of inhaled and intravenous colistin methanesulfonate in cystic fibrosis patients: targeting advantage of inhalational administration. Antimicrob Agents Chemother. 2014; 58:2570–9.
7. Jensen T, Pedersen SS, Garne S, Heilmann C, Høiby N, Koch C. Colistin inhalation therapy in cystic fibrosis patients with chronic Pseudomonas aeruginosa lung infection. J Antimicrob Chemother. 1987; 19:831–8.
8. Kollef MH, Niederman MS. Why is Acinetobacter baumannii a problem for critically ill patients? Intensive Care Med. 2015; 41:2170–2.
9. Bogović TZ, Budimir A, Bošnjak Z, Hrabač P, Baronica R, Tomašević B, et al. Inhalation plus intravenous colistin versus intravenous colistin alone for treatment of ventilator associated pneumonia. Signa Vitae. 2014; 9(Suppl 1):29–33.
11. Pogue JM, Lee J, Marchaim D, Yee V, Zhao JJ, Chopra T, et al. Incidence of and risk factors for colistin-associated nephrotoxicity in a large academic health system. Clin Infect Dis. 2011; 53:879–84.

12. Kaye KS, Kaye D. Polymyxins (polymyxin B and colistin). Principles and practice of infectious diseases. 6th ed. In : Mandell GL, Bennett JE, Dolin R, editors. Philadelphia: Elsevier;2005. p. 435–6.
13. Horan TC, Andrus M, Dudeck MA. CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control. 2008; 36:309–32.

14. Korbila IP, Michalopoulos A, Rafailidis PI, Nikita D, Samonis G, Falagas ME. Inhaled colistin as adjunctive to intravenous colistin for the treatment of microbiologically documented VAP: a comparative cohort study. Clin Microbiol Infect. 2010; 16:1230–6.
15. Knaus W, Draper E, Wagner D, Zimmerman J. APACHE II: a severity of disease classification system. Crit Care Med. 1985; 13:818–29.
16. Pugin J, Auckenthaler R, Mili N, Janssens JP, Lew PD, Suter PM. Diagnosis of ventilator-associated pneumonia by bacteriologic analysis of bronchoscopic and nonbronchoscopic “blind” bronchoalveolar lavage fluid. Am Rev Respir Dis. 1991; 143:1121–9.

17. Kofteridis DP, Alexopoulou C, Valachis A, Maraki S, Dimopoulou D, Georgopoulos D, Samonis G. Aerosolized plus intravenous colistin versus intravenous colistin alone for the treatment of ventilatorassociated pneumonia: a matched case-control study. Clinical Infect Dis. 2010; 51:1238–44.

18. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF, Feldman HI, et al. A New Equation to Estimate Glomerular Filtration Rate. Ann Intern Med. 2009; 150:604–12.

19. Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P; Acute Dialysis Quality Initiative work-group. Acute renal failure - definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care. 2004; 8:R204–12.
20. Hamer DH. Treatment of nosocomial pneumonia and tracheobronchitis caused by multidrug-resistant Pseudomonas aeruginosa with aerosolized colistin. Am J Respir Crit Care Med. 2000; 162:328–30.
21. Evans ME, Feola DJ, Rapp RP. Polymyxin B sulfate and colistin: old antibi-otics for emerging multiresistant gram-negative bacteria. Ann Pharmacother. 1999; 33:960–7.

22. DeRyke CA, Crawford AJ, Uddin N, Wallace MR. Colistin dosing and nephrotoxicity in a large community teaching hospital. Antimicrob Agents Chemother. 2010; 54:4503–5.

23. Imberti R, Cusato M, Villani P, Carnevale L, Iotti GA, Langer M, et al. Steady-state pharmacokinetics and BAL concentration of colistin in critically Ill patients after IV colistin methanesulfonate administration. Chest. 2010; 138:1333–9.

24. Steinfort DP, Steinfort C. Effect of long-term nebulized colistin on lung function and quality of life in patients with chronic bronchial sepsis. Intern Med J. 2007; 37:495–8.

25. Marchand S, Bouchene S, de Monte M, Guilleminault L, Montharu J, Cabrera M, et al. Pharmacokinetics of Colistin Methansulphonate (CMS) and Colistin after CMS Nebulisation in Baboon Monkeys. Pharm Res. 2015; 32:3404–14.

26. Sorlí L, Luque S, Grau S, Berenguer N, Segura C, Montero MM, et al. Trough colistin plasma level is an independent risk factor for nephrotoxicity: a prospective observational cohort study. BMC Infect Dis. 2013; 13:380–8.

27. Katzmarzyk PT, Reeder BA, Elliott S, Joffres MR, Pahwa P, Raine KD, et al. Body mass index and risk of cardiovascular disease, cancer and all-cause mortality. Can J Public Health. 2012; 103:147–51.

28. O’Brien JM Jr, Welsh CH, Fish RH, Ancukiewicz M, Kramer AM. Excess body weight is not independently associated with outcome in mechanically ventilated patients with acute lung injury. Ann Intern Med. 2004; 140:338–45.

29. Hogue CW Jr, Stearns JD, Colantuoni E, Robinson KA, Stierer T, Mitter N, et al. The impact of obesity on outcomes after critical illness: a meta-analysis. Intensive Care Med. 2009; 35:1152–70.

30. Arabi YM, Dara SI, Tamim HM, Rishu AH, Bouchama A, et al. The Cooperative Antimicrobial Therapy of Septic Shock (CATSS) Data-base Research Group. Clinical characteristics, sepsis interventions and outcomes in the obese patients with septic shock: an international multicenter cohort study. Crit Care. 2013; 17:R72.
Fig. 1.
Flow of patient screening and enrollment. MDR-GNB: multidrug-resistant Gram-negative bacteria; RRT: renal replacement therapy; AKI: acute kidney injury.
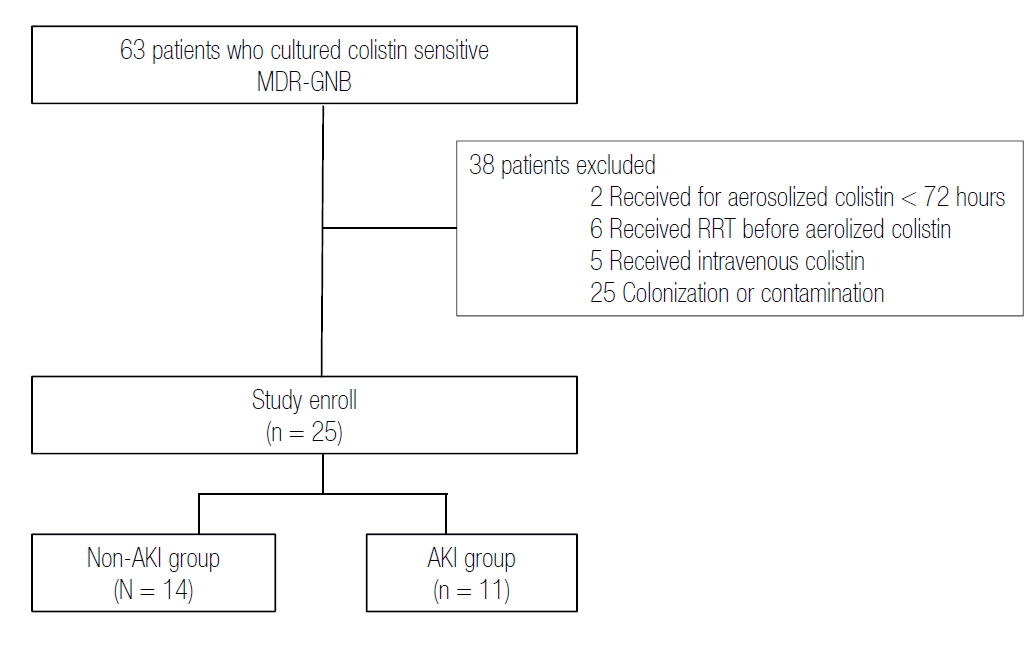
Fig. 2.
The area under a ROC curve to predict factors associated with mortality and AKI. For mortality, the area under the ROC curve was 0.817 for patients with a body mass index <18 kg/m2 (p = 0.015). ROC: receiver operating characteristic; AKI: acute kidney injury.
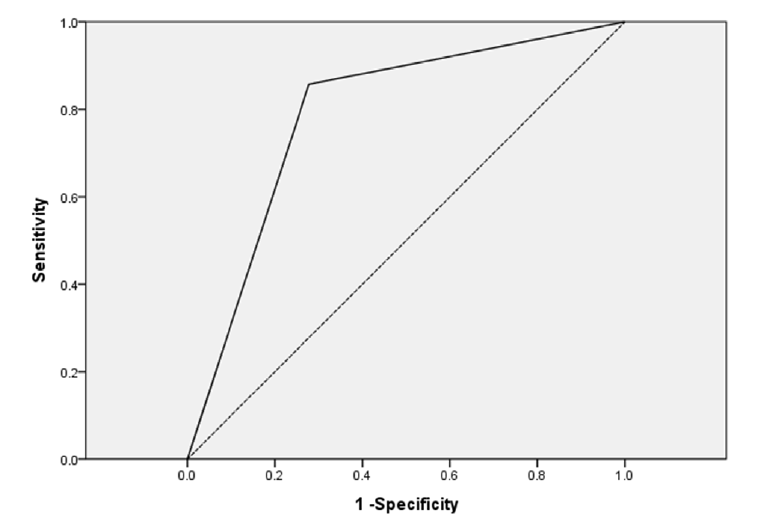
Fig. 3.
Relationship between the occurrence of AKI and concomitant use of nephrotoxic drugs, excluding vasopressors. Use of more than two nephrotoxic drugs concomitant with aerosolized colistin therapy increases the likelihood of developing AKI. AKI: acute kidney injury.
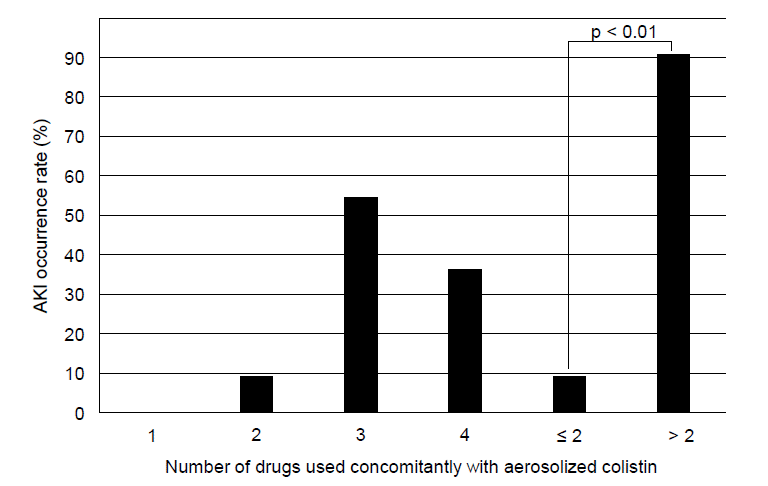
Fig. 4.
The area under a ROC curve to predict factors associated with mortality and AKI. For AKI, the area under the ROC curve was 0.790 for patients who used more than two nephrotoxic drugs concomitant with aerosolized colistin (p = 0.027). ROC: receiver operating characteristic; AKI: acute kidney injury.
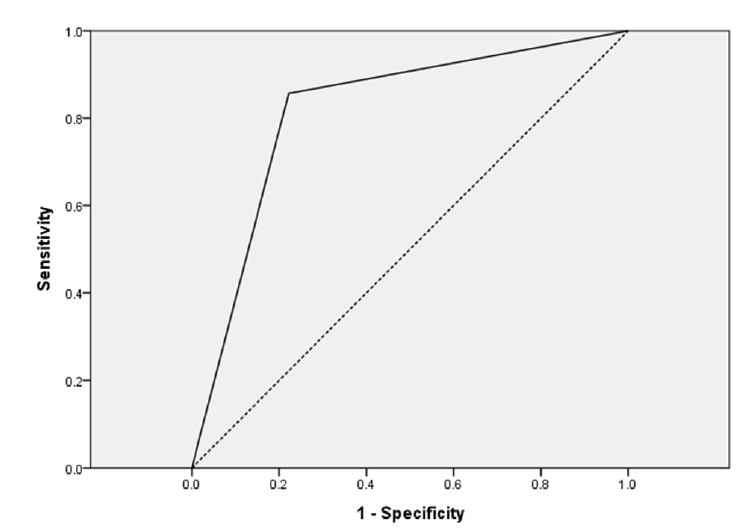
Table 1.
Baseline characteristics of patients
| Characteristics | Mean ± SD or n (%) |
|---|---|
| Demographics | |
| Age (years) | 66.6 ± 14.5 |
| Male | 16 (64.0) |
| Body weight (kg) | 56.1 ± 10.5 |
| Body Mass Index (kg/m2) | 20.4 ± 4.1 |
| Comorbidities | |
| Cardiovascular diseases | 13 (52.0) |
| Cerebrovascular diseases | 4 (16.0) |
| Diabetes mellitus | 3 (12.0) |
| Chronic obstructive pulmonary disease | 3 (12.0) |
| Length of MICU stay, days | 30.5 ± 21.9 |
| Mechanical ventilation, days | 21.8 ± 14.8 |
| APACHE II score* | 23.0 ± 6.8 |
| CPIS† | 7.9 ± 1.8 |
| PaO2/FiO2 ratio | 250.1 ± 9.35 |
| Isolated pathogens | |
| Acinetobacter baumannii | 22 (88.0) |
| Pseudomonas aeruginosa | 6 (24.0) |
| Klebsiella pneumoniae | 2 (8.0) |
| eGFR by CKD-EPI‡ | 135.6 ± 97.9 |
| Colistin | |
| Daily dose (mg) | 246.6 ± 141.4 |
| Cumulative dose (mg) | 2,714.0 ± 1,799.0 |
| Duration of administration (days) | 11.7 ± 7.1 |
Table 2.
Clinical parameters in survivors and non-survivors
| Clinical parameters | Survivors (n = 15) | Non-Survivors (n = 10) | p-value |
|---|---|---|---|
| Demographics | |||
| Age (years) | 66.7 ± 13.7 | 66.5 ± 16.4 | 0.97 |
| Male sex, n (%) | 8 (53.3) | 8 (80) | 0.67 |
| Body weight (kg) | 56.1 ± 11.0 | 56.1 ± 10.3 | 1.00 |
| Body Mass Index (kg/m2) | 22.1 ± 3.7 | 17.8 ± 3.1 | 0.006 |
| Comorbidities, n (%) | |||
| Cardiovascular diseases | 6 (40.0) | 7 (70.0) | 0.15 |
| Cerebrovascular diseases | 2 (13.3) | 2 (20.0) | 0.66 |
| Diabetes mellitus | 2 (13.3) | 1 (10.0) | 0.80 |
| COPD | 2 (13.3) | 1 (10.0) | 0.80 |
| Length of MICU stay (days) | 30.0 ± 27.0 | 30.5 ± 21.9 | 0.91 |
| Mechanical ventilation (days) | 20.1 ± 24.4 | 21.8 ± 14.8 | 0.48 |
| APACHE II score* | 21.7 ± 7.5 | 25.0 ± 5.2 | 0.24 |
| CPIS† | 8.5 ± 1.6 | 7.5 ± 1.8 | 0.16 |
| PaO2/FiO2 ratio | 279.1 ± 103.8 | 207.6 ± 56.1 | 0.06 |
| Septic shock | 7 (46.7) | 7 (70.0) | 0.25 |
| eGFR by CKD-EPI‡ | 92.0 ± 39.5 | 91.9 ± 35.9 | 0.99 |
| Acute Kidney Injury, n (%) | 5 (33.3) | 6 (60.0) | 0.24 |
| Colistin | |||
| Daily dose (mg) | 238.5 ± 82.0 | 270.0 ± 63.2 | 0.31 |
| Cumulative dose (mg) | 2,793.3 ± 2,120.5 | 2,595.0 ± 1,268.9 | 0.79 |
| Duration of treatment (days) | 12.0 ± 6.9 | 11.2 ± 7.6 | 0.79 |
| Concomitant nephrotoxic drugs, n (%) | |||
| Vancomycin | 10 (66.7) | 4 (40.0) | 0.19 |
| Aminoglycoside | 1 (6.7) | 5 (50.0) | 0.01 |
| Amphotericin B | 0 (0) | 3 (30.0) | 0.02 |
| Diuretics | 14 (93.3) | 9 (90.0) | 0.76 |
| Vasopressors | 7 (46.7) | 7 (70.0) | 0.25 |
Table 3.
Univariate and multivariate analyses of risk factors for mortality in patients with MDR-GNB pneumonia treated with aerosolized colistin
| Variables |
Univariate analyses |
Multivariate analyses |
||
|---|---|---|---|---|
| OR (95% CI) | p-value | OR (95% CI) | p-value | |
| Demographics | ||||
| Age > 65 years | 1.18 (0.2-6.93) | 0.85 | ||
| Male sex | 1.56 (0.28-8.53) | 0.61 | ||
| BMI < 18 | 21.00 (1.92-229.40) | 0.01 | 21.95 (1.59-302.23) | 0.02 |
| APACHE II* > 20 | 7.87 (0.79-78.68) | 0.08 | 0.92 (0.78-1.08) | 0.33 |
| PF ratio† < 300 | 7.87 (0.79-78.68) | 0.08 | 0.62 (0.98-3.98) | 0.62 |
| Septic shock | 2.67 (0.49-14.16) | 0.25 | ||
| AKI‡ | 0.33 (0.06-1.75) | 0.19 | ||
| Colistin | ||||
| Daily dose | 2.00 (0.28-14.18) | 0.49 | ||
| Total dose | 1.00 (0.99-1.00) | 0.78 | ||
| Duration | 0.98 (0.87-1.10) | 0.78 | ||
| Comorbidities | ||||
| CV disease | 0.29 (0.05-1.55) | 0.29 | ||
| Diabetes§ | 1.39 (0.11-17.67) | 0.80 | ||
| Concomitant nephrotoxic drugs | ||||
| Vancomycin | 3.00 (0.57-15.77) | 0.19 | ||
| Aminoglycoside | 14.00 (1.29-150.89) | 0.03 | 14.78 (0.98-222.86) | 0.052 |
| Amphotericin B | 0.00 (0.00-0.00) | 0.99 | ||
| Nephrotoxic drugs∥ > 2 | 2.67 (0.49-14.46) | 0.25 | ||
Table 4.
Changes in laboratory values of inflammatory markers with aerosolized colistin treatment
Table 5.
Clinical characteristics in patients with and without AKI
| Compare characteristics | Non-AKI group (n = 14) | AKI group (n = 11) | p-value |
|---|---|---|---|
| Demographics | |||
| Age (years) | 64.6 ± 14.8 | 69.2 ± 14.5 | 0.44 |
| Male sex, n (%) | 8 (57.1) | 8 (72.7) | 0.68 |
| Body weight (kg) | 56.1 ± 11.0 | 56.1 ± 10.3 | 1.00 |
| Body Mass Index (kg/m2) | 20.6 ± 3.9 | 20.2 ± 4.4 | 0.79 |
| Comorbidity, n (%) | |||
| Cardiovascular diseases | 7 (50.0) | 6 (54.6) | 0.82 |
| Cerebrovascular diseases | 3 (21.4) | 1 (9.1) | 0.40 |
| Diabetes mellitus | 2 (14.4) | 1 (9.1) | 0.69 |
| COPD | 3 (21.4) | 0 (0.0) | 0.10 |
| Length of MICU stay (days) | 32.8 ± 26.6 | 27.5 ± 14.7 | 0.57 |
| Mechanical ventilation (days) | 22.5 ± 13.7 | 20.9 ± 16.7 | 0.80 |
| MICU mortality, n (%) | 2 (14.3) | 4 (36.3) | 0.35 |
| Total in-hospital mortality, n (%) | 4 (28.6) | 6 (54.5) | 0.07 |
| APACHE II score* | 20.9 ± 7.0 | 25.7 ± 5.5 | 0.07 |
| CPIS† | 7.3 ± 1.5 | 8.6 ± 1.8 | 0.06 |
| PaO2/FiO2 ratio | 266.0 ± 101.3 | 230.1 ± 83.4 | 0.36 |
| Septic shock | 5 (35.7) | 9 (81.8) | 0.02 |
| eGFR by CKD-EPI‡ | 86.3 ± 45.3 | 45.9± 24.2 | 0.01 |
| Colistin | |||
| Daily dose (mg) | 253.6 ± 77.1 | 250.0 ± 75.0 | 0.91 |
| Cumulative dose (mg) | 2,993 ± 2,186 | 2,356 ± 1,144 | 0.39 |
| Duration of treatment (days) | 11.6 ± 6.7 | 11.8 ± 7.9 | 0.93 |
| Concomitant nephrotoxic drugs, n (%) | |||
| Vancomycin | 6 (42.9) | 8 (72.7) | 0.13 |
| Aminoglycoside | 1 (7.1) | 5 (45.5) | 0.03 |
| Amphotericin B | 1 (7.1) | 2 (18.2) | 0.40 |
| Diuretics | 12 (85.7) | 11 (100.0) | 0.19 |
| Vasopressors | 5 (35.7) | 9 (81.8) | 0.02 |
Table 6.
Univariate and multivariate analyses of risk factors for AKI in patients with MDR-GNB pneumonia treated with aerosolized colistin
| Variables |
Univariate analyses |
Multivariate analyses |
||
|---|---|---|---|---|
| OR (95% CI) | p-value | OR (95% CI) | p-value | |
| Demographics | ||||
| Age > 65 years | 2.50 (0.38-16.42) | 0.34 | ||
| Male sex | 2.00 (0.37-10.91) | 0.42 | ||
| BMI < 18 | 2.08 (0.40-10.95) | 0.39 | ||
| APACHE II* > 20 | 3.37 (0.52-21.72) | 0.20 | ||
| PF ratio† < 300 | 3.37 (0.52-21.72) | 0.20 | ||
| Septic shock | 12.88 (1.01-150.76) | 0.029 | 8.10 (1.23-53.20) | 0.04 |
| Colistin | ||||
| Daily dose | 0.99 (0.98-1.00) | 0.11 | ||
| Total dose | 1.00 (0.99-1.00) | 0.38 | ||
| Duration | 1.00 (0.89-1.13) | 0.93 | ||
| Comorbidities | ||||
| CV disease‡ | 1.20 (0.25-5.84) | 0.82 | ||
| Diabetes§ | 0.60 (0.47-7.63) | 0.69 | ||
| Concomitant nephrotoxic drugs | ||||
| Vancomycin | 3.56 (0.65-19.41) | 0.14 | ||
| Aminoglycoside | 10.83 (1.03-114.15) | 0.047 | 5.61 (0.42-76.07) | 0.19 |
| Amphotericin B | 2.89 (0.23-36.86) | 0.41 | ||
| Nephrotoxic drugs∥ > 2 | 57.59 (3.30-1003.58) | 0.005 | 15.03 (1.40-161.76) | 0.025 |




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download