Abstract
This report describes a case of systemic thromboembolism caused by left ventricular (LV) thrombosis that developed after placement of an implantable cardioverter-defibrillator (ICD). A 27-year-old male patient was diagnosed with idiopathic dilated cardiomyopathy and ventricular tachycardia, and underwent ICD implantation for the primary prevention of sudden cardiac death. Two weeks after ICD implantation, the patient experienced renal infarction. Transthoracic echocardiography revealed a mobile thrombus at the LV apex, and automated function imaging demonstrated deteriorated LV function after ICD implantation. The RV was not placed by ICD and the mechanical force which was occurred by ICD that led to induced dyssynchronous motion of the LV apex may have resulted in a systemic thromboembolism.
The incidence of thromboembolic events after implantation of intra-cardiac devices is 0.6–3.5%.[1] Right ventricular (RV) pacing can induce dyssynchronous contraction of the left ventricle (LV) and LV dysfunction.[2,3] Abnormal LV apical wall motion can result in thrombosis in LV cavity and systemic thromboembolism.[4,5] Most of thromboembolic events after implantation of implantable cardioverter-defibrillator (ICD) are venous thrombosis and arterial thromboembolic events are very rare[6] and thrombosis associated with pacemaker or ICD result from lead thrombosis.[7,8] This report demonstrates a systemic thromboembolism caused by thrombosis of the LV apex in a patient with dilated cardiomyopathy (DCMP) and ventricular tachycardia (VT). The event may be resulted from mechanical force of RV lead of an ICD that did not pace the RV apex.
A 27-year-old man was admitted to the hospital with shortness of breath and near syncope. He had experienced dyspnea, decreased exercise tolerance and dizziness for one month. His mother had died of sudden cardiac death at age 38. A chest X-ray showed mild cardiomegaly (Fig.1a). Laboratory data including complete blood cell count and blood chemistry were unremarkable. Mild elevation of N-terminal pro-brain natriuretic peptide (1344 pg/ml) was noted and cardiac enzymes such as troponin-I, CK-MB were within normal limits. Electrocardiography (ECG) demonstrated normal sinus rhythm and LV hypertrophy. Non-sustained VT was detected by 24-hour ambulatory holter monitoring (Fig.1b). We performed transthoracic echocardiography (TTE) to evaluate cardiac function. The LV end diastolic dimension was 62.77 mm (Fig. 2a), and LV ejection fraction (LVEF) measured by modified Simpson’s method was 32.7%. Global hypokinesis was noted (Fig. 2b). Coronary CT angiography demonstrated no significant stenosis. The patient was diagnosed with DCMP and treated with ramipril 5 mg, carvedilol 6.25 mg, furosemide 40 mg, and spironolactone 25 mg.
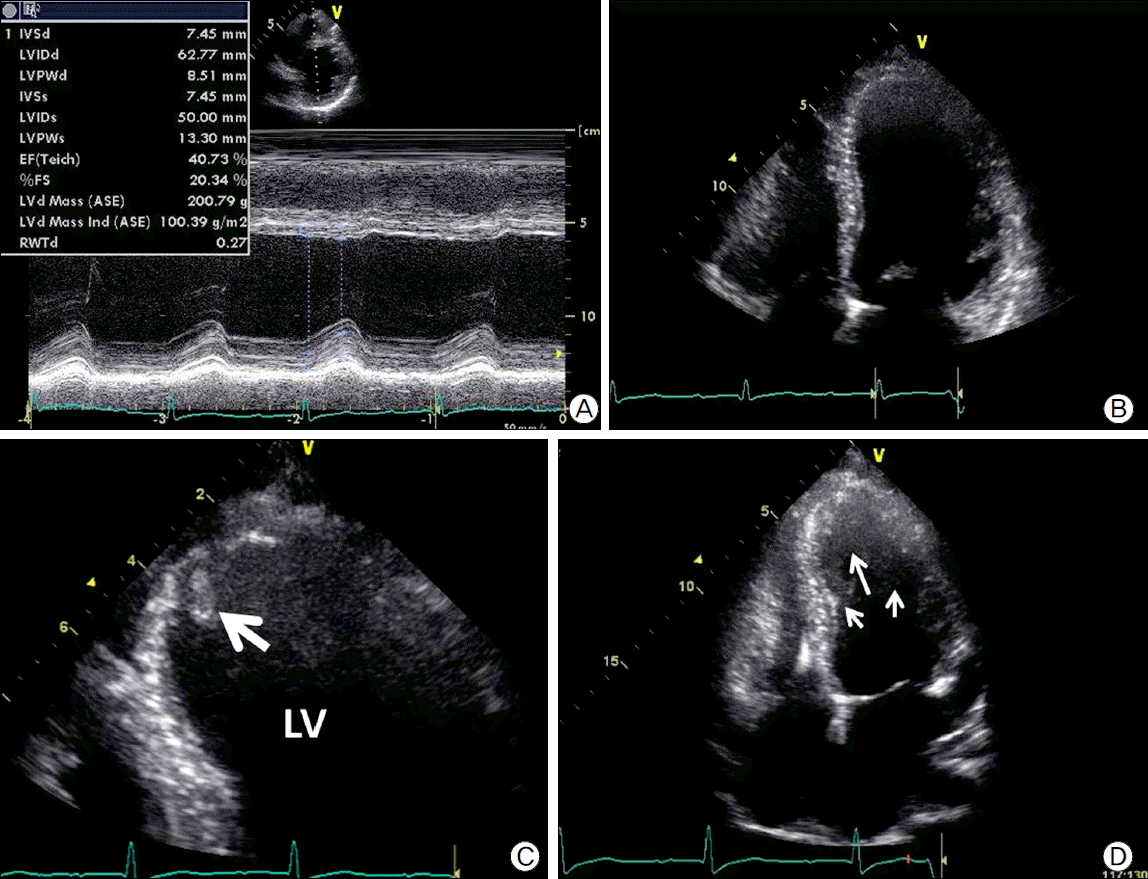
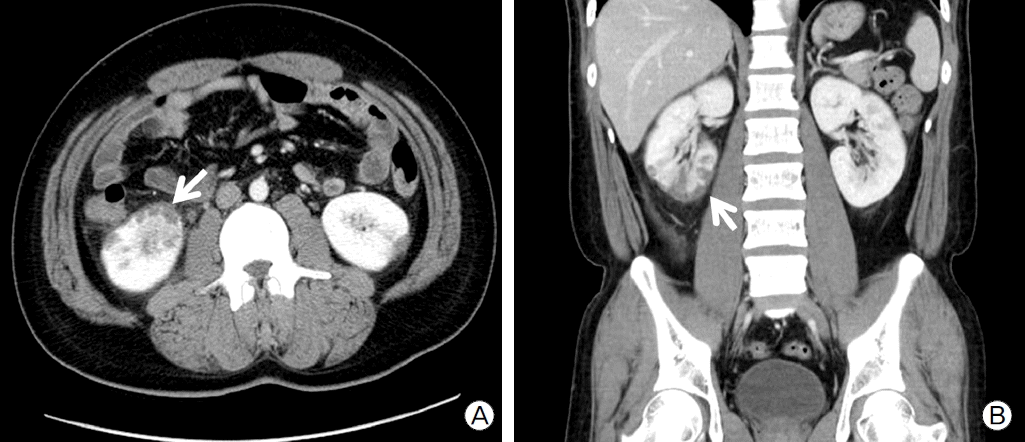
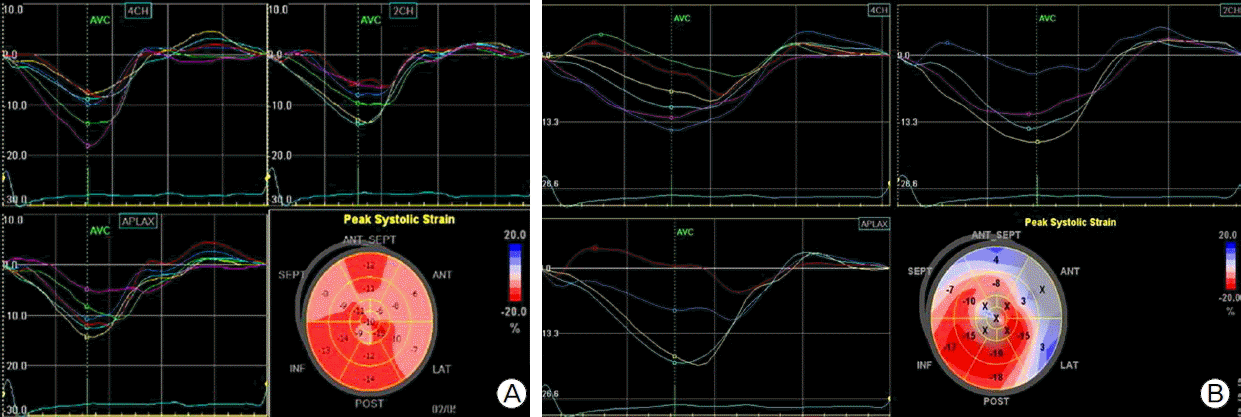
Considering the severe LV dysfunction, documented non-sustained VT, and family history, we decided to implant an ICD (Guidant VITALITYTM, Boston Scientific, Natick, MA, USA) for primary prevention of sudden cardiac death. The RV lead (ENDOTAK RELIANCEⓡ, Boston Scientific) was placed at RV apex (Fig. 1c). At 13 days after successful ICD implantation, the patient complained of sudden right flank pain. Laboratory findings showed leukocytosis, microscopic hematuria, elevated high-sensitivity C-reactive protein (60.38 mg/L). There was no elevation of cardiac enzymes and ECG showed no interval changes compared with prior ECG (Fig. 1d). Abdominal computed tomography showed multiple renal infarctions (Fig. 3a, Fig. 3b). TTE showed a mobile thrombus in the LV apex (Fig. 2c) and spontaneous echo contrast (SEC) in the LV apical cavity (Fig. 2d) which was not observed before ICD implantation. LVEF was decline to 27% and mid to apical anterior septal wall motion was akinetic, which was hypokinetic before ICD implantation. ICD setting for ventricular pacing was VVI mode and lower rate limit was 45 per minute and 75 per minute in post-shock bradycardia. We checked ICD and there was no ventricular tachycardia or fibrillation or pacing event. There is no significant change in medicine he has been taking (ramipril 5 mg, carvedilol 6.25 mg, furosemide 40 mg, and spironolactone 25 mg). We analyzed LV function by measuring myocardial deformation with automated function imaging (AFI) using post assessment program (EchoPac, General electric, USA) and AFI demonstrated deteriorated LV function during normal sinus rhythm after ICD implantation compared to prior study (Fig. 4a, Fig. 4b). We started the patient on anticoagulation therapy with warfarin and two weeks later, the thrombus was resolved.
An ICD lead is thicker and stiffer than a pacemaker lead. ICD leads can generate more mechanical force than pacemaker leads and have a higher incidence of lead perforation.[9] However, we did not expect that the mechanical force of the RV lead alone would affect LV contractility, resulting in dyssynchronous wall motion of the LV apex. In the case, contractility of the LV apex deteriorated after an RV lead of ICD was placed in the RV apex. Deterioration of LV contractility was demonstrated by using AFI, which is simplified method for assessment of myocardial strain throughout the LV.[10] LV apical cavity after ICD implantation was clearly in the thrombogenic condition, which was demonstrated by SEC that was not observed before the procedure. The Force, that deteriorated LV wall motion, might be affected by the length of intracardiac portion of the RV lead which can be determined by the position of lead tip and the bent portion of the lead in the right atrium. An excessively long intra-cardiac lead can generate mechanical force and compress the RV apex, affecting LV contractility. The diseased myocardium of DCMP patient could be a major contributor to dyssynchronus contraction of LV. In this case, the force generated by the RV lead might not be unusually high, but a diseased myocardium might not be able to overcome the tension generated by RV lead.
Although RV pacing induced LV dysfunction is well known complication, thrombosis in LV cavity is very rare in such patients. That is because myocardial contractility is generally preserved in patient with rhythm disorder. The dyssynchronous contraction of LV may reduce stroke volume and cardiac output, but is not enough for thrombogenesis. Severe LV dysfunction of patient in the case is a predisposing condition of intraventricular thrombosis and the incidence of LV thrombosis in patients with idiopathic DCMP ranges 11–44%.[11] In this case, 13 days is sufficient time for thrombogenesis even though there was no thrombus before procedure and inflammatory conditions after surgical procedure of ICD implantation may also predispose LV thrombosis. But, inflammatory markers were stable during peri-procedural period and leukocytosis and elevation of hsCRP was associated with renal infarction. Moreover, there was no elevation of pro-BNP. It is the problem of deterioration of contractility of the regional LV segment rather than decreased LV ejection fraction. Ventricular pacing burden may also affect the LV dysfunction and eventually LV thrombogenesis. In patients with diseased LV myocardium, such as DCMP or ischemic cardiomyopathy, it is possible that RV pacing induced LV dyssynchrony may result in thrombosis in LV cavity. But there was no pacing event in this case during 13 days before embolic event. There was no SEC in LV apex before ICD implantation and the LV myocardial strain measurement by AFI clearly demonstrated deterioration of LV contractility after ICD implantation which suggests ICD implantation influenced to LV contractility. We presume RV lead inserted in right side heart generated mechanical force and affected contractility of diseased myocardium of DCMP and result in thrombosis and embolic event.
In conclusion, LV thrombosis and systemic embolization after implantation of ICD has not been described previously in the literature. The mechanical force generated by an RV lead of ICD can influence LV contractility in a patient with DMCP with severe LV dysfunction who already has diseased myocardium. Therefore, proper monitoring for deterioration of LV wall motion and prophylactic anticoagulation should be considered after ICD implantation.
REFERENCES
1). Bailey SM, Wilkoff BL. Complications of pacemakers and defibrillators in the elderly. Am J Geriatr Cardiol. 2006; 15:102–7.

2). Pastore G, Noventa F, Piovesana P, Cazzin R, Aggio S, Verlato R, et al. Left ventricular dyssynchrony resulting from right ventricular apical pacing: relevance of baseline assessment. Pacing Clin Electrophysiol. 2008; 31:1456–62.

3). Te CC, Stavrakis S, Lozano P, Reynolds D. Apparent acute reversible right ventricular pacing-induced left ventricular dysfunction. J Cardiovasc Electrophysiol. 2013; 24:224–6.

4). Delewi R, Zijlstra F, Piek JJ. Left ventricular thrombus formation after acute myocardial infarction. Heart. 2012; 98:1743–9.

5). Shin SN, Yun KH, Ko JS, Rhee SJ, Yoo NJ, Kim NH, et al. Left ventricular thrombus associated with takotsubo cardiomyopathy: a cardioembolic cause of cerebral infarction. J Cardiovasc Ultrasound. 2011; 19:152–5.

6). Grimm W, Flores BF, Marchlinski FE. Complications of implantable cardioverter defibrillator therapy: follow-up of 241 patients. Pacing Clin Electrophysiol. 1993; 16:218–22.

7). Rozmus G, Daubert JP, Huang DT, Rosero S, Hall B, Francis C. Venous thrombosis and stenosis after implantation of pacemakers and defibrillators. J Interv Card Electrophysiol. 2005; 13:9–19.

8). Refaat M, Bermudez C, Reis S. Right atrial thrombus attached to the defibrillator lead. Nat Clin Pract Cardiovasc Med. 2008; 5:354.

9). Carlson MD, Freedman RA, Levine PA. Lead perforation: incidence in registries. Pacing Clin Electrophysiol. 2008; 31:13–5.

10). Belghitia H, Brette S, Lafitte S, Reant P, Picard F, Serri K, et al. Automated function imaging: a new operator-independent strain method for assessing left ventricular function. Arch Cardiovasc Dis. 2008; 101:163–9.
11). Sivri N, Yetkin E, Tekin GO, Yalta K, Waltenberger J. Anticoagulation in Patients With Left Ventricular Systolic Dysfunction and Sinus Rhythm: When? Clin Appl Thromb Hemost. 2013; Apr. 23. [Epub]. DOI: 10.1177/1076029613486017.
Fig. 1.
(A) Chest X-ray showing cardiomegaly. (B) 24-hour holter monitoring demonstrating ventricular tachycardia. (C) Chest X-ray showing implanted implantable cardioverter-defibrillator (ICD). (D) Electrocardiography after development of embolic event showing normal sinus rhythm.
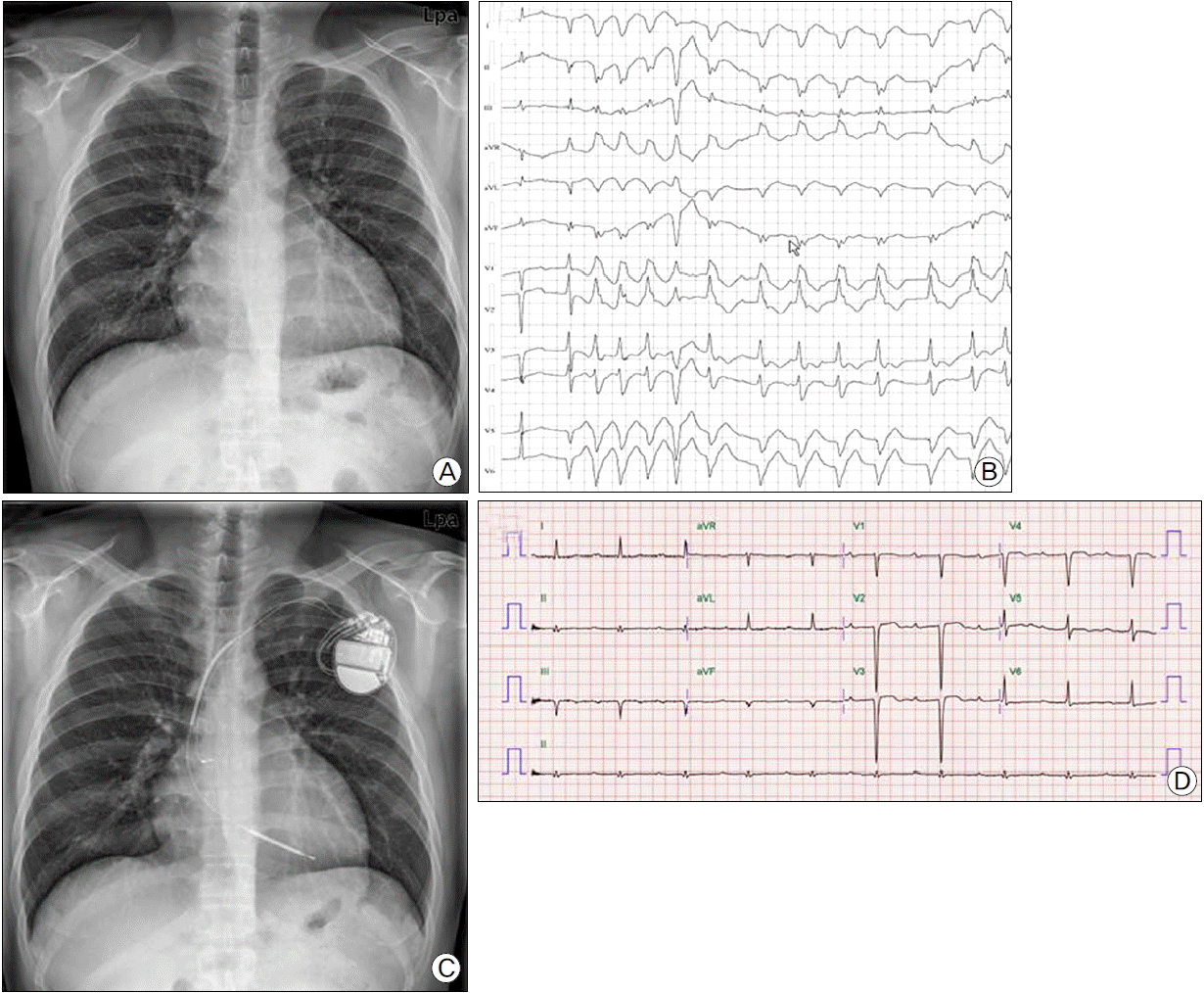
Fig. 2.
(A) M-mode image of tansthoracic echocardiography (TTE) demonstrating decreased contractility of the left ventricle and dilated chamber size. (B) Apical 4-chamber view of TTE demonstrating lack of thrombus on the apical wall.(C) Magnified image of apex showing mobile thrombus (white arrow). (D)Transthoracic echocardiography demonstratingright ventricular lead and spontaneous echo contrastin LV cavity (white arrows).




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download