Introduction
Materials and Methods
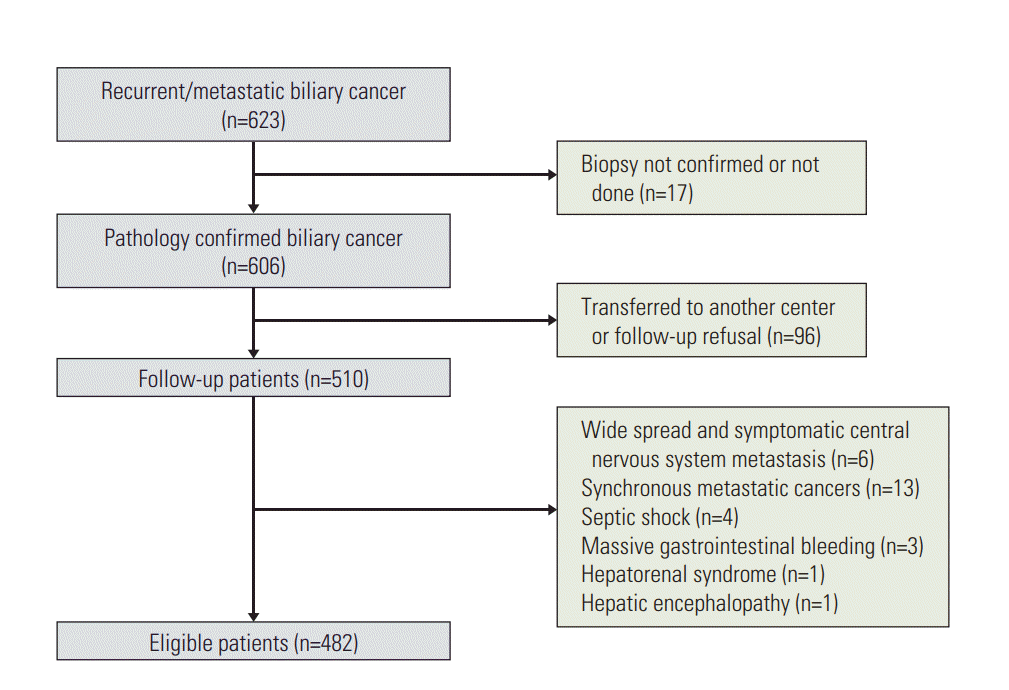
1. Patients
2. Data collection
3. Dataset allocation
4. Statistical analysis
Results
1. Baseline characteristics of investigational and validation datasets
Table 1.
| Characteristic | Total (n=482) | Investigation dataset (n=340) | Validation dataset (n=142) | p-value |
|---|---|---|---|---|
| Age (yr) | ||||
| < 67 | 235 (48.8) | 163 (47.9) | 72 (50.7) | 0.58 |
| ≥ 67 | 247 (51.2) | 177 (52.1) | 70 (49.3) | |
| Sex | ||||
| Male | 281 (58.3) | 201 (59.1) | 80 (56.3) | 0.573 |
| Female | 201 (41.7) | 139 (40.9) | 62 (43.7) | |
| Disease status | ||||
| Recurrent | 158 (32.8) | 117 (34.4) | 41 (28.9) | 0.238 |
| Metastatic | 324 (67.2) | 223 (65.6) | 101 (71.1) | |
| ECOG | ||||
| 0-2 | 418 (86.7) | 296 (87.1) | 122 (85.9) | 0.736 |
| 3-4 | 64 (13.3) | 44 (12.9) | 20 (14.1) | |
| Primary site | ||||
| Intrahepatic CC | 123 (25.5) | 87 (25.6) | 36 (25.4) | 1.00 |
| Hilar CC | 79 (16.4) | 56 (16.5) | 23 (16.2) | |
| Non-hilar CC | 87 (18.0) | 61 (17.9) | 26 (18.3) | |
| Gallbladder | 146 (30.3) | 103 (30.3) | 43 (30.3) | |
| Ampullary | 47 (9.8) | 33 (9.7) | 14 (9.9) | |
| BMI | 22.8 (20.6-25.0) | 22.7 (20.4-24.9) | 23.2 (21.0-25.1) | 0.223 |
| Complete blood count | ||||
| WBC count (/μL) | 7,090 (5,525-9,615) | 7,070 (5,555-9,650) | 7,130 (5,328-9,563) | 0.881 |
| Hemoglobin (g/dL) | 12.2 (11.1-13.4) | 12.1 (11.1-13.4) | 12.3 (11.1-13.4) | 0.771 |
| Platelet (×103/μL) | 243 (189-319) | 241 (186-314) | 250 (200-326) | 0.398 |
| NLR | 3.2 (2.0-5.8) | 3.3 (2.0-5.7) | 3.2 (2.1-6.1) | 0.738 |
| PLR | 171 (122-251) | 165 (120-234) | 181 (123-275) | |
| Blood chemistry profile | ||||
| Protein (g/dL) | 6.9 (6.5-7.4) | 6.9 (6.5-7.4) | 7.0 (6.5-7.4) | 0.804 |
| Albumin (g/dL) | 3.9 (3.4-4.3) | 3.9 (3.4-4.3) | 3.9 (3.4-4.3) | 0.775 |
| BUN (mg/dL) | 14.7 (11.5-19.2) | 14.8 (11.6-19.3) | 14.4 (11.3-18.9) | 0.671 |
| AST (IU/L) | 36 (22-75) | 37 (23-81) | 35 (22-68) | 0.433 |
| ALT (IU/L) | 30 (17-74) | 30 (17-72) | 29 (18-76) | 0.850 |
| Bilirubin (mg/dL) | 0.8 (0.5-3.4) | 0.8 (0.5-3.6) | 0.8 (0.5-2.7) | 0.650 |
| ALP (IU/L) | 146 (89-321) | 146 (88-321) | 141 (91-312) | 0.599 |
| Cholesterol (mg/dL) | 167 (134-198) | 168 (134-194) | 165 (132-202) | 0.900 |
| Tumor marker | ||||
| CEA (ng/mL) | 3.6 (1.9-16.0) | 3.2 (1.8-15.1) | 4.2 (2.4-18.9) | 0.056 |
| CA 19-9 (U/mL) | 160 (26-1,472) | 166 (25-1,587) | 145 (27-1,298) | 0.567 |
| Palliative Tx | ||||
| No | 148 (30.7) | 106 (31.2) | 42 (29.6) | 0.809 |
| Gem/Cis | 168 (34.9) | 120 (35.3) | 48 (33.8) | |
| Othersa) | 166 (34.4) | 114 (33.5) | 52 (36.6) |
Values are presented as number (%) or median (interquartile range). The Mann-Whitney U test was used for comparisons of continuous values. ECOG, Eastern Cooperative Oncology Group; CC, cholangiocarcinoma; BMI, body mass index; WBC, white blood cell; NLR, neutrophil-lymphocyte ratio; PLR, platelet-lymphocyte ratio; BUN, blood urea nitrogen; AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase; CEA, carcinoembryonic antigen; CA 19-9, carbohydrate antigen 19-9; Tx, treatment; Gem/Cis, combination chemotherapy with cisplatin and gemcitabine.
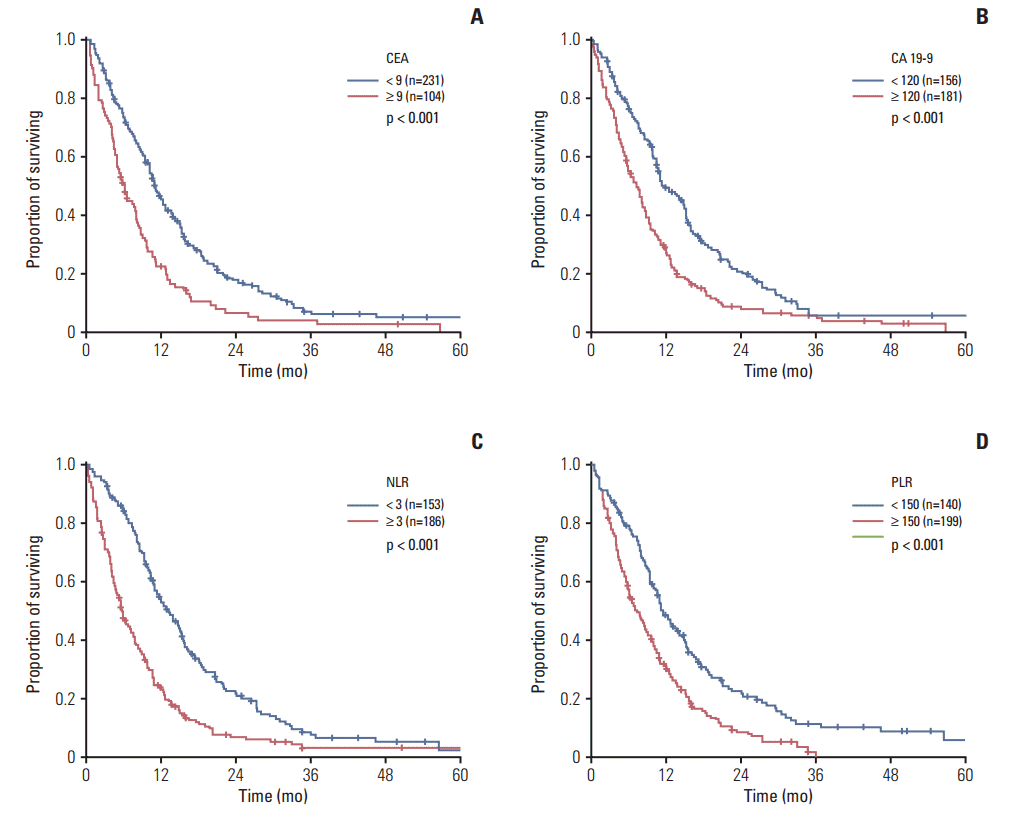
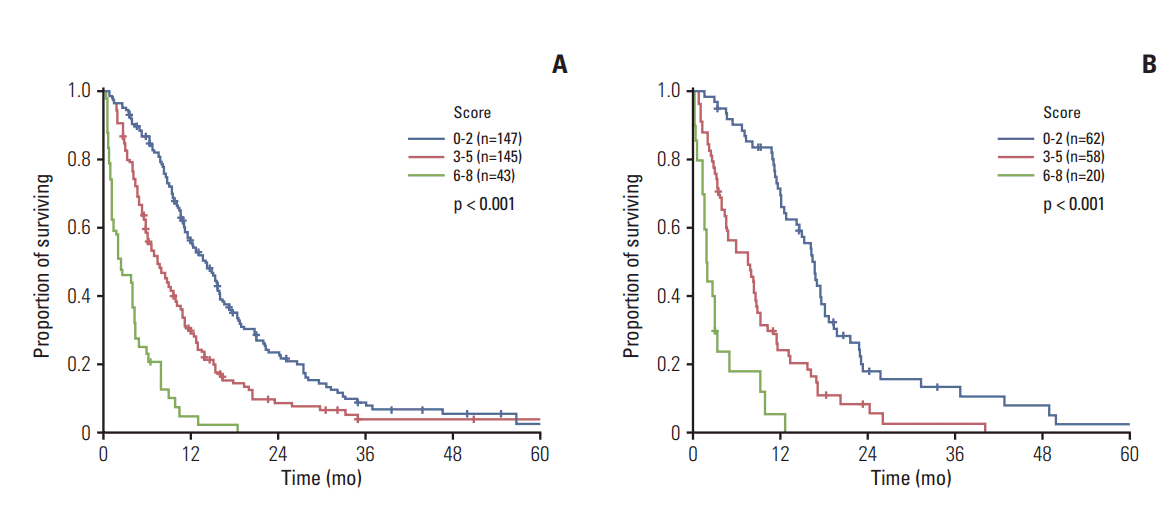
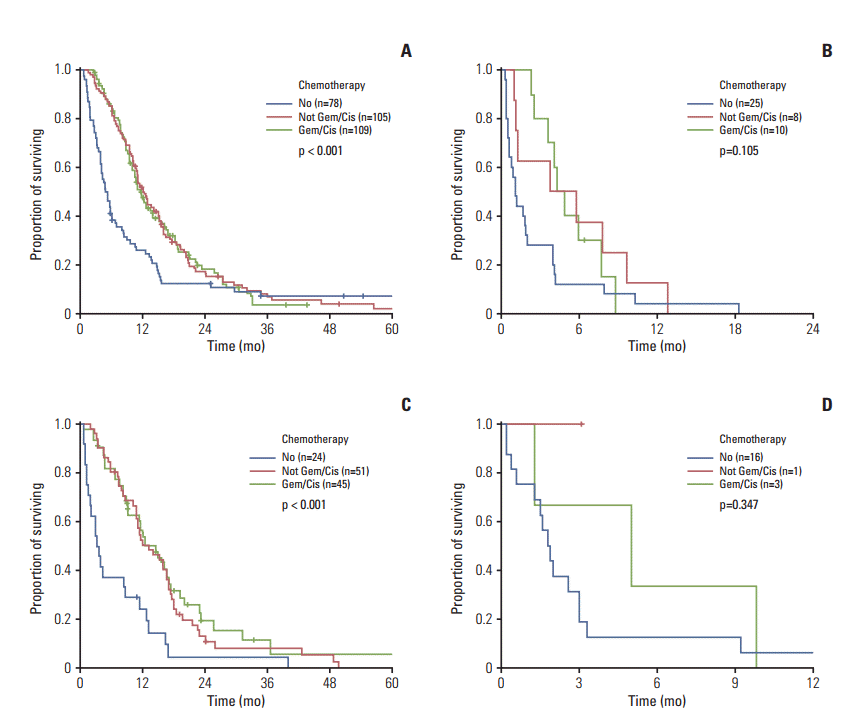
2. Assessment of the prognostic factors
Table 2.
| Characteristic |
Investigation dataset |
Validation dataset |
||||
|---|---|---|---|---|---|---|
| HR | 95% CI | p-value | HR | 95% CI | p-value | |
| Age (yr) | ||||||
| < 67 | 1 | 0.02 | 1 | 0.249 | ||
| ≥ 67 | 1.311 | 1.043-1.649 | 1.231 | 0.865-1.754 | ||
| Sex | ||||||
| Male | 1 | 0.515 | 1 | 0.937 | ||
| Female | 0.926 | 0.735-1.167 | 1.014 | 0.713-1.443 | ||
| Disease status | ||||||
| Recurrent | 1 | < 0.001 | 1 | 0.057 | ||
| Metastatic | 1.584 | 1.24-2.023 | 1.462 | 0.989-2.161 | ||
| ECOG | ||||||
| 0-2 | 1 | < 0.001 | 1 | < 0.001 | ||
| 3-4 | 4.499 | 3.205-6.316 | 5.276 | 3.102-8.972 | ||
| Primary site | ||||||
| Intrahepatic CC | 1 | 0.045 | 1 | 0.187 | ||
| Hilar CC | 0.802 | 0.563-1.144 | 0.834 | 0.479-1.451 | ||
| Non-hilar CC | 0.91 | 0.643-1.289 | 0.592 | 0.349-1.004 | ||
| Gallbladder cancer | 0.94 | 0.692-1.276 | 0.895 | 0.56-1.431 | ||
| Ampullary cancer | 0.514 | 0.331-0.8 | 0.519 | 0.255-1.057 | ||
| BMI | ||||||
| ≥ 18.5 | 1 | 0.001 | 1 | 0.024 | ||
| < 18.5 | 2 | 1.332-3.002 | 2.225 | 1.111-4.459 | ||
| WBC count (/μL) | ||||||
| Normal (< 10,800) | 1 | < 0.001 | 1 | < 0.001 | ||
| Abnormal (≥ 10,800) | 1.779 | 1.327-2.383 | 3.177 | 2.014-5.012 | ||
| Hemoglobin (g/dL) | ||||||
| Normal (≥ 12) | 1 | 0.005 | 1 | 0.209 | ||
| Abnormal (< 12) | 1.391 | 1.107-1.748 | 1.254 | 0.881-1.784 | ||
| Platelet (×103/μL) | ||||||
| Normal (≥ 150) | 1 | 0.086 | 1 | 0.814 | ||
| Abnormal (< 150) | 1.375 | 0.955-1.98 | 0.935 | 0.535-1.634 | ||
| Protein (g/dL) | ||||||
| Normal (≥ 6.9) | 1 | 0.013 | 1 | 0.006 | ||
| Abnormal (< 6.9) | 1.337 | 1.064-1.681 | 1.635 | 1.149-2.325 | ||
| Albumin (g/dL) | ||||||
| Normal (≥ 3.4) | 1 | < 0.001 | 1 | 0.011 | ||
| Abnormal (< 3.4) | 2.216 | 1.698-2.892 | 1.699 | 1.132-2.552 | ||
| BUN (mg/dL) | ||||||
| Normal (< 23) | 1 | 0.058 | 1 | 0.01 | ||
| Abnormal (≥ 23) | 1.401 | 0.988-1.988 | 1.956 | 1.178-3.25 | ||
| AST (IU/L) | ||||||
| Normal (< 30) | 1 | 0.151 | 1 | 0.176 | ||
| Abnormal (≥ 30) | 1.187 | 0.94-1.501 | 1.279 | 0.895-1.826 | ||
| ALT (IU/L) | ||||||
| Normal (< 33) | 1 | 0.773 | 1 | 0.237 | ||
| Abnormal (≥ 33) | 1.034 | 0.823-1.299 | 1.239 | 0.869-1.768 | ||
| Bilirubin (mg/dL) | ||||||
| Normal (< 1.2) | 1 | 0.435 | 1 | 0.198 | ||
| Abnormal (≥ 1.2) | 1.099 | 0.867-1.394 | 1.277 | 0.88-1.853 | ||
| ALP (IU/L) | ||||||
| Normal (< 123) | 1 | 0.043 | 1 | 0.043 | ||
| Abnormal (≥ 123) | 1.27 | 1.007-1.6 | 1.439 | 1.011-2.05 | ||
| Cholesterol (mg/dL) | ||||||
| Normal (< 139) | 1 | 0.005 | 1 | 0.022 | ||
| Abnormal (≥ 139) | 1.437 | 1.114-1.854 | 1.586 | 1.068-2.353 | ||
| CEA (ng/mL) | ||||||
| < 9 | 1 | < 0.001 | 1 | 0.016 | ||
| ≥ 9 | 1.812 | 1.414-2.321 | 1.604 | 1.094-2.353 | ||
| CA 19-9 (U/mL) | ||||||
| < 120 | 1 | < 0.001 | 1 | 0.441 | ||
| ≥ 120 | 1.668 | 1.323-2.104 | 1.149 | 0.807-1.637 | ||
| NLR | ||||||
| < 3 | 1 | < 0.001 | 1 | < 0.001 | ||
| ≥ 3 | 2.04 | 1.616-2.575 | 2.96 | 2.042-4.29 | ||
| PLR | ||||||
| < 150 | 1 | < 0.001 | 1 | 0.057 | ||
| ≥ 150 | 1.719 | 1.355-2.181 | 1.43 | 0.99-2.066 | ||
| Palliative Tx | ||||||
| No | 1 | < 0.001 | 1 | < 0.001 | ||
| Gem/Cis | 0.484 | 0.364-0.643 | 0.284 | 0.180-0.448 | ||
| Othersa) | 0.492 | 0.372-0.650 | 0.305 | 0.198-0.471 | ||
HR, hazard ratio; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; CC, cholangiocarcinoma; BMI, body mass index; WBC, white blood cell; BUN, blood urea nitrogen; AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase; CEA, carcinoembryonic antigen; CA 19-9, carbohydrate antigen 19-9; NLR, neutrophillymphocyte ratio; PLR, platelet-lymphocyte ratio; Tx, treatment; Gem/Cis, combination chemotherapy with cisplatin and gemcitabine.




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download