1. International Agency for Research on Cancer. GLOBOCAN 2012: estimated cancer incidence, mortality and prevalence worldwide in 2012 [Internet]. Lyon: IARC;2015. [cited 2015 Jul 26]. Available from:
http://globocan.iarc.fr/Default.aspx.
2. American Cancer Society. Colorectal cancer facts and figures 2011-2013. Atlanta, GA: American Cancer Society;2011.
3. Logan RF, Patnick J, Nickerson C, Coleman L, Rutter MD, von Wagner C. Outcomes of the Bowel Cancer Screening Programme (BCSP) in England after the first 1 million tests. Gut. 2012; 61:1439–46.

4. Korean National Cancer Center. Korea cancer facts and figures 2014. Goyang: Korean National Cancer Center;2014.
5. Asaria M, Griffin S, Cookson R, Whyte S, Tappenden P. Distributional cost-effectiveness analysis of health care programmes: a methodological case study of the UK Bowel Cancer Screening Programme. Health Econ. 2015; 24:742–54.
6. Korean Statistical Information Service [Internet]. Daejeon: Statistics Korea;2015. [cited 2015 Feb 28]. Available from:
http://kosis.kr/.
7. Park SM, Yun YH, Kwon S. Feasible economic strategies to improve screening compliance for colorectal cancer in Korea. World J Gastroenterol. 2005; 11:1587–93.

8. Markowitz AJ, Winawer SJ. Management of colorectal polyps. CA Cancer J Clin. 1997; 47:93–112.

9. Brenner H, Altenhofen L, Katalinic A, Lansdorp-Vogelaar I, Hoffmeister M. Sojourn time of preclinical colorectal cancer by sex and age: estimates from the German national screening colonoscopy database. Am J Epidemiol. 2011; 174:1140–6.

10. Djalalov S, Rabeneck L, Tomlinson G, Bremner KE, Hilsden R, Hoch JS. A review and meta-analysis of colorectal cancer utilities. Med Decis Making. 2014; 34:809–18.

11. Ladabaum U, Allen J, Wandell M, Ramsey S. Colorectal cancer screening with blood-based biomarkers: cost-effectiveness of methylated septin 9 DNA versus current strategies. Cancer Epidemiol Biomarkers Prev. 2013; 22:1567–76.

12. Shin A, Choi KS, Jun JK, Noh DK, Suh M, Jung KW, et al. Validity of fecal occult blood test in the national cancer screening program, Korea. PLoS One. 2013; 8:e79292.

13. Lansdorp-Vogelaar I, Knudsen AB, Brenner H. Cost-effectiveness of colorectal cancer screening. Epidemiol Rev. 2011; 33:88–100.

14. Whyte S, Walsh C, Chilcott J. Bayesian calibration of a natural history model with application to a population model for colorectal cancer. Med Decis Making. 2011; 31:625–41.

15. Mandel JS, Bond JH, Church TR, Snover DC, Bradley GM, Schuman LM, et al. Reducing mortality from colorectal cancer by screening for fecal occult blood. Minnesota Colon Cancer Control Study. N Engl J Med. 1993; 328:1365–71.
16. Shaukat A, Mongin SJ, Geisser MS, Lederle FA, Bond JH, Mandel JS, et al. Long-term mortality after screening for colorectal cancer. N Engl J Med. 2013; 369:1106–14.

17. Shankaran V, McKoy JM, Dandade N, Nonzee N, Tigue CA, Bennett CL, et al. Costs and cost-effectiveness of a low-intensity patient-directed intervention to promote colorectal cancer screening. J Clin Oncol. 2007; 25:5248–53.

18. Hewitson P, Ward AM, Heneghan C, Halloran SP, Mant D. Primary care endorsement letter and a patient leaflet to improve participation in colorectal cancer screening: results of a factorial randomised trial. Br J Cancer. 2011; 105:475–80.

19. Korea Ministry of Health and Welfare. Notification 2014-2: National medicial checkup guideline. Sejong: Korea Ministry of Health and Welfare;2014.
21. Korea National Health Insurance Corporation. Korea national insurance statistical yearbook 2012. Wonju: Korea National Health Insurance Corporation;2013.
22. Mariotto AB, Yabroff KR, Shao Y, Feuer EJ, Brown ML. Projections of the cost of cancer care in the United States: 2010-2020. J Natl Cancer Inst. 2011; 103:117–28.

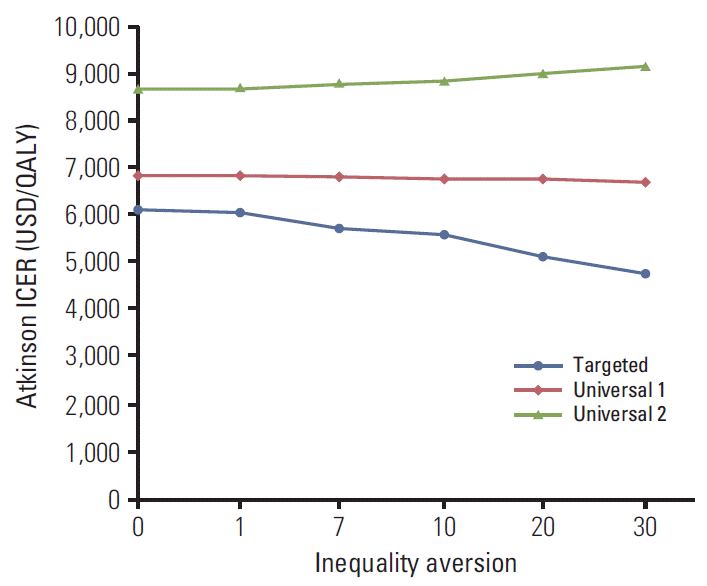
23. Atkinson AB. On the measure of inequality. J Econ Theor. 1970; 2:244–63.
24. Pirttila J, Uusitalo R. A ‘Leaky Bucket’ in the real world: estimating inequality aversion using survey data. Economica. 2010; 77:60–76.
25. Lambert PJ, Millimet DL, Slottje D. Inequality aversion and the natural rate of subjective inequality. J Public Econ. 2003; 87:1061–90.





 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download