Abstract
Purpose
The aim of this study was to evaluate the role of radiotherapy (RT) in the management of Ewing sarcoma family tumors (ESFT).
Materials and Methods
Retrospective analysiswas performed on 91 patientswith localized ESFT treated from 1988 to 2012. Primary tumor size was ≥ 8 cm in 33 patients. Surgery, RT, and combined surgery with RT were applied in 37, 15, and 33 patients, respectively.
Results
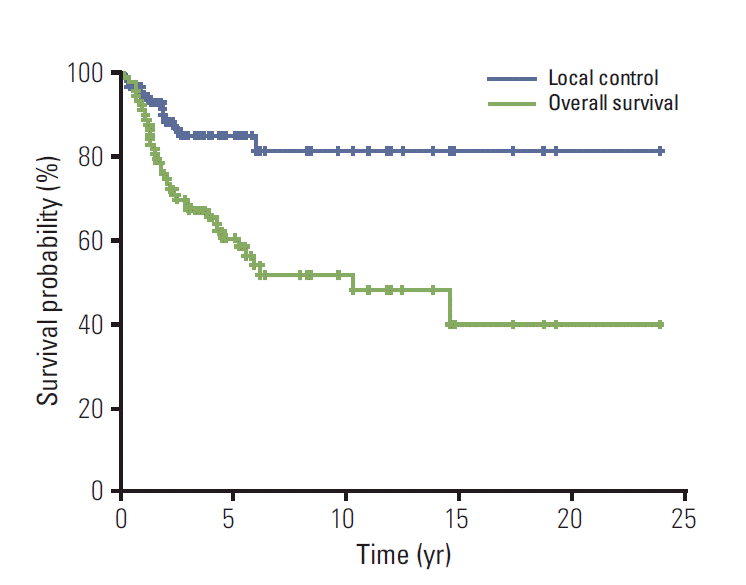
Median follow-up was 43.8 months. Forty-three patients (47.3%) showed recurrence or progressive disease. Twelve patients (13.2%) showed local failure after initial treatment. Thirty-nine patients (42.9%) experienced distant metastases. The 5-year overall survival (OS), progression-free survival, and local control (LC) were 60.5%, 58.2%, and 85.1%, respectively. According to treatment, 5-year LCwas 64.8% with RT and 90.2% with combined surgery and RT (p=0.052). Prognostic factors for OS were tumor size (≥ 8 cm, p < 0.001) and surgical resection (p < 0.001). In large tumors (≥ 8 cm), combined surgery and RT produced better LC compared to RT (p=0.033). However, in smaller tumors (< 8 cm), RT without surgery resulted in a similar LC rate as RT with surgery (p=0.374).
Conclusion
RT used for patients with unfavorable risk factors resulted in worse outcome than for patientswho received surgery. Smallertumors could be controlled locallywith chemotherapy and RT. For large tumors, combined surgery and RT is needed. Proper selection of local treatment modality, RT, surgery, or both is crucial in the management of ESFT.
The Ewing sarcoma family tumors (ESFT) include Ewing sarcoma, peripheral primitive neuroectodermal tumor (PNET), and Askin’s tumor of the chest wall [1]. These sarcomas are related to and originate from unique mesenchymal stem cells. A common chromosomal translocation that results in EWS-ETS has been implicated in 80% to 95% of cases of ESFT [2,3].
Current standard treatment is neoadjuvant chemotherapy followed by local treatment such as surgery, radiotherapy (RT), or a combination of both modalities [2]. The application of neoadjuvant chemotherapy increases the possibility of surgical resection by shrinking the primary tumor. Doxorubicin, cyclophosphamide, vincristine, and dactinomycin are known to be effective. A more effective chemotherapy regimen including etoposide and ifosfamide was introduced in 2003 [2,4]. Although ESFT are radiosensitive tumors [5] and RT has been the treatment choice for local treatment in ESFT [6], the role of resection has increased with an improvement of surgical technique [7] and there are concerns over secondary malignancies and local failures after RT. Surgery has been chosen rather than RT for “dispensable bones” (e.g., fibula, rib, and smaller lesions of the hands or feet) and has been often considered in young patients, in whom radiation late effect was expected to be severe [8]. Moreover, the use of neoadjuvant chemotherapy often decreases tumors [9] and results in an increase of complete tumor resection rate [10].
This study was performed to find the role of RT in the multidisciplinary management of patients with ESFT by retrospective review. ESFT is especially rare in Asia [11]. We collected the patients from two large-scale institutions and could thus analyze a substantial number of Korean ESFT patients in this study. This study's findings could provide valuable clinical information about Asian ESFT patients.
Ninety-one patients who were diagnosed with localized ESFT and treated in Severance Hospital, Yonsei University Health System, and Samsung Medical Center, Sungkyunkwan University School of Medicine between July 1988 and July 2012 were collected for evaluation. Included pathologic diagnoses were osseous Ewing’s sarcoma, extraosseous (extraskeletal) Ewing’s sarcoma, and peripheral PNET.
Basically, all patients were evaluated by computed tomography (CT) scan and the primary tumor size was defined as the greatest tumor dimension (maximal diameter in axial or perpendicular plane) in the CT scan. Patients were divided into two groups by tumor size (< 8 cm vs. ≥ 8 cm) [12] and primary tumor size was ≥ 8 cm in 33 patients. According to tumor location or extent, magnetic resonance imaging (MRI), bone scan, and positron emission tomography were conducted in 80, 44, and 47 patients, respectively.
Ages ranged from 1 to 66 years with a median age of 18 years. The sites of primary tumor were as follows: 23 patients in extremities, 13 in spine or paravertebral regions, 11 in pelvis, nine in head and skull, and 35 in trunk or other sites (Table 1).
All patients received chemotherapy basically comprised of a combination of vincristine, dactinomycin, cyclophosphamide, and doxorubicin. Etoposide and/or ifosfamide was added in 65 patients. High-dose chemotherapy with peripheral blood stem cell transplantation was applied in 13 patients; among them, 10 patients underwent stem cell transplantation as second-line treatments, especially for distant failure (n=7).
As for the local treatment, surgery, RT, and combined surgery with RT were performed in 37, 15, and 33 patients, respectively (Fig. 1). In regard to extent of surgical resection, R0, R1, and R2 resections were performed in 48, 8, and 14 patients, respectively. Six patients did not receive local therapy after chemotherapy due to refusal, progression, or toxicity of chemotherapy (Fig. 1). Among the patients who underwent combined surgery with RT in localized disease (n=33), 32 patients received postoperative RT and one patient received preoperative RT (Fig. 1). Among the patients who underwent surgery without RT (n=37), seven patients (18.9%) received incomplete resections. However, 15 patients (46 9%) among those treated with postoperative RT (n=32) received incomplete resections. Surgical resection without postoperative RT was applied more in patients with tumors in extremities (13/23, 56.5%) than in those with tumors in spine or paravertebral area (3/13, 23.1%). Among the 32 patients with large tumors (≥ 8 cm), 8, 12, and 7 patients underwent surgery, combined surgery and RT, and RT for local treatment, respectively. Five patients with tumors ≥ 8 cm did not receive local therapy due to progression, treatment refusal, or toxicity.
Forty-eight patients received RT with or without surgery for the local control (LC) of primary tumor. Those treated with RT without surgery (n=15) largely consisted of ≥ 8-cm tumor (n=8) and pelvic tumor (n=6).
Clinical treatment volume (CTV) was defined as the prechemotherapy tumor volume or postoperative tumor bed plus margin, usually 1-3 cm [13,14]. Gross tumor volume (GTV) was defined as postchemotherapy tumor volume. Modification of CTV margin was allowed for tumors located in epiphysis, spine, head and neck to avoid excessive radiation. Among patients who received RT with field reduction, GTV was irradiated with a reduced field with a median dose of 54.0 Gy (range, 45.0 to 59.4 Gy) after irradiation of CTV with median dose of 41.4 Gy (range, 30.6 to 50.4 Gy). For the other patients who underwent RT without field reduction, CTV was irradiated with median dose of 50.4 Gy (range, 40.0 to 66.6 Gy). One patient with a huge tumor involving ribs received 80 Gy to GTV and 50 Gy to CTV with intensity-modulated RT technique, instead of surgical resection.
Median follow-up was 43.8 months. The median follow-up period for surviving patients was 60.7 months. Treatment response of patients was evaluated at the end of local therapy, 1 and 3 months after local therapy, and semiannually thereafter. Follow-up assessment included CT or MRI of primary tumor.
Progression was defined as clinical or radiographic increase in the size of primary or metastatic tumor or new appearance of metastatic lesion. Progression-free survival (PFS) was defined as the time interval from date of histological diagnosis to date of first event (relapse or progression or death).
PASW Statistics ver. 18.0 (SPSS Inc., Chicago, IL) was used for statistical analysis. Chi-square test was used to evaluate treatment and prognostic factors for LC. Survival and LC rates were calculated using Kaplan-Meier estimation. The log-rank test was used for group comparisons. A Cox proportional hazards model was used to clarify independent predictive factor in multivariate analysis. Statistical significance was defined as a p-value of < 0.05.
During the follow-up period, 43 patients (47.3%) showed recurrence or progressive disease: 12 patients with local failure (Table 2) and 39 patients with distant metastasis (8 patients experienced both local and distant failure). Five patients who experienced local failure had large primary tumors (≥ 8 cm). Among these five patients with large tumors, one patient underwent surgical resection, and two patients underwent RT for local therapy. In addition, the other two patients did not receive any local treatment due to early progression. The common sites of local failure were pelvis (n=3) and extremity (n=3). Frequent metastatic sites were lung in 19 patients and bone in 13 patients. The rate of distant metastasis was higher in patients with tumors primarily located in spinal or paravertebral regions (7/13, 53.8%). Among these 43 patients with progression, 26 patients died, 12 patients were alive with disease 7.3-62.4 months after diagnosis, and five patients were saved with surgical resection.
The primary tumors were not controlled in 12 patients (Table 2) and 5-year actuarial LC rate was 85.1% (Fig. 2). Poorer responders to chemotherapy tended to show shorter LC (Table 2). LC was different according to application of local treatment modalities (n=85, p=0.042) (Fig. 3). LC rate was higher in patients with combined surgery and RT compared to those with RT alone (5-year LC 90.2% vs. 64.8%, p=0.052). Subset analyses were performed in patients with localized disease who received RT with or without surgery (n=48). In patients with tumors ≥ 8 cm (n=20), combined RT with surgery showed superior LC than RT without surgery (p=0.033) (Fig. 4A). However, there was no statistical difference in LC between the two groups among patients with tumors < 8 cm (n=28, p=0.374) (Fig. 4B).
On top of that, radiation dose did not affect LC. The dose-response relationship (< 50 Gy vs. ≥ 50 Gy) for LC was not observed in patients with postoperative RT or with definitive RT without surgery (p=0.227 and p=0.180, respectively).
Further subset analyses for LC and resection margin status were performed to find the role of adjuvant RT in patients who underwent surgery (n=69, among 70 patients who underwent surgery±RT, one patient who underwent preoperative RT and complete resection was excluded from the analyses). Fig. 5A presents the LC according to adjuvant RT in patients with incomplete resection (n=22, 8 patients with R1 resection and 14 patients with R2 resection). Adjuvant RT did not significantly improve the LC in cases with incomplete resection (p=0.254) (Fig. 5A), nor did adjuvant RT provide a significant benefit on LC in cases with complete resection (n=47, p=0.241) (Fig. 5B).
The 5-year overall survival (OS) and PFS were 60.5% (Fig. 2) and 58.2%, respectively. The 5-year PFS was different according to the application of local modality (n=85, p=0.007). The 5-year PFS was 55.9% in patients treated with combined RT and surgery and 39.4% in those treated with RT without surgery.
Univariate analysis showed that prognostic factors for OS were primary tumor size (p < 0.001) and surgical resection (p < 0.001). Tumor location did not affect the survival outcome (extremity vs. nonextremity, p=0.597; spine/paravertebral lesion vs. nonspine/paravertebral lesion, p=0.902; and pelvis vs. nonpelvis, p=0.424). Multivariate analysis showed that primary tumor size (p=0.001) and surgical resection (p=0.024) were independent prognostic factors for OS (Table 3).
In the subset analysis with patients who received RT with or without surgery (n=48), surgical resection did not significantly improve OS (p=0.161) and PFS (p=0.068) compared to RT without surgery. However, in patients with tumors ≥ 8 cm (n=20), RT combined with surgery showed superior PFS to RT without surgery (p=0.016).
Two patients (2.2%) experienced secondary malignancies after treatment. Acute myeloid leukemia and diffuse large B-cell lymphoma (DLBCL) each occurred in one patient, respectively. Thirty-eight patients expired during the follow-up period. Among them, 33 deaths were associated with progression of ESFT. One death was related to uncontrolled DLBCL. Three patients died due to peripheral blood stem cell transplantation-related toxicities (sepsis). One death was related to pneumonia. As for late complications, two cases of limb shortage and one case of stress fracture in bone were observed during the follow-up period.
Local treatment for ESFT can be accomplished by surgery, RT, or both. However, no randomized trials have compared surgery and RT. Although the role of surgery is emphasized in some studies [15,16], RT still plays an important role in patients who need functional preservation or who have unresectable tumors. Combined surgery and RT can be used in cases with large tumors or with positive resection margins.
Several retrospective studies have suggested that surgery yields better LC and survival than RT [15,16]. In the present study, PFS was better in patients who received surgery than in patients who did not. Especially in large-sized tumors (diameter, ≥ 8 cm), RT without surgery demonstrated inferior LC (p=0.033) (Fig. 4A) and PFS (p=0.016) compared to combined surgery with RT. However, direct comparison between RT and surgery was difficult due to patient selection bias [14]. The patients who received RT without surgery had more unfavorable prognostic factors such as tumor location or size, not amenable to resection. As in the current series, many surgical series included patients with lower risks, such as smaller tumors and tumors at dispensable bones [16,17], and the advantage and disadvantages of surgery and RT are different. Compared with RT, which can cause secondary malignancy and bone growth disturbances in young children, surgical resection could result in functional deficits or adverse cosmetic effects. Therefore, surgery and RT are not competitive modalities, but complimentary modalities in the management of ESFT. Proper combination of function-preserving surgery and RT with optimized radiation volume and dose is mandatory for local treatment of ESFT. The selection of local treatment modality should be based on not only LC rate but also rehabilitation capacity and psychological effect.
RT can be used as an alternative or adjuvant treatment modality for surgery in ESFT. RT could be considered for treatment of tumors located in the spine or paravertebral area [18], where complete surgical resection is rarely possible. Presently, combined RT and surgery are mainly performed in patients with spine or paravertebral tumors. As a result, compared to a previous report [19] that documented poor treatment outcome in spine tumors, the current study showed that OS of spine and paravertebral ESFT were similar to that of other locations (p=0.902), as shown in Table 3. In addition, RT can be applied in cases of expected high morbidity after surgical resection such as tumors located in weight-bearing bones or pelvis [20]. Functional impairment in the case of shoulder joints or cosmetic adverse effects in cases of head and neck tumor-related resection [21] could influence the preference of RT.
The primary tumor burden and location also affected the treatment selections. Those who were treated with RT without surgery mostly had large tumors (≥ 8 cm, 8/15, 53.3%) and/or pelvic tumors (6/15, 40.0%). RT used for patients with unfavorable risk factors resulted in worse outcome than for patients who received surgery. Adequate combination of RT and less-ablative surgery could help patients who are at high risk for surgical morbidity or mortality (e.g., pelvis tumors) and improve treatment outcome.
In the current study, patients with tumors exceeding 8 cm showed poorer outcome. Although local failure rate was similar between small- and large-sized tumors (12.0% for < 8 cm vs. 15.2% for ≥ 8 cm), the distant failure rate was higher in large-sized tumors (36.2% for < 8 cm vs. 54.5% for ≥ 8 cm). This means that patients with a large tumor have greater possibility of systemic disease and poor response to chemotherapy resulting in distant failure as well as local failure.
The role of chemotherapy is robust in the management of ESFT to eradicate systemic disease and to achieve LC. Effective systemic chemotherapy shrinks the tumor and facilitates surgical resection, and eventually reduces radiation dose and volume or even omits RT. Most of the local failures in this study showed poor response to chemotherapy. Therefore, response to chemotherapy is a strong prognostic factor [22].
Although chemotherapy with etoposide and/or ifosfamide improves survival [2], distant metastases are still the main cause of treatment failure [23] and a more effective systemic treatment modality is needed. High-dose chemotherapy with peripheral blood stem cell transplantation has been tried for consolidation therapy or salvage therapy. However, it was not as effective [24].
Secondary malignancy after alkylating agents is another concern. According to an Italian sarcoma group experience [8], 2.8% of patients experienced secondary malignancies and radiation-induced osteosarcomas were the most common secondary malignancy. Dunst et al. [25] reported a 4.7% cumulative incidence of secondary malignancy after 15 years in patients with Ewing’s sarcoma using data in the CESS-81 and CESS-86. Two patients experienced secondary malignancies (2.2%). There was no secondary solid tumor related to radiation and two cases were hematologic malignancies.
As for limitations, this study did not fully deal with the functional outcomes after RT. During follow-up, we observed two cases of limb shortage and one case of stress fracture in bone. However, evaluation of functional outcome demands close long-term follow-up with standardized evaluation tools and a multidisciplinary team approach. Minimizing late complications is as important as maximizing treatment efficacy. Further studies focused on functional status in survival are warranted. Innovation in RT is still needed to improve the functional outcomes.
Furthermore, this study was based on data collected over 25 years. Therefore, it has inherent limitations that include changes of standard systemic chemotherapy regimens, RT technologies, and treatment strategies for LC.
The development of chemotherapy during the study period resulted in improved survival. Grier et al. [2] reported in a randomized trial that addition of ifosfamide and etoposide improved the survival outcome for patients without metastatic disease. It was noteworthy that the treatment benefit was greater among high-risk patients with huge tumors or tumors in pelvis [2]. However, it is not easy to conclude that the overall improvement of treatment outcome is mainly caused by the development of chemotherapy. RT changed from cobalt-60 therapy to three-dimensional conformal RT, and surgical techniques have also been improved in the same period of time.
The collaboration of a multidisciplinary team could contribute to increased survival rate of patients with ESFT. Since most ESFT failures are systemic failures, more effective chemotherapy for systemic control is still warranted. RT and surgery can be seen not as competitive but as complimentary. Maximum cytoreduction with chemotherapy and surgical resection followed by RT could improve LC, especially in large tumors. Small tumors could be successfully treated by surgery as well as RT. RT was effective in LC, and proper selection of local treatment modality, RT, surgery, or both, is crucial in the management of ESFT.
References
2. Grier HE, Krailo MD, Tarbell NJ, Link MP, Fryer CJ, Pritchard DJ, et al. Addition of ifosfamide and etoposide to standard chemotherapy for Ewing's sarcoma and primitive neuroectodermal tumor of bone. N Engl J Med. 2003; 348:694–701.

3. Delattre O, Zucman J, Melot T, Garau XS, Zucker JM, Lenoir GM, et al. The Ewing family of tumors: a subgroup of small-round-cell tumors defined by specific chimeric transcripts. N Engl J Med. 1994; 331:294–9.
4. Miser JS, Krailo MD, Tarbell NJ, Link MP, Fryer CJ, Pritchard DJ, et al. Treatment of metastatic Ewing's sarcoma or primitive neuroectodermal tumor of bone: evaluation of combination ifosfamide and etoposide: a Children's Cancer Group and Pediatric Oncology Group study. J Clin Oncol. 2004; 22:2873.
5. Sailer SL. The role of radiation therapy in localized Ewing' sarcoma. Semin Radiat Oncol. 1997; 7:225–35.
6. Krasin MJ, Rodriguez-Galindo C, Billups CA, Davidoff AM, Neel MD, Merchant TE, et al. Definitive irradiation in multidisciplinary management of localized Ewing sarcoma family of tumors in pediatric patients: outcome and prognostic factors. Int J Radiat Oncol Biol Phys. 2004; 60:830–8.

7. Cotterill SJ, Ahrens S, Paulussen M, Jurgens HF, Voute PA, Gadner H, et al. Prognostic factors in Ewing's tumor of bone: analysis of 975 patients from the European Intergroup Cooperative Ewing's Sarcoma Study Group. J Clin Oncol. 2000; 18:3108–14.

8. Longhi A, Ferrari S, Tamburini A, Luksch R, Fagioli F, Bacci G, et al. Late effects of chemotherapy and radiotherapy in osteosarcoma and Ewing sarcoma patients: the Italian Sarcoma Group Experience (1983-2006). Cancer. 2012; 118:5050–9.
9. Bacci G, Picci P, Ferrari S, Mercuri M, Brach del Prever A, Rosito P, et al. Neoadjuvant chemotherapy for Ewing's sarcoma of bone: no benefit observed after adding ifosfamide and etoposide to vincristine, actinomycin, cyclophosphamide, and doxorubicin in the maintenance phase. Results of two sequential studies. Cancer. 1998; 82:1174–83.
10. Shamberger RC, LaQuaglia MP, Gebhardt MC, Neff JR, Tarbell NJ, Marcus KC, et al. Ewing sarcoma/primitive neuroectodermal tumor of the chest wall: impact of initial versus delayed resection on tumor margins, survival, and use of radiation therapy. Ann Surg. 2003; 238:563–7.
11. Lee JA, Kim DH, Cho J, Lim JS, Koh JS, Yoo JY, et al. Treatment outcome of Korean patients with localized Ewing sarcoma family of tumors: a single institution experience. Jpn J Clin Oncol. 2011; 41:776–82.

12. Indelicato DJ, Keole SR, Shahlaee AH, Shi W, Morris CG, Marcus RB Jr. Definitive radiotherapy for ewing tumors of extremities and pelvis: long-term disease control, limb function, and treatment toxicity. Int J Radiat Oncol Biol Phys. 2008; 72:871–7.

13. Donaldson SS. Ewing sarcoma: radiation dose and target volume. Pediatr Blood Cancer. 2004; 42:471–6.

14. La TH, Meyers PA, Wexler LH, Alektiar KM, Healey JH, Laquaglia MP, et al. Radiation therapy for Ewing's sarcoma: results from Memorial Sloan-Kettering in the modern era. Int J Radiat Oncol Biol Phys. 2006; 64:544–50.

16. Schuck A, Ahrens S, Paulussen M, Kuhlen M, Konemann S, Rube C, et al. Local therapy in localized Ewing tumors: results of 1058 patients treated in the CESS 81, CESS 86, and EICESS 92 trials. Int J Radiat Oncol Biol Phys. 2003; 55:168–77.

17. Dunst J, Hoffmann C, Ahrens S, Jurgens H. Surgery versus radiotherapy in Ewing's sarcoma with good prognosis: analysis of the CESS-86 data. Strahlenther Onkol. 1996; 172:244–8.
18. Schuck A, Ahrens S, von Schorlemer I, Kuhlen M, Paulussen M, Hunold A, et al. Radiotherapy in Ewing tumors of the vertebrae: treatment results and local relapse analysis of the CESS 81/86 and EICESS 92 trials. Int J Radiat Oncol Biol Phys. 2005; 63:1562–7.

19. Bacci G, Boriani S, Balladelli A, Barbieri E, Longhi A, Alberghini M, et al. Treatment of nonmetastatic Ewing's sarcoma family tumors of the spine and sacrum: the experience from a single institution. Eur Spine J. 2009; 18:1091–5.

20. Burgers JM, Oldenburger F, de Kraker J, van Bunningen BN, van der Eijken JW, Delemarre JF, et al. Ewing's sarcoma of the pelvis: changes over 25 years in treatment and results. Eur J Cancer. 1997; 33:2360–7.

21. Whaley JT, Indelicato DJ, Morris CG, Hinerman RW, Amdur RJ, Mendenhall WM, et al. Ewing tumors of the head and neck. Am J Clin Oncol. 2010; 33:321–6.

22. Picci P, Rougraff BT, Bacci G, Neff JR, Sangiorgi L, Cazzola A, et al. Prognostic significance of histopathologic response to chemotherapy in nonmetastatic Ewing's sarcoma of the extremities. J Clin Oncol. 1993; 11:1763–9.

23. Burgert EO Jr, Nesbit ME, Garnsey LA, Gehan EA, Herrmann J, Vietti TJ, et al. Multimodal therapy for the management of nonpelvic, localized Ewing's sarcoma of bone: intergroup study IESS-II. J Clin Oncol. 1990; 8:1514–24.

Fig. 1.
Patient arrangement according to local treatment methods. ESFT, Ewing sarcoma family tumors; RT, radiotherapy.
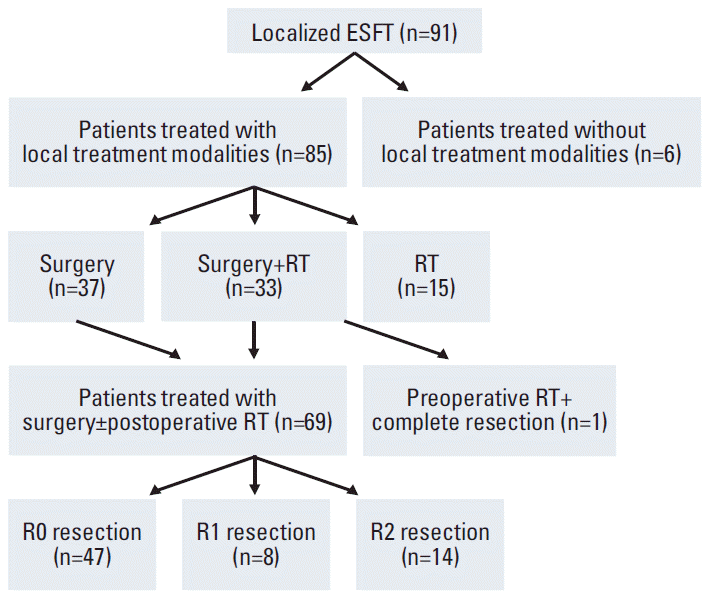
Fig. 3.
Local control in localized disease; comparison among surgery alone (n=37), combined surgery with radiotherapy (RT; n=33), and RT without surgery (n=15).
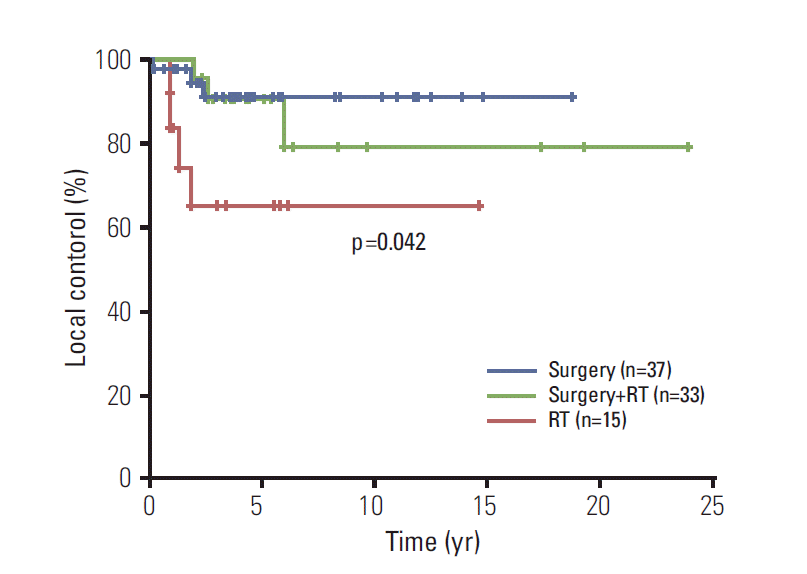
Fig. 4.
. Local control in localized tumors ≥ 8 cm according to treatment methods (A); comparison between combined surgery and radiotherapy (RT) (n=12) and RT without surgery (n=8), and local control in localized tumors < 8 cm according to treatment methods (B); comparison between combined surgery and RT (n=21) and RT without surgery (n=7).
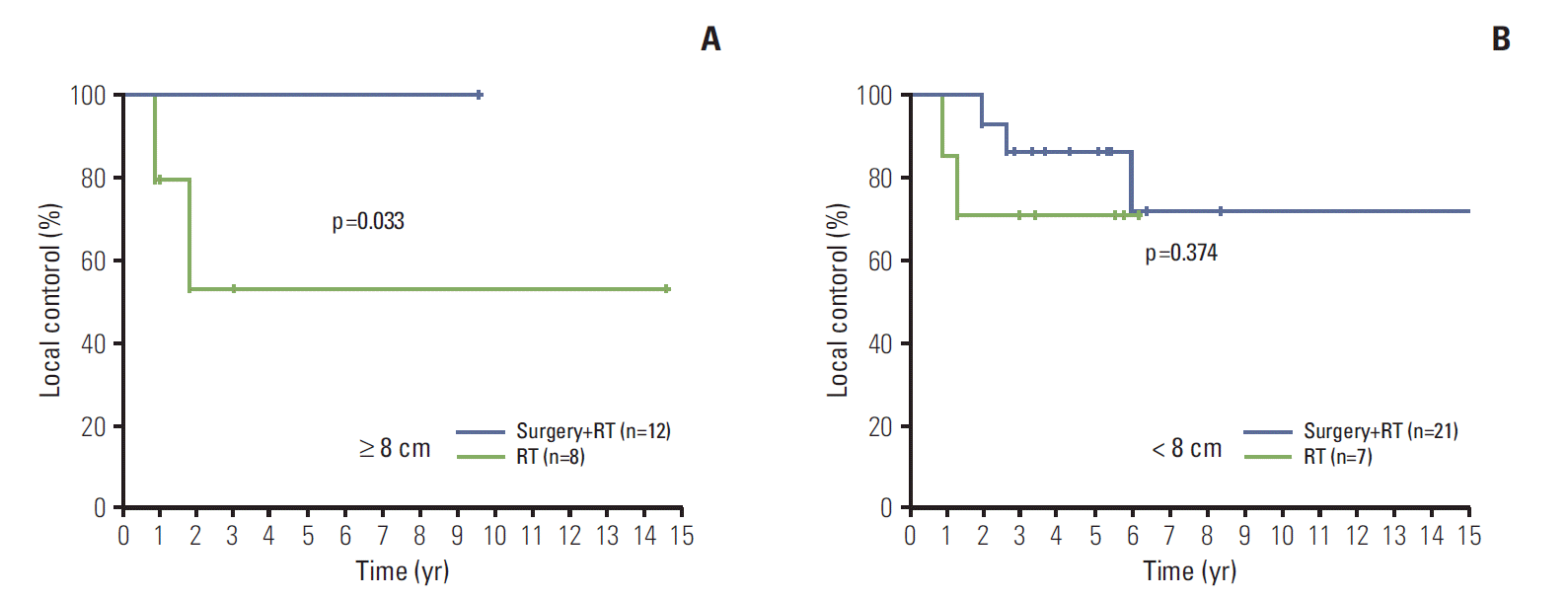
Fig. 5.
Local control in patients who underwent surgical resection according to implementation of adjuvant radiotherapy (RT) (A); comparison between surgery (n=7) and combined surgery and RT (n=15) in patients with incomplete resection (B); comparison between surgery (n=30) and combined surgery and RT (n=17) with complete resection.
Table 1.
Patient characteristics (n=91)
Table 2.
Characteristics of 12 patients with local failure
Table 3.
Univariate and multivariate analysis for overall survival (n=91)




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download