Introduction
Ifosfamide is an alkylating agent frequently used to treat ovarian, testicular, cervical, and head and neck cancers, as well as lymphomas and soft tissue sarcomas. Frequent side effects of ifosfamide include myelotoxicity, hemorrhagic cystitis and neurotoxicity (mental confusion, drowsiness, hallucinations, disorientation, and coma) [
1]. Although the use of ifosfamide was initially limited by severe hemorrhagic cystitis, this problem was resolved by the concurrent administration of sodium 2-mercaptoethane sulfonate (mesna), a synthetic thiol compound that binds to toxic species of ifosfamide in the bladder to form stable nontoxic thioether compounds. Ifosfamide and mesna can both have adverse effects in the central nervous system (CNS), with 10-30% of ifosfamide-treated patients experiencing CNS toxicity [
2-
6]. These neurotoxicities are dose dependent and may be fatal. Furthermore, the mechanism of action and factors predisposing to ifosfamide-induced encephalopathy are poorly understood. Methylene blue (MB) can be used to treat these encephalopathies, but its mechanism of action is unclear. In this report, we describe two patients with recurrent epithelial ovarian cancer who experienced fatal ifosfamide-induced encephalopathy, as well as a review of the proposed pathophysiology of ifosfamide-induced encephalopathy and the use of MB in its management.
Go to :

Case Reports
1. Case 1
A 54-year-old, gravida 2, para 2, married woman presented with recurrent epithelial ovarian cancer (endometrioid adenocarcinoma). She had noninsulin dependent diabetes mellitus, diabetic retinopathy, and hypertension. Seven years earlier, she underwent coronary artery bypass grafting due to coronary artery disease.
Three years ago, she was transferred to our center after laparoscopic bilateral salpingo-oophorectomy which led to a diagnosis of epithelial ovarian cancer. She underwent a debulking operation, including total abdominal hysterectomy and pelvic and paraaortic lymph node dissection, total omentectomy, appendectomy, peritoneal mass excision, and left ureter resection and anastomosis. The tumor was categorized as International Federation of Obstetrics and Gynecology (FIGO) stage IIIc and cytoreductive surgery was suboptimal. Following the surgery, she received 6 cycles of adjuvant chemotherapy with paclitaxel/carboplatin. However, because there was disease progression in this patient, she was further treated with 3 cycles of topotecan, followed by 3 cycles of docetaxel. An abdomino-pelvic computed tomography (CT) scan showed aggravation of disease and her serum carbohydrate antigen 125 (CA-125) concentration was elevated from 534 to 1,270 U/mL. We decided to change her chemotherapy regimen to ifosfamide monotherapy.
She was started on ifosfamide 1,200 mg/m
2/day, administered via continuous infusion on days 1-3 of each 4 week cycle, along with mesna 300 mg/m
2/day. On the third day she received ifosfamide, her mental status worsened, with a change from drowsiness to stupor, followed by disorientation, stereotyped movements, and emotional instability. She had no lateralizing neurologic signs. Her blood test results, including measurements of serum glucose, brain natriuretic peptide, and electrolytes, were normal, and her cerebrospinal fluid was negative for malignant cells. In addition, brain magnetic resonance imaging (MRI) findings were normal. An electroencephalogram showed frequent 1-1.5 Hz periodic or single triphasic waves on both hemispheres (
Fig. 1). She was diagnosed with ifosfamide-induced encephalopathy, and her chemotherapy was discontinued immediately. Her neurologic symptoms and signs did not improve. Despite the supportive care, her encephalopathy became aggravated, and she developed ifosfamide-induced acute tubular necrosis. She was discharged without any hope for recovery and died within 1 month.
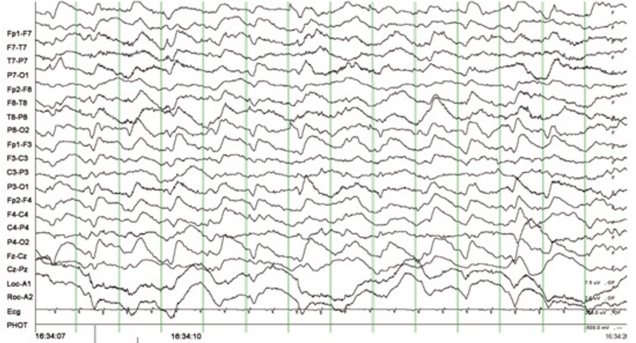 | Fig. 1Electroencephalogram (EEG) recording in case 1, showing frequent diffuse 1 Hz periodic triphasic waves and rhythmic delta slowing (EEG recording parameters: amplitude, 1 cm=70 µV; interval between two thick vertical lines=1 sec). 
|
2. Case 2
A 46-year-old, gravida 2, para 2, married woman with recurrent epithelial ovarian cancer (serous adenocarcinoma) was referred to our institution for further management. She had no specific medical history.
At initial diagnosis of epithelial ovarian cancer, she underwent bilateral salpingo-oophorectomy, partial omentectomy and incidental appendectomy at another hospital, and cytoreductive surgery was suboptimal. She received 12 cycles of adjuvant chemotherapy with paclitaxel and carboplatin. However, an abdomino-pelvic CT scan showed residual peritoneal carcinomatosis and her serum CA-125 concentration was elevated, from 517 to 1,020 U/mL. After referral to our center, she underwent optimal cytoreductive surgery, including total abdominal hysterectomy, pelvic and paraaortic lymph node dissection, low anterior resection, and right diaphragmatic stripping. After surgery, she received 3 cycles of adjuvant chemotherapy with topotecan and cisplatin. Due to disease progression, her regimen was switched to 3 cycles of docetaxel and carboplatin. Due to the disease progression on this regimen, we changed her chemotherapy regimen to a combination of ifosfamide and etoposide. She received the first cycle of etoposide (75 mg/m2/day in 3-hour infusions on days 1-5) and ifosfamide (1,200 mg/m2/day in 3-hour infusions on days 1-5), along with 300 mg/m2/day mesna, without any complications. On day 6, her speech became unintelligible but her orientation was intact. On day 7, she developed delirium, disorientation, visual and auditory hallucinations, stereotyped movements, and emotional instability. At that time, her estimated glomerular filtration rate, serum creatinine and blood urea nitrogen were 27 mL/min (normal, >60 mL/min), 2.3 mg/dL (normal, 0.7 to 1.4 mg/dL) and 33 mg/dL (normal, 10 to 26 mg/dL), respectively, and her renal function decreased gradually. Brain MRI was normal. After referral to a nephrologist, her electrolyte imbalance was corrected and her uremia was improved by hemodialysis. However, her mental impairments showed no improvement. She was diagnosed as having severe ifosfamide-induced encephalopathy. She never recovered and was discharged in a hopeless condition on day 10 and died within 1 month.
Go to :

Discussion
Ifosfamide is an oxazaphosphorine antineoplastic agent (a nitrogen mustard derivative) and is a structural analog of cyclophosphamide [
3,
7]. Ifosfamide is a prodrug that requires hepatic activation to its cytotoxic metabolite, ifosfamide mustard [
7,
8]. The latter is metabolized by cytochrome p450 to generate the active alkylating agents, 4-hydroxy-ifosfamide and isophosphoramide mustard [
8]. Ifosfamide and its metabolites can penetrate the blood brain barrier (BBB), with CNS toxicity occurring in 10-40% of patients receiving high doses of the drug [
2-
6].
The exact pathophysiological mechanisms responsible for the development of ifosfamide-induced encephalopathy are unclear, but one of the causes is thought to be the dechloroethylation of Ifosfamide to form chloroacetaldehyde (CAA) [
8-
10]. Hypotheses for the possible pathophysiological pathways leading to the development of ifosfamide-induced encephalopathy [
2,
11,
12] were based on the finding of glutaric acid and sarcosine in the urine of a patient with ifosfamide-induced encephalopathy (
Fig. 2) [
11,
13]. The accumulation of glutaric acid may lead to an increase in chloroethyl amine, the metabolism of which may in turn disturb the mitochondrial respiratory chain. Thus, the accumulation of nicotinamide adenine dinucleotide hydrogen (NADH) may prevent the dehydrogenation of aldehydes such as the ifosfamide metabolite CAA, a potentially neurotoxic substance [
9]. CAA, which is structurally similar to both chloral hydrate and acetaldehyde, can cross the BBB and therefore be responsible for ifosfamide-induced encephalopathy [
7]. The neurotoxic effects of CAA may be due to 1) a direct neurotoxic effect, 2) depletion from the CNS of glutathione, and/or 3) the inhibition of mitochondrial oxidative phosphorylation resulting in impaired fatty acid metabolism [
2].
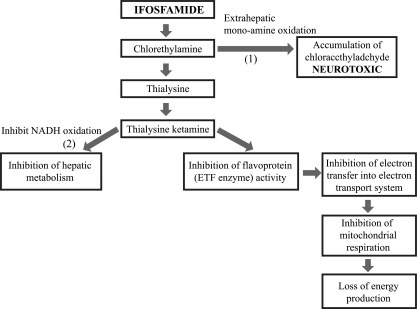 | Fig. 2Proposed pathogenesis pathways involved in ifosfamide neurotoxicity and the proposed sites of action of methylene blue: (1) Inhibition of extrahepatic mono-amine oxidation of chloroethylamine to chloroacetaldehyde; (2) Restoration of hepatic nicotinamide adenine dinucleomide hydrogen (NADH) oxidative functions; (3) Substitute for electron transport flavoprotein (ETF) enzyme (electron acceptor) (ada-pted from Donegan S. J Oncol Pharm Pract. 2001; 6:153-65 [ 13], with permission). 
|
CNS toxicity displays a wide spectrum of signs and symptoms. Manifestations of ifosfamide-induced encephalopathy include cerebellar ataxia, mental confusion, complex visual hallucinations, extrapyramidal signs, seizures and/or mutism. Less common CNS manifestations of ifosfamide are asterixis, nonconvulsive status epilepticus, manic episodes and cerebellar and temporo-frontal cortical degeneration [
6].
According to the National Cancer Institute Toxicity Grading for Encephalopathy [
14], there are five grades of encephalopathy. Most patients with ifosfamide-induced encephalopathy show grade 1 or 2 symptoms. Many patients with ifosfamide-induced encephalopathy recover spontaneously and completely within 48-72 hours following the onset of discontinuation of ifosfamide and conservative management such as adequate hydration. Some patients with ifosfamide-induced encephalopathy present with grade 3 or 4 symptoms. However, most patients with grade 3 or 4 symptoms can also recover spontaneously and completely. In rare cases, grade 3 or 4 encephalopathy leads to death. Because ifosfamide-induced encephalopathy research is limited to case reports and small retrospective case series, the prognosis and sequelae according to the toxicity grade, has not been clearly established [
10].
Although factors predisposing to ifosfamide-induced encephalopathy have not yet been revealed, rates of neurotoxicity may be increased by pre-existing renal or hepatic failure, low albumin and poor performance status [
4,
15]. These two cases were considered as grade 4 ifosfamide-induced encephalopathy according to the National Cancer Institute Toxicity Grading for Encephalopathy [
14] and both resulted in death within 1 month. In case 1, the electroencephalogram (EEG) finding was also revealed as Meanwell grade IV [
5]. Both patients' clinical courses may have been aggravated by their decreased renal function [
10].
Ifosfamide-induced encephalopathy is a clinical diagnosis. The differential diagnoses of ifosfamide-induced encephalopathy include infection, metabolic abnormalities and concomitant drug-induced syndromes. It is supported by normal brain imaging, EEG findings of metabolic encephalopathy and the absence of other causes [
6]. EEG characteristics have been correlated with the clinical grading of ifosfamide-induced encephalopathy [
5]. For example, Meanwell grade 3 toxicity has been associated predominantly with delta wave activity, with or without sharp complex waveforms, whereas patients with grade 4 toxicity exhibit continuous delta activity, complex waveforms and triphasic waves. The EEG recording in case 1 showed evidence of grade 4 toxicity (
Fig. 1).
In patients experiencing acute ifosfamide-induced encephalopathy, the mainstays of management include the prompt cessation of ifosfamide infusion and intravenous hydration. Although ifosfamide-induced encephalopathy usually reverses completely within 2-3 days after the cessation of drug administration, it may not in some patients with high grade toxicity.
Because ifosfamide-induced encephalopathy can resolve spontaneously 48-72 hours following its onset, patients with mild symptoms may not need treatment with MB. Patients with more severe symptoms and with grade 3-4 toxicity may derive greater benefit from MB. However, as the encephalopathy resolves spontaneously in most cases, even in patients with severe symptoms, and since controlled clinical trials have not been conducted to determine the efficacy of MB in severe ifosfamide-induced encephalopathy, the evidence to support its use is not strong [
10]. In most cases, MB is considered for use in the management of acute symptoms [
12]. While some patients have experienced improvements in CNS function as rapidly as within 10 minutes, others have required several days to recover [
10].
MB has been increasingly used both for the treatment and prevention of ifosfamide-induced encephalopathy [
8,
11]. MB is thought to counteract some of the metabolic pathways of ifosfamide [
11]. It may act as an alternative electron acceptor, replacing the inhibited flavoproteins and thus restoring the mitochondrial respiratory chain. It may also oxidize NADH, allowing the dehydrogenation of aldehydes. MB has also been found to inhibit plasma and extrahepatic monoamine oxidases (
Fig. 2) [
3,
8,
13]. The recommended dose of intravenous MB for treatment of ifosfamide-induced encephalopathy is 50 mg every 4 hours (1% aqueous solution over 5 minutes), whereas the dose for secondary prophylaxis of ifosfamide-induced encephalopathy is 50 mg every 6 hours, either intravenously or orally [
8]. However, the efficacy of MB in patients with severe ifosfamide-induced encephalopathy is still controversial.
Go to :






 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download