This article has been
cited by other articles in ScienceCentral.
Abstract
Purpose
We evaluated clinical outcomes after drainage for malignant pericardial effusion with imminent or overt tamponade.
Materials and Methods
Between August 2001 and June 2007, 100 patients underwent pericardiocentesis for malignant pericardial effusion. Adequate follow-up information on the recurrence of pericardial effusion and survival status was available for 98 patients.
Results
Recurrence of effusion occurred in 30 patients (31%), all of whom were diagnosed with adenocarcinoma. Multivariate analysis indicated that adenocarcinoma of the lung (hazard ratio [HR], 6.6; 95% confidence interval [CI], 1.9 to 22.3; p=0.003) and progressive disease despite chemotherapy (HR, 4.3; 95% CI, 1.6 to 12.0; p=0.005) were independent predictors of recurrence. Survival rates three months after pericardiocentesis differed significantly with the type of primary cancer; the rates were 73%, 18%, 90% and 30% in patients with adenocarcinoma of the lung, squamous cell carcinoma of the lung, breast cancer and other cancers, respectively.
Conclusion
Recurrence and survival of patients with malignant pericardial effusion are dependent on the type of primary cancer and response to chemotherapy. Patients with adenocarcinoma of the lung may be good candidates for surgical drainage to avoid repeated pericardiocentesis, but pericardiocentesis is considered effective as palliative management in patients with other cancers.
Go to :

Keywords: Neoplasms, Pericardial effusion, Pericardiocentesis, Prognosis
Introduction
Malignant pericardial effusion is a serious manifestation of advanced malignancies. A large autopsy series has reported that 8.1% of patients with malignancy had pericardial involvement and 2.7% experienced pericardial effusion (
1). Malignant pericardial effusion can lead to various adverse events including, chest discomfort, dyspnea, tachycardia, hypotension and cardiogenic shock, although some patients remain undiagnosed until death. Because the amount of pericardial fluid can only be semi-quantitatively estimated through imaging modalities, it is difficult to evaluate the relationship between pericardial fluid volume and its affect on the heart.
The most effective method of managing malignant pericardial effusion with imminent or recurrent tamponade has not been determined, although repeated pericardiocentesis (PCC) and surgical drainage (pericardiostomy or pericardial window operation) have been widely used. Pericardial instillation of anti-cancer drugs has also been recommended (
2-
6). The selection of a management plan for these patients depends on the patient's expected survival time and the probability of recurrence. Patients with pericardial effusion with imminent or recurrent tamponade have shown poor survival outcomes (
7-
9). Therefore, pericardial effusion in cancer patients has been considered a very grave sign. However, recent improvement in systemic treatment, including chemotherapy and molecular-targeted therapy, has enabled us to re-evaluate clinical outcomes for patients with malignant pericardial effusion. In addition, little is known about recurrence and survival after drainage, although a report has described practice patterns and outcomes for all types of pericardial effusion (
10). In contrast to malignant pericardial effusion, other kinds of effusion, including those caused by surgery, infection and connective tissue disease are relatively uncommon, rarely recur, and can be relatively easily treated. Recent improvements in systemic treatment, including chemotherapy and molecular-targeted therapy, have allowed a re-evaluation of clinical outcomes for patients with malignant pericardial effusion. We have therefore investigated clinical outcomes after drainage for malignant pericardial effusion with imminent or overt tamponade.
Go to :

Materials and Methods
1. Patient population
We retrospectively identified 100 consecutive patients who underwent successful PCC between August 2001 and June 2007 as initial treatment for hemodynamically significant or symptomatic malignant pericardial effusion. Hemodynamic significance was defined as diastolic right ventricular outflow collapse and exaggerated right atrial collapse during atrial systole (
11). Of the 100 patients, two were lost to follow up after PCC; thus, 98 were included in this analysis. Malignant pericardial effusion was defined as the presence of malignant cells in pericardial effusion or the drainage of exudate effusion in patients with malignancy, in the absence of other causes, such as post-operative effusion or tuberculosis. Patients were classified by primary cancer type into four categories: adenocarcinoma of the lung (ADL-L), squamous cell carcinoma of the lung (SQ-L), breast cancer, and other cancers.
Clinical characteristics and the cytology and biochemistry of effusion fluid were evaluated. The study end points were recurrence of pericardial effusion after PCC and death from any cause. Recurrence was defined as a reaccumulation of pericardial effusion requiring another pericardial procedure, either PCC, pericardial window operation or surgical pericardiostomy. Information on patient vital status and survival was collected from medical records and death certificate data from the Korea National Statistical Office. We calculated the interval between the first PCC and the next drainage of pericardial effusion. Except for one patient who had adenocarcinoma of lung (ADC-L) and poor performance status, all patients received chemotherapy after PCC. Chemotherapy responses were assessed after two or three treatment cycles using Response Evaluation Criteria in Solid Tumors (RECIST) criteria (
12).
2. PCC procedures
All PCCs were performed in the medical intensive care unit under the guidance of two-dimensional echocardiography. The point of minimal distance between the skin and maximal fluid accumulation was chosen as an entry site. After insertion of a catheter (18F), using the standard Seldinger technique, pericardial effusion was aspirated as much as possible on site and the drainage catheter was allowed to remain in place. Successful PCC, defined as adequate catheter positioning and effective drainage, was verified by echocardiography and chest radiography. The catheter position was confirmed by injection of agitated saline contrast, if necessary. Subsequently, the effusion was drained naturally (without negative pressure) for two days, and the catheter was removed when there was no significant pericardial effusion by echocardiography. No chemical agent was instilled.
3. Statistical analysis
Categorical variables, presented as counts and percentages, were compared using the chi-square test for equality of proportions. Continuous variables were presented as mean±standard deviation and range. Recurrence-free survival and overall survival rates were assessed using the Kaplan-Meier method and between group differences in survival curves were assessed using the log-rank test. Mean follow-up time was calculated among censored observations only. Predictors of recurrence-free survival and overall survival were investigated by fitting univariate and multivariable Cox regression models. Variables, including age, sex, primary cancer type, response to subsequent chemotherapy, history of radiotherapy (before and after PCC), signs of tamponade, drained effusion volume and results of effusion analysis, were entered into a backward and forward stepwise Cox proportional-hazards model to identify predictors of recurrence. Variables with a probability value <0.2 were retained. All p-values were two sided and p<0.05 was considered statistically significant. Stata/SE 10.0 for Windows (Stata Corp LP, College Station, TX) was used for all statistical analyses.
Go to :

Results
1. Patient characteristics
Patient baseline demographic and clinical characteristics are listed in
Table 1. Fifty seven patients had ADC-L, 11 had SQ-L, 10 had breast cancer, and 20 had other kinds of cancer; including 3 with small cell carcinoma of the lung, 2 each with unspecified non-small cell lung cancer, uterine cancer, stomach cancer, and colon cancer, and 1 each with lymphoma, large cell carcinoma of the lung, mesothelioma, osteosarcoma, bladder cancer, prostate cancer, bile tract cancer, esophageal cancer and thymic cancer. Therefore, the predominant tumor type was lung cancer, present in 74 of 98 (76%) patients, including 57 with ADC-L, 11 with SQ-L, 3 with small cell lung cancer, 1 with large cell lung cancer and 2 with non-specified lung cancer. Malignant cells were observed in the pericardial effusion of 75 patients (77%). Although 23 patients (23%) were negative for malignant cells, exudate effusion was drained from the patients with malignancy and without other causes, such as post-operative effusion or tuberculosis. Therefore, these patients were regarded as having malignant pericardial effusion. According to RECIST criteria, 74 patients had progressive disease after first chemotherapy, 2 had complete response, 10 had partial response and 12 had stable disease. Of the 75 patients with lung cancer, 13 (17%) received targeted therapy. The time from diagnosis to first PCC also varied. Including the 15 patients (15%) who underwent PCC as the initial manifestation of cancer, the median time between cancer diagnosis and drainage of pericardial effusion was 10.2 months, and the mean volume of drained pericardial effusion was 1,148±494 mL (range, 370 to 2,300 mL). Most pericardial fluid samples (69/98, 70%) were grossly bloody. The mean effusion to serum ratio of red blood cell (RBC) concentration was 0.3±0.3 (range, 0 to 1.4) and was>0.5 in 17% (14/84) of patients. In 3/84 patients (3.6%), RBCs were more concentrated in the pericardial fluid than in the serum, but there was no evidence of PCC-related hemorrhage. Interestingly, effusion recurred only in patients with ADC-L or adenocarcinoma of other organs (prostate, stomach, and colon).
Table 1
Baseline characteristics according to the type of primary cancer

2. Predictors of recurrence of pericardial effusion
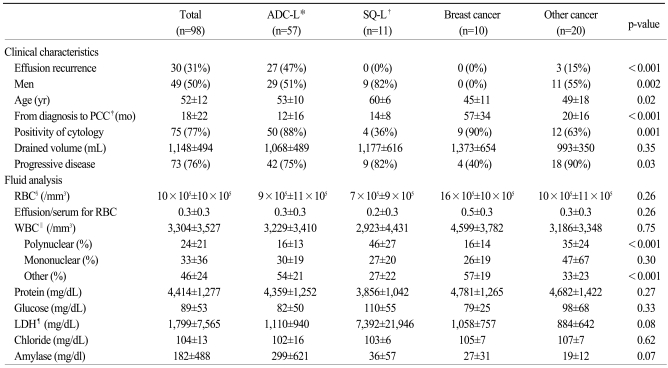
Pericardial effusions recurred in 30 patients (31%); of these, 12 underwent urgent surgical drainage, 11 underwent repeated PCC with subsequent pericardial window operations, and 7 underwent repeated PCC without subsequent pericardial window operations. The mean follow-up time in these 30 patients was 9.7 months. The cumulative probabilities of recurrence were 30% at 6 months and 52% at 12 months (
Fig. 1A). Recurrence was dependent on the type of primary cancer (
Fig. 1B) and response to chemotherapy (
Fig. 1C). Univariate analysis showed that ADC-L, progressive disease after chemotherapy, and a lower proportion of polynuclear cells in the effusion fluid were significantly associated with time to recurrence. Positive cytology and the effusion to serum ratio of RBC tended to show associations with recurrence, but these associations did not reach statistical significance (
Table 2). Although, some patients received radiotherapy before (19%) and/or after (8%) PCC, radiotherapy did not affect recurrence of pericardial effusion (
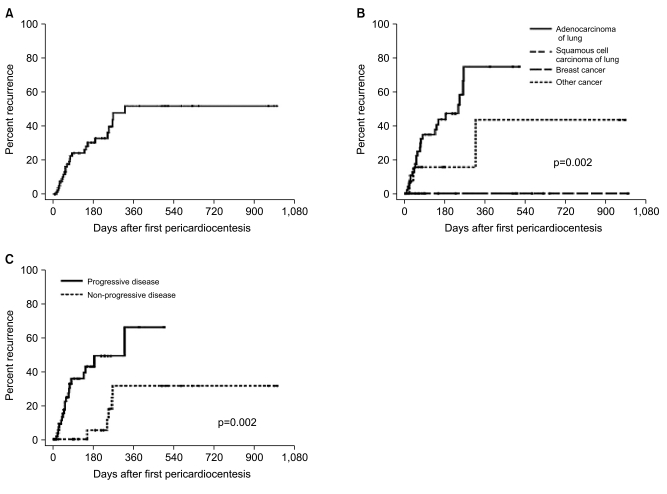
Table 2). In multivariable analysis, ADC-L (hazard ratio [HR], 6.6; 95% confidence interval [CI], 1.9 to 22.3; p=0.003) and progressive disease (HR, 4.3; 95% CI, 1.6 to 12.0; p=0.005) remained significant predictors for the recurrence of pericardial effusion. Among lung cancer patients, those with adenocarcinomas were vulnerable to recurrence of effusion, whereas those with squamous cell carcinoma were not.
 | Fig. 1Recurrences of malignant pericardial effusion. (A) Overall recurrence rates after first pericardiocentesis. (B) Recurrence rates in patients with different types of primary cancer. (C) Association between recurrence rate and response to chemotherapy. No recurrence of pericardial effusion was observed in patients with breast cancer and squamous cell carcinoma of the lung. Therefore, two lines were overlaid in (B). 
|
Table 2
Univariate and multivariable cox regression analyses of time to effusion recurrence

3. Survival after PCC
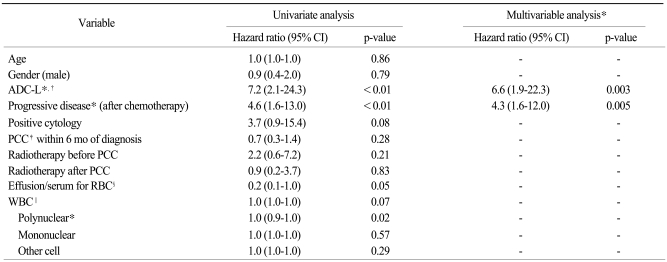
Median overall survival after PCC was 5.0 months. Deaths were observed in 89 patients (91%). The probability of overall survival was 45% at 6 months and 26% at 12 months (
Fig. 2A). The probability of overall survival differed significantly by the type of primary cancer and response to chemotherapy. Breast cancer patients showed significantly better overall survival than did other groups (vs. ADL-L, p=0.02; vs. SQ-L, p=0.002; vs. other cancers, p=0.02) (
Fig. 2B), which may have been due to the lower rate of progressive disease in breast cancer patients (4/10, 40%). Overall survival was better in patients with ADC-L than those with SQ-L or other cancers. Median survival after PCC was 6.8 months in patients with ADC-L, 1.9 months in patients with SQ-L, 16.5 months in patients with breast cancer, and 1.7 months in patients with other cancers. The probabilities of survival 3 months after PCC in these 4 groups were 73%, 18%, 80% and 30%, respectively. In addition, the survival rates 1 year after PCC were not so discouraging in patients with ADC-L (26%) and breast cancer (47%). Progressive disease after PCC was also significantly associated with overall survival time (
Fig. 2C).
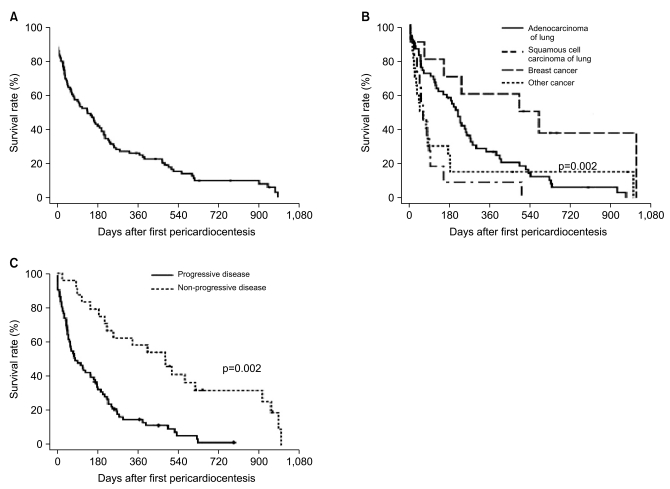 | Fig. 2Survival outcomes. (A) Overall survival rate after first pericardiocentesis. (B, C) Survival rates according to the type of primary cancer (B) and response to chemotherapy (C). 
|
Go to :

Discussion
PCC-related major complications, including chamber laceration, injury to intercostal vessels, pneumothorax, ventricular tachycardia and bacteremia have been reported to be rare (14/1,127, 1.2%), as has PCC-related mortality (1/1,127, 0.09%) (
10). PCC is considered safe for patients with significant pericardial effusion, although it requires experience and skill and is only a temporary resolution. Therefore, PCC is quite effective for patients with rarely recurrent pericardial effusion, including those with idiopathic effusion, heart failure, uremia, tuberculosis, myocardial infarction, connective tissue disease, or with pericardial effusion due to well controlled diseases such as hypothyroidism (
13). In contrast, recurrent malignant pericardial effusion is common, and the life expectancy of these patients is limited due to advanced cancer (
Table 3).
Table 3
Summary of the studies for clinical outcomes of malignant pericardial effusion
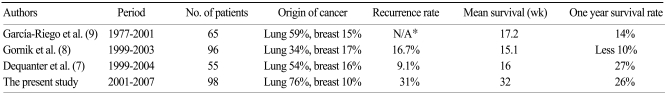

We found that the recurrence rate after PCC differed according to the type of primary cancer and response to chemotherapy. To our knowledge, the present study is the largest to date to assess long-term outcomes in patients with malignant pericardial effusion, and to show that long-term outcome depends on the type of primary cancer. Patients with malignant pericardial effusion from breast cancer showed longer survival times and lower recurrence rates than did other patients, suggesting that PCC may be an effective solution for patients with malignant pericardial effusion from breast cancer. Patients with malignant pericardial effusion from SQ-L had a poor survival rate (18% at 3 months) and there may be little chance of recurrence. PCC is also effective as palliative management in patients with such poor survival. However, patients with ADC-L had better survival rates than those with SQ-L or other cancers, and pericardial effusion recurred in many patients with ADC-L. ADC-L was significantly associated with both recurrence and higher survival rate, probably due to a better response to chemotherapy. Thus, patients with ADC-L may be good candidates for surgical drainage to avoid repeated PCC.
Although most surgical drainages are performed under general anesthesia, they are quite effective in avoiding the recurrence of pericardial effusion (
14). Another independent predictor of recurrence was progressive disease. Therefore, response to chemotherapy should be considered when selecting a therapeutic option for pericardial effusion. Malignant cells were more frequently observed in patient with recurrent effusions than those with non-recurrent effusion, but the difference was not statistically significant.
Abnormal cytology has been reported to predict poor prognosis (
8). In that study, however, cytology analyses were not performed for all patients, and the proportion of patients with lung cancer, which had the highest positive cytology rate, was lower than in our study. A lower proportion of polynuclear cells is indicative of higher proportions of other cells, including malignant cells, but these findings also did not correlate with recurrence. Thus, the correlation between recurrence and cytology requires further investigation.
The effusion to serum ratio of RBC concentration varied considerably and was even greater than in some patients without evidence of hemorrhage. Therefore, unlike patients with hemothorax, it may not be appropriate to differentiate among patients with pericardial fluid by their effusion to serum RBC ratio.
Go to :

Conclusion
The overall recurrence rate of malignant pericardial effusion was 31% and the median survival after first PCC was 5.0 months, although survival times varied considerably, from 0 to more than 10 years. Recurrences of pericardial effusion occurred exclusively in patients with adenocarcinoma (lung, prostate, stomach and colon), with ADC-L and progressive disease being significant predictors of recurrence. Median survival after first PCC in patients with ADC-L was -6.8 months, and this may be increased due to recent advances in chemotherapy. These findings highlight the importance of appropriate management of malignant pericardial effusion, along with active treatment of the primary cancer. Surgical interventions, such as pericardial window operation, may be warranted in selected patients, especially those with ADC-L, to prevent recurrent pericardial tamponade. Although this study is retrospective in design and involves patients from a single center, our findings suggest that the approach to malignant pericardial effusion should be individualized based on the type of primary cancer and patient response to chemotherapy.
Go to :






 PDF
PDF Citation
Citation Print
Print






 XML Download
XML Download