INTRODUCTION
Xerostomia is one of the most common toxicities after radiation therapy for treating head and neck cancer. Xerostomia predisposes patients to infection and it disturbs eating, speaking and sleeping (1~3). Moreover, three-dimensional conformal radiation therapy (3D-CRT) with using non-coplanar beams was developed to minimize the radiation dose to the parotid glands during RT (4,5).
Intensity modulated radiation therapy (IMRT) is now known to preserve the parotid glands and to diminish the incidence of xerostomia. However, the IMRT results show that xerostomia could develop to some degree following IMRT, and that the salivary function may show recovery at 12 months after IMRT (6~8). Previous studies on dose-response relationships and the effects of the volume of the irradiated glands on gland function have revealed that the mean parotid dose is the most important predictor for developing xerostomia (6~10).
A well-known study by Emami et al. (11) suggested employing TD 5/5 (the probability of a 5% complication rate within five years of treatment) doses of 32 Gy and TD 50/5 (the probability of a 50% complication rate within five years from treatment) doses of 46 Gy, respectively, for more than 2/3 parotid irradiation. However, these estimates of TD 5/5 and TD 50/5 were based on clinical experiences. In addition, others have attempted to determine the dose levels that preserve the salivary output after parotid sparing 3D-CRT and IMRT (4~8).
This study was undertaken to evaluate the changes in the xerostomia questionnaire score (XQS), the LENT/SOMA scale (LSS), the unstimulated salivary flow rate (USFR), and the stimulated salivary flow rate (SSFR) after IMRT, and we wanted to identify the optimal parotid dose that's required to substantially preserve the salivary function relative to the pretreatment baseline function.
Go to : 
MATERIALS AND METHODS
The study subjects consisted of 34 patients who suffered with head and neck cancer and who were treated with IMRT from February 2003 through to June 2004. All the patients had an Eastern Cooperative Oncology Group (ECOG) performance status scale (12) score of 0~2. All the patients signed an informed consent and this study was approved by the institutional review board of National Cancer Center. The clinical evaluation of the patients included physical examinations, chest X-rays, head and neck computed tomography (CT) scans, head and neck magnetic resonance imaging (MRI) and laryngoscopy. All 34 patients received primary or postoperative IMRT that was planned to spare the parotid gland. Among 14 patients who received primary treatment, 10 patients (29.4%) received concurrent IMRT and cisplatin-based chemotherapy for treating locally-advanced head and neck cancer.
1) Patient immobilization and CT simulation
Patients were placed in the supine position on a custom-made head and shoulder support device, i.e., the Jeonsung® fixation device. This device is a type of reinforced aquaplast face mask that minimizes movement to 2~3 mm. CT images were acquired using a CT simulator (Marconi AcQsim, Marconi Medical System, Inc., Cleveland, OH). The transverse images represented 4-mm thick slices from 5 cm superior to the skull base to the clavicle heads. The acquired images were directly transferred to a 3D planning system (AcQPlan version 4.2, Philips Medical Systems, Cleveland, OH).
2) Target definition and treatment planning
Treatment of the bilateral neck was indicated for all the patients. The targets were then delineated after first delineating the normal structures. The targets in all patients were defined in accordance with the ICRU 50 guidelines (International Commission on Radiation Units and Measurements Report). The gross tumor volume (GTV) encompassed all detectable tumors and lymph nodes that were less than <0.5 cm in short axis diameter as determined from the CT or magnetic resonance (MR) images. The clinical target volume (CTV) encompassed the GTV and regional lymphatics, including those areas considered to harbor potential microscopic disease. The bilateral jugular chain (Level II~IV) lymph nodes were included in most cases. Due to considerations for the treatment setup and the patients' weight loss, the planned target volumes (PTVs) included the CTVs and a margin (usually 5~10 mm).
The patients were treated with a 6 megavolt linear accelerator. By employing the sliding window technique, IMRT was planned using Helios (6.3.6 version, Varian Medical Systems, Palo Alto). For the definitive radiation therapy, the prescribed doses were about 64.8 Gy to the PTVs, and they were treated by accelerated fractionation (2.4 Gy/fraction). For the postoperative irradiation, about 60 Gy was prescribed to the PTVs, and this was administered by conventional fractionation (2 Gy/fraction). The dose limits of the normal tissues were set as follows; brain stem: 54 Gy, optic nerve/chiasm: 54 Gy, spinal cord: 45 Gy, and the total parotid gland: 35 Gy. Generally, 5 or 7 beams were required to cover the PTV and avoid the normal tissue. The dose-volume histograms (DVH) were analyzed for determining the coverage of the PTV and the normal tissues. The mean radiation dose to each gland was calculated by summing the doses to the voxels that comprised each gland volume and by dividing the sum by the number of voxels (voxel size: 3×3 mm). The total parotid mean dose was defined by dividing the sum of the mean doses to the parotid glands by two. We optimized the doses using the superficial part of the parotid, and after optimization, we outlined the whole parotid as a separate structure to determine its DVH. Ten patients with locally advanced head and neck cancer were treated with concurrent IMRT and their chemotherapy consisted of intravenous cisplatin at 100 mg/m2/day; it was infused over 8 hours on days 1 to 4 during the first and fifth week of radiation therapy. Depending on the patient's tolerability and clinical assessment of toxicity, a third cycle of chemotherapy was planned 4 to 5 weeks after the second cycle, upon completion of the radiation therapy.
3) Evaluation of the xerostomia parameters
Xerostomia was evaluated using a questionnaire and by conducting salivary flow measurements. These evaluations were conducted before IMRT (pre-IMRT) and at 1, 3 and 6 months after IMRT.
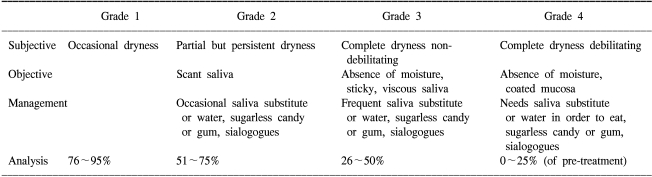
To evaluate the subjective symptoms, we employed a xerostomia questionnaire that consisted of four questions related to the patients' reported dryness while communicating, eating and sleeping and during normal periods (Table 1). The xerostomia questionnaire score (XQS) were determined by summing the scores of the 4 questions.
As objective parameters, the salivary flow rates (USFR and SSFR) for each patient were measured at the time of administering the questionnaires. The patients refrained from eating, drinking and anything else that would stimulate the parotid gland for a minimum of 90 minutes prior to salivary collection. Unstimulated saliva was collected first, and then stimulated saliva was measured by applying 2% citric acid to the dorsolateral tongue surface at 30 second intervals for 120 seconds. The unstimulated and stimulated saliva were converted to flow rates (ml/min) and these were recorded by assuming a specific gravity of 1.0. To control for different salivary flow rates among different individuals, all the subjects' flow rates were converted to unstimulated/stimulated flow rate ratios at baseline.
The LENT SOMA scale (LSS) was also administered at the time of the above measurements to reflect the subjective and objective symptoms (13). The LSS consisted of 4 domains (subjective, objective, management and analysis) and it was graded by the investigator (Table 2). The LENT SOMA scale (LSS) was also determined by summing the grades of its 4 domains. For both the XQS and LSS, the patients were asked to respond to a questionnaire with respect to the extent to which they experienced problems with the listed items. The mean XQS and mean LSS of all 34 patients at each measurement time point were analyzed. Higher scores mean higher levels of xerostomia symptoms. We defined the parotid tolerance dose as the total parotid mean dose at all times during follow-up that showed significant XQS, LSS and salivary flow rate preservation versus the pretreatment baseline values.
4) Statistical analysis
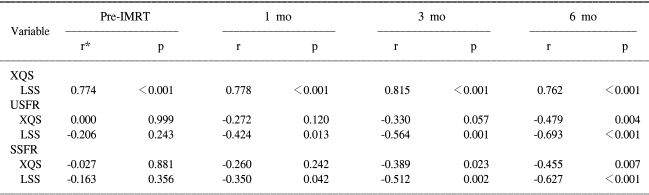
The changes in the scores and salivary flow rates between the subgroups were tested using the Mann-Whitney test. The changes at follow-up times and baseline were illustrated graphically and they were assessed using the Wilcoxon signed rank test. Correlations between the XQS, LSS and salivary flow rates were evaluated and analyzed by using Pearson correlation coefficients (r). All statistical tests were two-sided and statistical significance was accepted for p-values ≤0.05.
Go to : 
RESULTS
All 34 patients received comprehensive IMRT for their head and neck cancer. We tested the changes in the scores and salivary flow rates between the subgroups and we founded that the patients' age (≥60 or <60), gender, tumor site, stage, chemotherapy (yes or no) and surgery (yes or no) did not show any significant difference (data not shown). The study group was composed of 23 men and 11 women, and their ages ranged from 29 to 78 years, with a mean (±standard deviation) age of 58.5 (±11.5 years). Twenty patients received postoperative irradiation, and the other 14 patients underwent definitive radiation therapy. The patient, tumor and treatment characteristics are shown in Table 3.
1) Xerostomia questionnaire (XQS and LSS) results
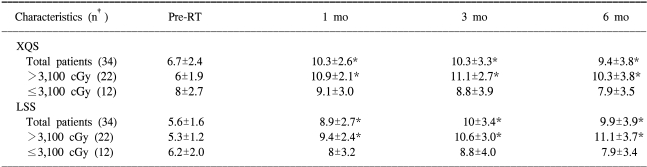
When the total parotid mean dose was not taken into account, the mean XQS and LSS for all 34 patients showed significant changes at 1, 3 and 6 months after IMRT compared to baseline. However, when we analyzed the patients after dividing the 34 patients into two groups according to the total parotid mean dose, no significant differences between the XQS baseline values at 1 (p=0.326), 3 (p=0.663), and 6 (p=0.592) months after IMRT were found for the 12 patients who received a total parotid mean dose of ≤3,100 cGy. Yet the 22 patients that received >3,100 cGy showed significant increases of the XQS at 1 (p<0.001), 3 (p<0.001), and 6 (p=0.001) months after IMRT. The increases in the LSS from baseline to each time point (1 (p<0.001), 3 (p<0.001), and 6 (p<0.001) months) after IMRT were also significant for the 22 patients who received a total parotid mean dose of >3,100 cGy. However, no significant increase in the LSS at any follow-up time point (1 (p=0.125), 3 (p=0.113), and 6 (p=0.220) months) after IMRT was observed for the 12 patients who received ≤3,100 cGy. The relation between the mean XQS, LSS and time is shown in Table 4.
2) Salivary flow rates (USFR and SSFR)
Saliva flow rate measurements were taken for up to 6 months after completing radiation therapy for all the patients. The median unstimulated and stimulated salivary flow rates at pre-IMRT were 0.159 ml/min (mean: 0.243 ml/min, standard deviation: 0.272 ml/min) and 0.175 ml/min (mean: 0.237 ml/ min, standard deviation: 0.211 ml/min), respectively, and these diminished after initiating IMRT. To represent the saliva changes in a graphic form, the flow rates were converted into flow ratios based on the pre-IMRT salivary flow rates. At 6 month after IMRT, the mean salivary flow ratios were 0.604 of the baseline when they were unstimulated, and 0.503 when they were stimulated for the 34 patients; at 1, 3, and 6 months after IMRT, the mean salivary flow ratios of the 34 patients were significantly lower than the baseline values (Fig. 1, 2). The salivary flow ratios of 7 patients that received less than the total parotid mean dose, i.e., 2,750 cGy, were significantly preserved at 6 months after IMRT. For these patients, the mean salivary flow ratios were 0.725 when unstimulated, and 0.735 when stimulated at 6 months, and no significant differences were observed between the baseline values and the 6 month values in these patients. However, the salivary flow ratios in the 27 patients who received >2,750 cGy were significantly diminished after IMRT and at 6 months post IMRT, and the mean values were statistically distinguishable from the baseline values (Fig. 1, 2). At 1 month after IMRT, the unstimulated (p=0.046) and stimulated (p=0.019) salivary flow ratios were significantly lower than those at baseline, and this remained true at 3 months (unstimulated, p<0.001; stimulated, p<0.001) and at 6 months (unstimulated, p<0.001; stimulated, p<0.001). For 27 patients at 6 months after IMRT, the mean salivary flow ratios were 0.463 unstimulated, and 0.367 stimulated. Moreover, these flow ratio patterns were similar for both the USFR and SSFR.
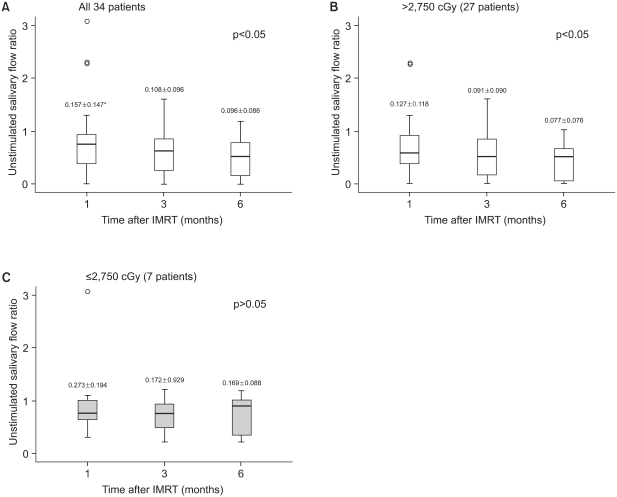 | Fig. 1Changes in the unstimulated salivary flow ratio with time. (A) All patients, (B) the total parotid mean dose >2,750 cGy, and (C) the total parotid mean dose ≤2,750 cGy. The median is denoted by a solid line. Ratios were obtained by dividing the measured USFR at each time point by its pretreatment (baseline) value. *unstimulated salivary flow rate (mean±standard deviation, ml/min) |
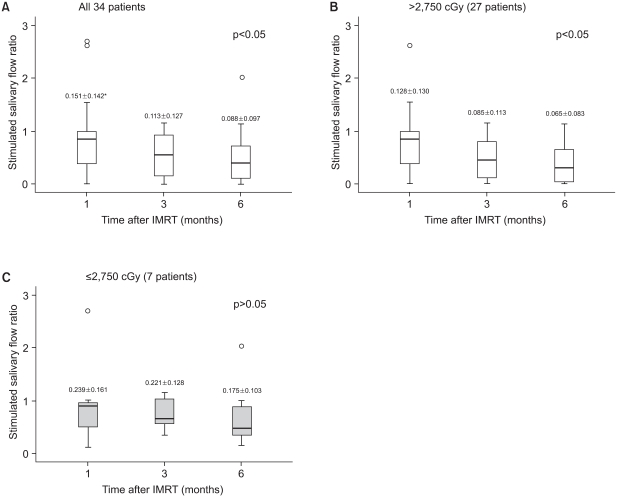 | Fig. 2Changes in the stimulated salivary flow ratio with time. (A) All patients, (B) the total parotid mean dose >2,750 cGy, (C) the total parotid mean dose ≤2,750 cGy. The median is denoted by a solid line. Ratios were calculated by dividing the estimated SSFR at each time point by its pretreatment value. *stimulated salivary flow rate (mean±standard deviation, ml/min). |
3) Correlations between the XQS, LSC and Salivary flow rates (USFR and SSFR)
The correlation between the XQS and LSS are shown in Table 5. Pearson's correlation analysis revealed that the XQS were significantly correlated with the LSS at 1, 3 and 6 months post-IMRT. In terms of the relation between the XQS and the salivary flow rates, a significant correlation was observed at 6 months after IMRT between the XQS and USFR, and at 3 and 6 months between the XQS and SSFR. At all the follow-up points after IMRT, the LSS and salivary flow rate values were found to be significantly correlated.
Go to : 
DISCUSSION
The results obtained in the present study indicate that in the case of IMRT, the total parotid mean dose is the most significant predictor for the development of xerostomia, so this can be used as a planning guideline to preserve salivary function. The validated patient-reported xerostomia questionnaire of the University of Michigan (14) was the basis of our xerostomia questionnaire (Table 1): this consisted of four questions concerning the patient's reported mouth dryness while communicating, eating, sleeping and during normal periods, and so this was used in our study to evaluate the subjective symptoms.
A review of the retrospective results concerning the parotid's radiation tolerance revealed that the salivary flow rates in the patients who received 30~45 Gy substantially recovered at 6 and 18 months after radiation therapy, whereas irreversible salivary function losses were observed in the patients that received >65 Gy (6,7,11,13,15,16). However, the patients were not treated by 3D-CRT in the majority of the previous studies. Therefore, these studies were limited in terms of their abilities to analyze parotid volumes in two dimensions because they employed only orthogonal planar radiographs and these do not provide dose volume information. With the advent of 3D-CRT, targets and surrounding normal organs can be accurately delineated, and accordingly, the dose-volume relations can be determined. Furthermore, the introduction of IMRT makes the preservation of normal organs such as the parotid glands more feasible than does 3D-CRT. Eisbruch et al. (6) enrolled 88 patients with head and neck cancer who were irradiated with 3D-CRT and IMRT, and Eisbruch reported that the total parotid gland mean dose should be limited to <26 Gy, if substantial sparing of the gland function is required. In that study, they selectively measured the salivary flow rates from each parotid gland before and periodically after RT, and they identified the entire parotid volumes to compute the DVHs for analysis. They reported that glands that had received a mean dose equal or lower to the threshold dose 26 Gy showed a tendency to increase saliva output over time after RT. Yet the majority of glands that received a higher dose showed a reduced saliva output at the first month, and there was no recovery over time, regardless of the dose level. Eisbruch surmised that the dose threshold with respect to saliva stimulation appears to be more clinically important than the threshold for unstimulated secretion for the major role of the parotid glands, i.e., saliva secretion after stimulation by food.
Chao et al. (7) reported that the stimulated saliva flow at 6 months after treatment was reduced exponentially, for each gland, at a rate of approximately 4% per Gy of the mean parotid dose. This implies that approximately 50% or more of the baseline saliva flow can be retained if both parotid glands receive a mean dose of <16 Gy. If both parotid glands receive a mean dose of 32 Gy, the model predicts that stimulated saliva levels would be 25% of the pretreatment values, which is equivalent to Grade 4 xerostomia according to the LENT SOMA scale. They also used the entire parotid volume to compute the DVH, as we did in the present study. Although they attempted to spare the superficial lobe of the parotid glands to avoid underdosing those tumor targets in the parapharyngeal space, they reported that the responses to quality of life (QOL) questions on eating/speaking function were significantly correlated with the stimulated and unstimulated saliva flow at 6 month. These results are in close agreement with the results of the present study, which found that the XQS and LSS are significantly correlated with the SSFR and USFR at various times after IMRT. These results indicate that evaluation of xerostomia by using the XQS, LSS, SSFR and USFR is efficacious and reasonable.
Roesink et al. (17) reported tolerated parotid doses that were somewhat higher than those reported by others researchers. They used a normal tissue complication probability (NTCP) model (18) to identify the threshold in 108 patients who were treated by 3D-CRT, and they regarded a post-treatment parotid flow ratio of <25% as a complication, according to their data and the RTOG/EORTC Late Effects Consensus Conference (19). Their results revealed a linear correlation between the post-radiotherapy flow ratio and the parotid gland dose, and there was a strong relation between the dose and saliva volume. They concluded that the mean parotid gland dose should be <39 Gy for radiation planning, which was associated with a complication probability of 50%.
A recently published study by Munter et al. (20) was performed to evaluate the salivary gland function after IMRT for head and neck tumors. However, this study used quantitive scintigraphy to quantify the functions of individual salivary glands before and after RT, and it didn't use quantitative saliva flow measurement techniques, which have generally been used in clinical studies. In that study, the parotid glands and the submandibular glands were delineated on CT images for each patient. They found that the relative saliva excretion rates fell significantly when the parotid glands received a mean dose of 26 Gy. In terms of changes in the maximal activity uptake, a statistically significant difference between before and after treatment was observed at a dose threshold of 30 Gy. Thus, they concluded that doses <30 Gy significantly preserved salivary gland function. Saarilahti et al. (8) recently reported a D50 (the 50% complication probability dose) for an impaired stimulated parotid gland saliva flow rate of 25.5 Gy, and they concluded that a reduction in the cumulative dose (<26 Gy) administered to the parotid gland resulted in the preservation of both the basal (unstimulated) and stimulated flow rates.
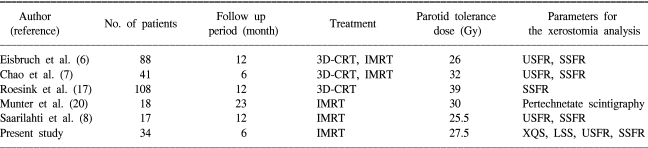
The results of the above studies are summarized in Table 6. However, direct comparisons and interpretations between different studies are somewhat difficult. First, different questionnaires were used to estimate xerostomia, and the follow-up periods differed as well. We used the XQS and LSS, and we measured salivary flow rates for the xerostomia analysis, whereas the majority of previous studies (6~8,17) predominantly used salivary flow rates to determine the tolerated parotid doses. Xerostomia is generally defined as the subjective complaint of a dry mouth, and this is only poorly correlated with objective salivary dysfunction findings (21,22). Second, the RT techniques, doses and fractionation schemes that were used varied between the studies. In the present study, the dose and fractionation scheme we used involved the delivery of 60~64.8 Gy in 2.0~2.4 Gy fractions to targets with using a SMART (simultaneous accelerated radiation therapy) beam, which was relatively consistent. On the other hand, most of the previous studies (6,7,17) included patients that received 3D-CRT as well as IMRT, which caused fractionation scheme inconsistencies and made comparisons difficult. However, debate among physicians and researchers continues concerning the effect of fractionation on the parotid gland (23).
Our study also has its limitations. In the present study, no volume analysis was performed using the NTCP model, as was been done by several other studies (6~8,17), and the follow-up time was relatively short (6 months) (6,8,17,20). In our future work, we will focus on identifying the dose-volumetric parameters most associated with a risk of severe xerostomia in the patients with head and neck tumors that are treated by IMRT.
Go to : 
CONCLUSIONS
The salivary function questionnaire (XQS and LSS) results were significantly correlated with the mean dose administered to the parotid glands, and this demonstrates that dosimetric sparing of the parotid glands improves subjective xerostomia. The findings obtained from this study also suggest the dose dependency of the parotid gland function. We suggest that the total parotid mean dose should be limited to ≤2,750 cGy to preserve the USFR and SSFR and to improve the QOL after IMRT.
Go to : 




 PDF
PDF Citation
Citation Print
Print




 XML Download
XML Download