Abstract
Purpose
The benefit of consolidation high-dose chemotherapy (HDC) for high-risk primary breast cancer is controversial. We evaluated the efficacy and safety of consolidation HDC with cyclophosphamide, thiotepa and carboplatin (CTCb) followed by autologous stem-cell transplantation (ASCT) in resected breast cancer patients with 10 or more positive lymph nodes.
Materials and Methods
Between December 1994 and April 2000, 22 patients were enrolled. All patients received 2 to 6 cycles of adjuvant chemotherapy after surgery for breast cancer. The HDC regimen consisted of cyclophosphamide 1,500 mg/m2/day, thiotepa 125 mg/m2/day and carboplatin 200 mg/m2/day intravenous for 4 consecutive days.
Results
With a median follow-up of 58 months, 11 patients recurred and died. The median disease-free survival (DFS) and median overall survival (OS) were 49 and 69 months, respectively. The 5-year DFS and OS rates were 50% and 58%, respectively. The 12 patients with 10 to 18 involved nodes had better 5-year DFS (67%) and OS (75%) than 10 patients with more than 18 involved nodes (30% and 38%, respectively). The most common grade 3 or 4 nonhematologic toxicity was diarrhea, which occurred in 5 patients (23%). No treatment-related death was observed.
The prognosis of breast cancer patients with 10 or more positive lymph nodes is particularly poor. Adjuvant chemotherapy has led to an improvement in the outcome of these patients, but the recurrence rate at 5 years is still high (> 50%) (1~3).
There is a clear relationship between the dose intensity of adjuvant chemotherapy for breast cancer and the relapse-free survival (4). In addition, autologous stem-cell transplantation (ASCT) allows dose escalations within the myeloablative range, which is needed to overcome tumor cell resistance and produce better outcomes. During the 1990s, several nonrandomized studies reported extended survival rates after consolidation high-dose chemotherapy (HDC) with ASCT in breast cancer patients with 10 or more involved axillary lymph nodes (5,6). Therefore, HDC has become a popular treatment for high-risk patients (7), but a number of randomized studies reported to date have failed to show a significant survival benefit for HDC in this population (8~13).
In 1998, we reported the preliminary results of a phase II study of HDC with cyclophosphamide, thiotepa and carboplatin (CTCb) followed by ASCT for 11 patients with metastatic or high-risk breast cancers (14). This study was closed down in July 2000, due to the lack of evidence from randomized studies demonstrating any survival benefit from HDC (15,16). Recently, several randomized trials showed a trend in favor of HDC, with respect to disease-free survival (DFS), especially in selected patient subgroups (11~13).
We report the updated outcomes of 22 high-risk patients of the phase II study after a median follow-up of 58 months, with a maximum of 9 years.
Female patients were considered eligible if they had histologically proven primary breast cancer; 10 or more positive axillary lymph nodes; had undergone a mastectomy or breast-conserving operation with axillary lymph node dissection; had no distant metastases; were 16 to 65 years of age; had an Eastern Cooperative Oncology Group (ECOG) performance status of 0 to 2; and had adequate organ function (defined as leukocytes ≥ 4,000/µl, platelets ≥ 100,000/µl, transaminases ≤ 2 times the upper normal limit, total bilirubin ≤ 2.0 mg/dl, serum creatinine ≤ 2.0 mg/dl and a cardiac ejection fraction ≥ 50%). The study was approved by our Institutional Review Board, in accordance with the Helsinki Declaration, and the written informed consent was obtained from each patient.
All patients received 2 to 6 cycles of adjuvant chemotherapy. CAF (cyclophosphamide 600 mg/m2, doxorubicin 60 mg/m2, and 5-fluorouracil 600 mg/m2 every 3 weeks) was the preferred regimen used in 18 patients (82%). Three patients received single-agent doxorubicin (75 mg/m2 every 3 weeks), and one patient was treated with the MFL regimen (methotrexate 100 mg/m2, 5-fluorouracil 600 mg/m2, and leucovorin 60 mg on days 1 and 8 every 4 weeks). Following a breast-conserving operation, radiotherapy was administered to the breast and regional nodes.
Peripheral-blood stem cell mobilization was induced by chemotherapy plus granulocyte colony-stimulating factor (G-CSF, 10 µg/kg/day subcutaneously). The chemotherapy regimens were cyclophosphamide 4 g/m2 or CAF. The peripheral-blood stem cells were collected by leukapheresis until at least 3×106 CD34+cells/kg had been harvested (17).
The HDC regimen consisted of cyclophosphamide 1,500 mg/m2/day, thiotepa 125 mg/m2/day, and carboplatin 200 mg/m2/day intravenously on days -7 to -4. G-CSF was initiated the day after stem cell infusion and it was continued until the absolute neutrophil count reached 1,000/µl or more on 2 consecutive days.
All patients received prophylactic antimicrobials/antivirals consisting of oral ciprofloxacin and acyclovir. The patients underwent transfusions with irradiated red blood cells and platelets to maintain their hemoglobin levels and platelet counts at greater than 8 g/dl and 20,000/µl, respectively.
Patients were examined every 3 months, and annual mammography and chest roentgenography were performed. For symptomatic patients, a bone scan and liver ultrasonography were also checked. Recurrence was defined as local (operation scar or chest wall), regional (axillary or supraclavicular lymph nodes) or distant.
The DFS and overall survival (OS) were estimated using the Kaplan-Meier method and calculated from the date of stem cell infusion. The DFS was measured until the date of first recurrence or death from any cause, and the OS until the date of death from any cause. For patients not experiencing these events, their data were censored at the time of their last follow-up visit. The log-rank test was used to compare the survival times between the patient subgroups, and the Cox proportional hazards model was used to estimate the hazard ratios and to perform a regression analysis. p values of 0.05 or less were considered significant.
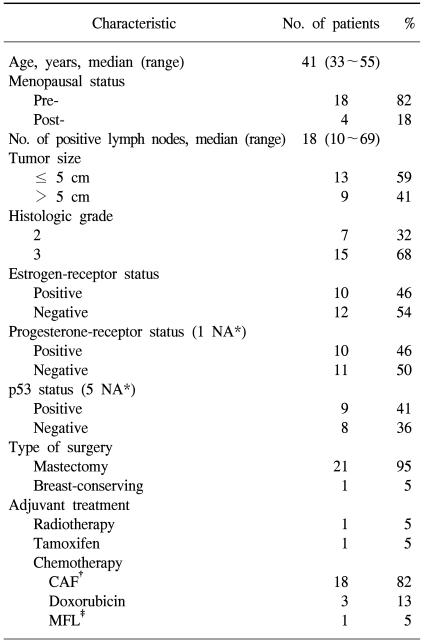
Between December 1994 and April 2000, 22 patients were enrolled. The patients' baseline clinical features are summarized in Table 1. The median age was 41 years (range, 33 to 55), and 82% were premenopausal. The hormone-receptors were positive in 46% of patients, and the median number of positive lymph nodes was 18 (range, 10 to 69).
A median 4 sessions of leukapheresis (range, 3 to 19) was required to obtain at least 3×106 CD34+ cells/kg (median yield, 5.4×106; range, 2.2×106 to 40.8×106). In 1 patient (5%), less than the target number of cells was harvested. The median times to neutrophil engraftment (≥ 500/µl) and platelet transfusion independence (≥ 20,000/µl) were 12 (range, 9 to 33) and 19 days (range, 8 to 45), respectively. There was 1 case of platelet engraftment failure. The median in-patient stay after stem-cell infusion was 26 days (range, 14 to 56).
The HDC was terminated early in 1 patient due to a seizure. All patients became transfusion-dependent and developed an episode of neutropenic fever requiring intravenous antibiotics. There were 5 cases (23%) of documented infection (urinary tract infection, central-line infection, sinusitis, cellulites and bacteremia). The most common grade 3 or 4 nonhematologic toxicity was diarrhea, which occurred in 5 patients (23%). Grade 3 mucositis occurred in one patient and grade 3 hepatotoxicity occurred in 3. There was 1 case of hemorrhagic cystitis, despite the concomitant administration of mesna. No treatment-related death was observed.
After the completion of HDC, radiotherapy was administered to 10 patients, and tamoxifen was given to 10 with a positive hormone-receptor tumor.
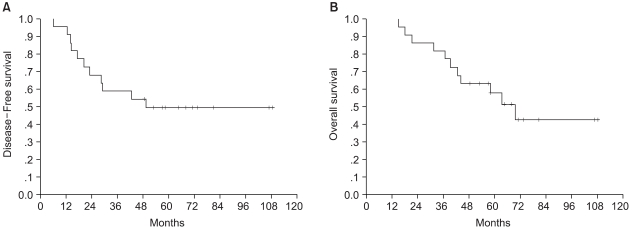
With a median follow-up of 58 months from the time of ASCT (range, 15 to 108), 11 patients recurred and died. Of these 11 recurrences, 2 were isolated locoregional and 9 were distant with or without simultaneous locoregional failure, as first event (bone in 3, lung in 2, pleura in 1, liver in 1 and brain in 2). The median durations of DFS and OS were 49 and 69 months (95% CI, 51 to 88), respectively (Fig. 1). The 5-year DFS and OS rates were 50±11% and 58±11%, respectively.
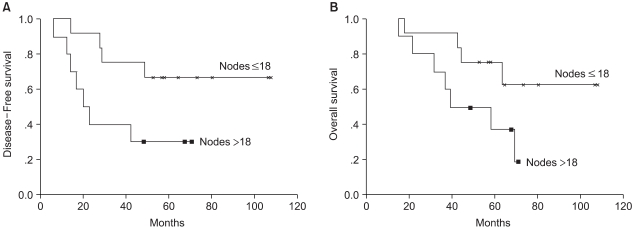
Univariate analysis was performed to determine the factors predictive of DFS and OS. Factors examined were: age (≤ 40 years vs. > 40 years), menopausal status, number of involved lymph nodes (10 to 18 vs. more than 18), tumor size (≤ 5 cm vs. > 5 cm), histological grade, and hormone-receptor status. Only the number of involved lymph nodes was predictive of DFS (p=0.036) (Fig. 2). In the 12 patients with 10 to 18 involved nodes, the 5-year DFS and OS rates were 67±14% and 75±15%, respectively, while in the 10 patients with more than 18 positive nodes there were 30±15% and 38±16%, respectively. The hazard ratio for recurrence was significantly higher in patients with more than 18 positive nodes (3.51; 95% CI, 1.01 to 12.24; p=0.048), and that for death (2.90; 95% CI, 0.84 to 9.95) only showed a trend in favor of patients with 10 to 18 positive nodes.
Consolidation HDC with cyclophosphamide, thiotepa and carboplatin (CTCb) followed by ASCT for primary breast cancer with 10 or more positive lymph nodes had an acceptable toxicity; however, it does not show promising survival. After a median follow-up of 58 months from the date of stem-cell infusion, the 5-year DFS and OS rates were 50% and 58%, respectively.
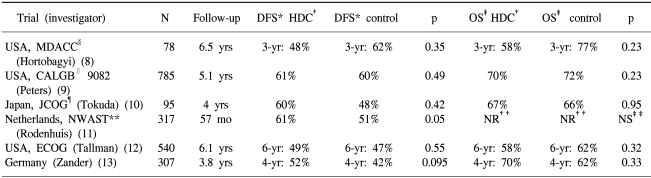
The outcomes of patients with more than 10 involved nodes receiving conventional-dose adjuvant chemotherapy have been poor. The 5-year DFS rate was average 38% (range, 17% to 56%) (18). Compared with conventional-dose chemotherapy, HDC may improve the chance of eradicating micrometastases in patients at high risk of recurrence, thus increasing the impact of adjuvant therapy on survival. Several phase II trials (5,6) have reported encouraging results after HDC, showing an average 5-year DFS rate of 58% (range, 50% to 64%) (18). However, of the 6 HDC randomized trials on primary breast cancer with more than 10 involved nodes reported to date, none have shown a significant survival benefit (Table 2).
In the Intergroup Cancer and Leukemia Group B 9082 trial, comprised of 785 enrolled patients (9), there were fewer relapses in the HDC arm compared with non-HDC arm (28.9% vs. 39.1%). However, there were 32 treatment-related deaths (8.1%) in the HDC arm, offsetting, in part, the benefit from HDC. Similarly, in the Eastern Cooperative Oncology Group trial (12), 9 patients died of transplantation-related complications, and myelodysplastic syndrome or acute myeloid leukemia developed in a further 9. No significant difference in survival was observed, although the time to recurrence was longer among patients in the group assigned to HDC with stem-cell transplantation. Transplantation-related mortality could be eliminated through the use of peripheral-blood stem cells, noncarmustine-based regimens to reduce the pulmonary toxicity, and less potentially leukemogenic regimens. A longer follow-up period may also be required before a definitive conclusion relating to survival can be made, as breast cancer can potentially recur up to 15 to 20 years after treatment. In the first analysis of the German trial (19), Zander et al. reported a substantial DFS advantage in the transplant arm compared with the standard-dose chemotherapy arm for patients with the longest follow-up, with 6-year actuarial DFS rates of 50% and 25%, respectively. However, no DFS or OS differences were noted in the whole study of 302 patients, with a median follow- up of 3.7 years (20). Moreover, it is possible that the benefit from HDC was restricted to only certain subgroups of patients, resulting in a small observed difference for the whole study group. In the German trial (13), there was a trend favoring HDC with respect to event-free survival in premenopausal patients and those with a grade 3 tumor, but this effect was not significant. The same trend was also observed in younger patients and in those with a HER2/neu negative tumor (11), and another group of patients, which might benefit more from HDC, could be. Therefore, a meta-analysis of updated data from these 6 randomized trials could be warranted.
Our 5-year DFS rate of 50% was superior to the results with conventional-dose adjuvant chemotherapy (18), although such comparisons need to be conducted carefully due to the differences in patient selection and the rigorous staging of patients, etc. Although the regimens were heterogeneous, and the trial designs variable, our 5-year DFS and OS of 50% and 58% were comparable to the results of other phase II trials (5, 6) and the outcomes of the HDC arm in randomized trials (8~13). However, our DFS and OS were not superior to those of the conventional-dose chemotherapy arm in the randomized trials (8~13) or the survival achieved with the same CTCb regimen used in our trial. After a median follow-up of 51 months (range, 27 to 87), Stemmer et al. (21) reported DFS and OS rates of 72% and 81%, respectively. The reasons for this are unclear, but then it is possible that our consolidation regimen was inadequate. While they used single-agent doxorubicin as the adjuvant chemotherapy, followed by a HDC as a consolidation, most patients in this series was administered a cyclophosphamide-based regimen as an adjuvant chemotherapy, which could theoretically have induced resistance to a high-dose cyclophosphamide-containing transplant protocol. Furthermore, the CTCb regimen may not be a true high-dose regimen. While the thiotepa dose was significantly elevated above its standard dose (approximately eight times), the carboplatin dose was less than twice the single-agent dose. In addition, cyclophosphamide and thiotepa were given as continuous infusions, which may have significantly decreased the peak plasma level and/or area under the curve of the active metabolites of cyclophosphamide (22,23).
The toxicities were reversible and no treatment-related mortality was observed. This was attributed to the selection of a noncarmustine-based transplant protocol, the use of peripheral blood stem-cell support, and better supportive care. This low treatment-related mortality was in accordance with other studies (8,10,11,13,18,21,24).
In this series, the Cox multivariate analysis demonstrates that only the number of involved lymph nodes was an independent predictor of the DFS, but not of the OS. Although this was inconsistent with another report (12), it was not surprising that patients with 10 to 18 positive nodes showed a better 5-year DFS rate than those with more than 18 positive nodes (75% vs. 30%, p=0.036). The number of involved lymph nodes (> 9 vs. 4 to 9 vs. < 4) is a strong predictive factor for recurrence or survival. Similarly, the outcome of patients with more than 18 positive nodes might be inferior to that of patients with 10 to 18 positive nodes, and HDC might be inappropriate for very high-risk populations (more than 18 nodes). Because this study was small, and not a true population-based study, firm statements regarding survival statistics are impossible. However, our results are worth pursuing in a trial of adequate size. In contrast to other reports (11,13), age, menopausal status, and histological grade were not an important prognostic factors for survival.
This was a study with a prolonged follow-up period (median 58 months and maximum 9 years). Adjuvant HDC for resected breast cancer is not currently accepted as a standard therapy. Nevertheless, the breast cancer patients in this study with more than 10 nodes had 5-year DFS and OS rates of 50% and 58%, respectively, after consolidation HDC with ASCT.
References
1. Jones SE, Moon TE, Bonadonna G, Valagussa P, Rivkin S, Buzdar A, et al. Comparison of different trials of adjuvant chemotherapy in stage II breast cancer using a natural history data base. Am J Clin Oncol. 1987; 10:387–395. PMID: 3310603.

2. Buzdar AU, Kau SW, Hortobagyi GN, Ames FC, Holmes FA, Fraschini G, et al. Clinical course of patients with breast cancer with ten or more positive nodes who were treated with doxorubicin-containing adjuvant therapy. Cancer. 1992; 69:448–452. PMID: 1728373.

3. Davidson NE, Abeloff MD. Adjuvant systemic therapy in women with early-stage breast cancer at high risk for relapse. J Natl Cancer Inst. 1992; 84:301–305. PMID: 1531367.

4. Hryniuk W, Levine MN. Analysis of dose intensity for adjuvant chemotherapy trials in stage II breast cancer. J Clin Oncol. 1986; 4:1162–1170. PMID: 3525765.

5. Peters WP, Ross M, Vredenburgh JJ, Meisenberg B, Marks LB, Winer E, et al. High-dose chemotherapy and autologous bone marrow support as consolidation after standard-dose adjuvant therapy for high-risk primary breast cancer. J Clin Oncol. 1993; 11:1132–1143. PMID: 8501500.

6. Gianni AM, Siena S, Bregni M, Di Nicola M, Orefice S, Cusumano F, et al. Efficacy, toxicity, and applicability of high-dose sequential chemotherapy as adjuvant treatment in operable breast cancer with 10 or more involved axillary nodes: five-year results. J Clin Oncol. 1997; 15:2312–2321. PMID: 9196145.

7. Gradishar WJ, Tallman MS, Abrams JS. High-dose chemotherapy for breast cancer. Ann Intern Med. 1996; 125:599–604. PMID: 8815759.

8. Hortobagyi GN, Buzdar AU, Theriault RL, Valero V, Frye D, Booser DJ, et al. Randomized trial of high-dose chemotherapy and blood cell autografts for high-risk primary breast carcinoma. J Natl Cancer Inst. 2000; 92:225–233. PMID: 10655439.

9. Peters W, Rosner G, Vredenburgh J, Shpall EJ, Crump M, Marks L, et al. Updated results of a prospective, randomized comparison of two doses of combination alkylating agent (AA) as consolidation after CAF in high-risk primary breast cancer involving ten or more axillary lymph nodes (LN): CALGB 9082/SWOG 9114/NCIC MA-13. Proc Am Soc Clin Oncol. 2001; 20:67b. (abstr 2016).
10. Tokuda Y, Tajima T, Narabayashi M, Takeyama K, Watanabe T, Fukutomi T, et al. Randomized phase III study of high- dose chemotherapy (HDC) with autologous stem cell support as consolidation in high-risk postoperative breast cancer: Japan Clinical Oncology Group (JCOG9208). Proc Am Soc Clin Oncol. 2001; 20:38a. (abstr 148).
11. Rodenhuis S, Bontenbal M, Beex LV, Wagstaff J, Richel DJ, Nooij MA, et al. High-dose chemotherapy with hematopoietic stem-cell rescue for high-risk breast cancer. N Engl J Med. 2003; 349:7–16. PMID: 12840087.

12. Tallman MS, Gray R, Robert NJ, LeMaistre CF, Osborne CK, Vaughan WP, et al. Conventional adjuvant chemotherapy with or without high-dose chemotherapy and autologous stem-cell transplantation in high-risk breast cancer. N Engl J Med. 2003; 349:17–26. PMID: 12840088.

13. Zander AR, Kroger N, Schmoor C, Kruger W, Mobus V, Frickhofen N, et al. High-dose chemotherapy with autologous hematopoietic stem-cell support compared with standard-dose chemotherapy in breast cancer patients with 10 or more positive lymph nodes: first results of a randomized trial. J Clin Oncol. 2004; 22:2273–2283. PMID: 15111618.

14. Cho SH, Kim SW, Min YJ, Choi SJ, Kim JK, Kim TW, et al. High dose cyclophosphamide, thiotepa, and carboplatin followed by autologous peripheral stem cell rescue in patients with responsive metastatic or high-risk primary breast cancer. J Korean Cancer Assoc. 1998; 30:100–105.
15. Bezwoda WR, Seymour L, Dansey RD. High-dose chemotherapy with hematopoietic rescue as primary treatment for metastatic breast cancer: a randomized trial. J Clin Oncol. 1995; 13:2483–2489. PMID: 7595697.

16. Bezwoda W. Randomised, controlled trial of high dose chemotherapy (HD-CNVP) versus standard dose (CAF) chemotherapy for high risk, surgically treated, primary breast cancer. Proc Am Soc Clin Oncol. 1999; 18:2a. (abstr 4).

17. Suh C, Kim HJ, Kim SH, Kim S, Lee SJ, Lee YS, et al. Low-dose lenograstim to enhance engraftment after autologous stem cell transplantation: a prospective randomized evaluation of two different fixed doses. Transfusion. 2004; 44:533–538. PMID: 15043569.

18. Damon LE, Wolf JL, Rugo HS, Gold E, Zander AR, Cassidy M, et al. High-dose chemotherapy (CTM) for breast cancer. Bone Marrow Transplant. 2000; 26:257–268. PMID: 10967563.

19. Zander A, Kruger W, Kroger N, Schmoor C, Mobus V, Frickhofen N, et al. High-dose chemotherapy with autologous hematopoietic stem-cell support (HSCS) vs. standard-dose chemotherapy in breast cancer patients with 10 or more positive lymph nodes: first results of a randomized trial. Proc Am Soc Clin Oncol. 2002; 21:415a. (abstr 1658).
20. Nieto Y. The verdict is not in yet. Analysis of the randomized trials of high-dose chemotherapy for breast cancer. Haematologica. 2003; 88:201–211. PMID: 12604410.
21. Stemmer SM, Hardan I, Raz H, Adamou AK, Inbar M, Gottfried M, et al. Adjuvant treatment of high-risk stage II breast cancer with doxorubicin followed by high-dose chemotherapy and autologous stem-cell transplantation: a single-institution experience with 132 consecutive patients. Bone Marrow Transplant. 2003; 31:655–661. PMID: 12692605.

22. Anderson LW, Chen TL, Colvin OM, Grochow LB, Collins JM, Kennedy MJ, et al. Cyclophosphamide and 4-Hydroxycyclophosphamide/ aldophosphamide kinetics in patients receiving high-dose cyclophosphamide chemotherapy. Clin Cancer Res. 1996; 2:1481–1487. PMID: 9816324.
23. Huitema AD, Kerbusch T, Tibben MM, Rodenhuis S, Beijnen JH. Reduction of cyclophosphamide bioactivation by thiotepa: critical sequence-dependency in high-dose chemotherapy regimens. Cancer Chemother Pharmacol. 2000; 46:119–127. PMID: 10972481.

24. Fisher DC, Vredenburgh JJ, Petros WP, Hussein A, Berry DA, Elkordy M, et al. Reduced mortality following bone marrow transplantation for breast cancer with the addition of peripheral blood progenitor cells is due to a marked reduction in veno-occlusive disease of the liver. Bone Marrow Transplant. 1998; 21:117–122. PMID: 9489626.





 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download