MATERIALS AND METHODS
Case selection
Immunohistochemical study
Nested real time polymerase chain reaction (nested reverse transcription polymerase chain reaction)
Ethics statement
RESULTS
Case descriptions
Patient A
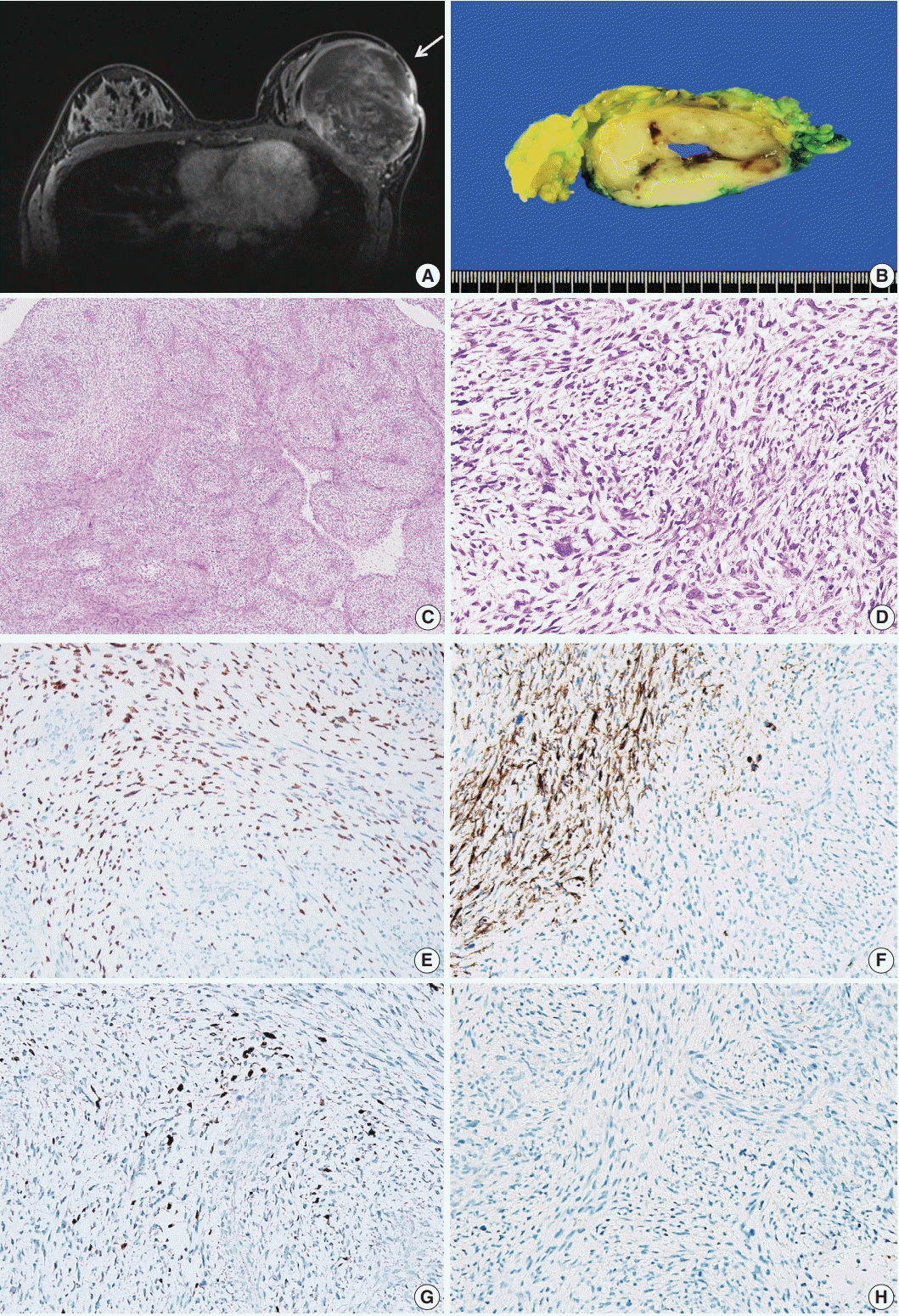 | Fig. 1.Primary breast sarcoma of patient A. (A) A 12×9.5×8.3-cm-sized heterogeneously enhancing mass in the left breast (yellow arrow). (B) Grayish-yellow cut surface of the mass with multifocal hemorrhagic spots and central cavity formation. (C, D) Spindle cells exhibiting a fascicular or storiform growth pattern with a small amount of myxoid matrix. The image showing highly pleomorphic elongated nuclei and pale to eosinophilic cytoplasm, with high mitotic rates (up to 94/10 high power fields). (E–H) Immunohistochemical staining showing positivity of tumor cells for MyoD1 (E), desmin (F), and myogenin (G) and negativity for MNF-116 (H). |
Patient B
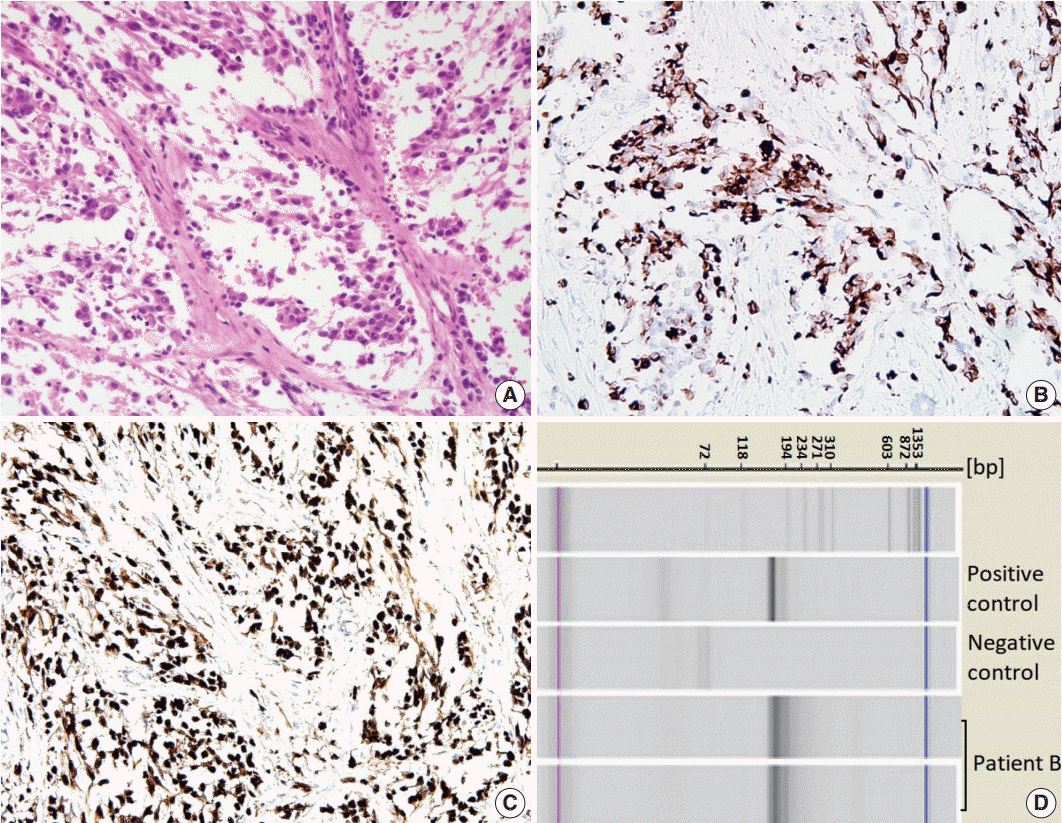 | Fig. 2.Primary breast sarcoma of patient B. (A) Primitive round-to-polygonal cells comprising the nests separated by fibrous septa and showing a loss of cellular cohesion. (B, C) Immunohistochemical staining for desmin (B) and myogenin (C). (D) PAX3-FKHR (FOXO1) fusion identified by PAX-3/PAX-7-FKHR (FOXO1) nested reverse transcription polymerase chain reaction analysis. |
Patient C
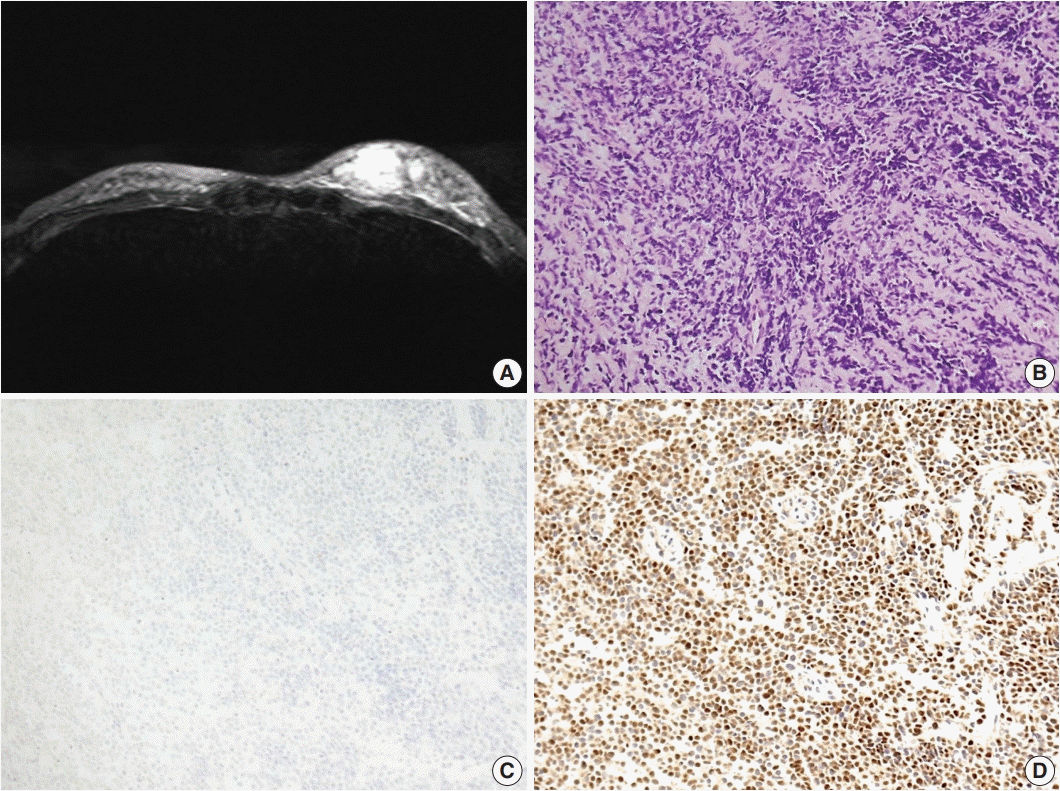 | Fig. 3.Primary breast sarcoma of patient C. (A) Magnetic resonance imaging of the breast showing multifocal, infiltrating, ill-defined, enhancing masses in the left lower inner, upper inner, and lower outer quadrants. (B) Hyperchromatic small round cells with scanty cytoplasm exhibiting solid-sheet and cord-like growth patterns. (C, D) Immunohistochemical staining showing negativity for desmin (C) and positivity for myogenin (D). |
Other PBSs
Table 1.
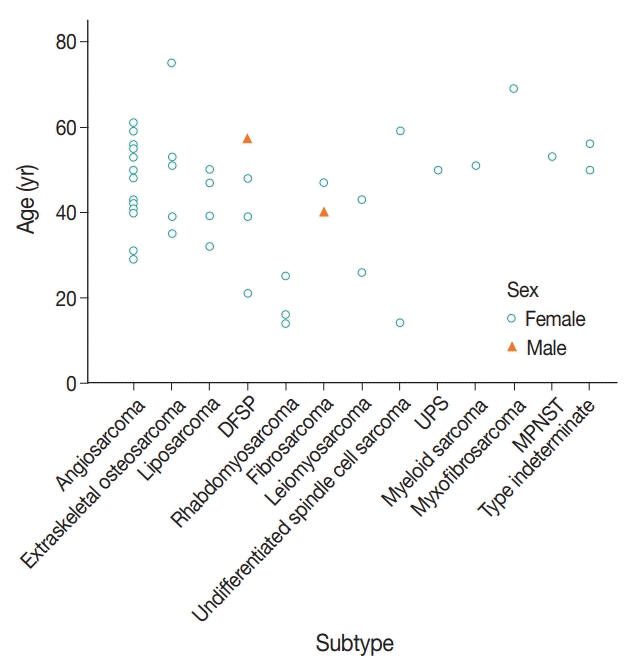 | Fig. 4.Age distribution of primary breast sarcoma (PBS) according to subtype. Angiosarcoma was the most common sarcoma, followed by extra-skeletal osteosarcoma, liposarcoma, and dermatofibrosarcoma protuberans (DFSP). The most prevalent age-group was 50–60 years. PBS was rare in people aged less than 30, and rhabdomyosarcoma was the most common subtype in this age-group. UPS, undifferentiated pleomorphic sarcoma; MPNST, malignant peripheral nerve sheath tumor. |
DISCUSSION
Table 2.
| Study | Age (yr) | Subtype | Size (cm) | LN | Meta | Surgical procedure | Other treatment | Prognosis |
|---|---|---|---|---|---|---|---|---|
| Hays et al. (1997) [18] | 13.6 | Alveolar | 3 | + | + | NI | RTx | DOD 79 wk |
| 16.9 | Alveolar | 15 | + | + | Excision | CTx, RTx | DOD 42 wk | |
| 15.5 | Alveolar | 21 | + | – | Radical mastectomy | CTx | NED 252 wk | |
| 16.9 | Alveolar | 8 | – | – | Radical mastectomy | RTx | NED 365 wk | |
| 15.2 | Alveolar | 6.5 | – | – | Mastectomy | RTx | NED 150 wk | |
| 15 | Alveolar | NI | – | + | NI | NI | DOD 124 wk | |
| 14.9 | Embryonal | 10 | – | – | Excision | RTx | NED 362 wk | |
| Hererra and Lugo-Vicente (1998) [19] | 13 | Embryonal | 6 | – | – | NI | CTx | NED 1 yr |
| Binokay et al. (2003) [20] | 16 | Alveolar | 10 | + | – | MRM | NI | NI |
| Vishnevskaia et al. (2004) [21] | 14 | Alveolar | NI | + | + | NI | NI | DOD 3 yr 1 mo |
| Italiano et al. (2005) [14],a | 46 | Embryonal | 3.5 | NI | NI | Quadrantectomy | CTx, RTx | NED 18 mo |
| Nogi et al. (2007) [23] | 13 | Alveolar | 13 | + | – | Total mastectomy | CTx | DOD 8 mo |
| Attili et al. (2007) [5] | 40 | Embryonal | 4 | + | – | MRM | CTx | NED 1 yr |
| Rasinariu et al. (2011) [9] | 58 | Spindle cell/sclerosing | 11 | NI | NI | Mastectomy | NI | NI |
| Li et al. (2012) [12] | 30 | Alveolar | 2.5 | + | – | MRM | neoCTx | DOD 29 mo |
| 17 | Embryonal | 10 | – | – | Simple mastectomy | NI | NED 34 mo | |
| Valera et al. (2013) [24] | 17 | Alveolar | 3.1 | NI | + | None | CCRT | NI |
| Bhosale et al. (2013) [25] | 60 | NI | 8 | + | – | MRM | CTx | NED 6 mo |
| Mondal et al. (2014) [26] | 49 | Pleomorphic | 7 | – | – | MRM | None | NED 12 mo |
| Kallianpur et al. (2015) [27] | 19 | NI | 30 | – | – | Mastectomy | CTx, RTx | NED 2 mo |
| Pareekutty et al. (2016) [28] | 12 | Alveolar | 9 | + | – | MRM | CTx, RTx | NED 35 mo |
| Audino et al. (2017) [7] | 16 | Alveolar | 12.5 | + | + | MRM | CTx, RTx | Recur 19 mo |
| Kim et al. (2017) [10] | 17 | Alveolar | 3.1 | – | + | Mastectomy | CTx | Recur 5 mo |
| Yuan et al. (2017) [29] | 34 | NI | 3.5 | NI | NI | Mastectomy | CTx | NED 23 mo |
| Bayramoglu et al. (2018) [30] | 12 | Alveolar | NI | + | – | NI | NI | NI |
| Jean-Louis (2018) [31] | 16 | Alveolar | 8.1 | + | – | Excision | NI | NI |
| Current study | 14 | Spindle cell/sclerosing | 16.7 | – | + | Mastectomy | CTx, RTx | Recur 4 mo |
| 16 | Alveolar | 9.5 | + | + | Mastectomy | CTx | DOD 22 mo | |
| 25 | Embryonal | 3 | + | – | - | CTx, RTx | DOD 18 mo |
LN, lymph node status; Meta, metastasis; NI, not identified; RTx, radiotherapy; DOD, dead of disease; CTx, chemotherapy; NED, no evidence of disease; MRM, modified radical mastectomy; neoCTx, neoadjuvant chemotherapy; CCRT, concurrent chemoradiation therapy.
a This was an identical case of Dausse et al. (2005) [22].




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download