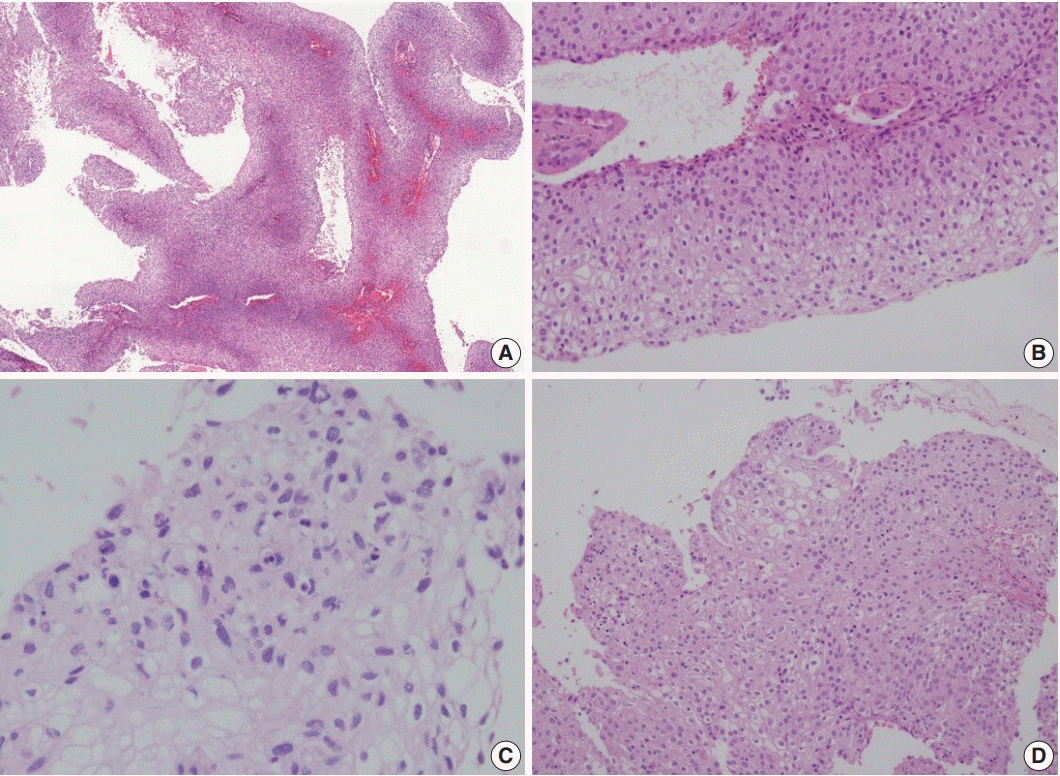
1. Randall ME, Andersen WA, Mills SE, Kim JA. Papillary squamous cell carcinoma of the uterine cervix: a clinicopathologic study of nine cases. Int J Gynecol Pathol. 1986; 5:1–10.
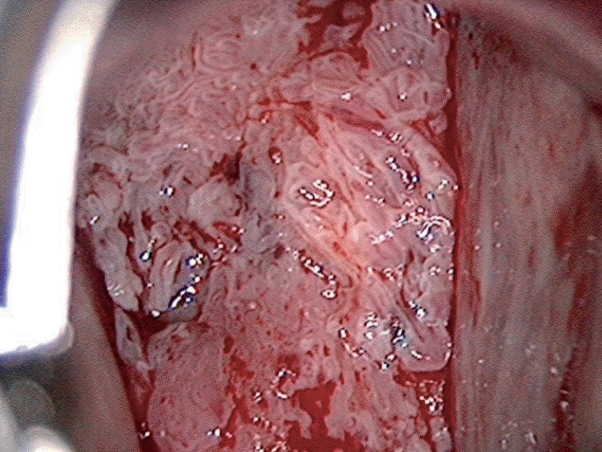
2. Al-Nafussi AI, Al-Yusif R. Papillary squamotransitional cell carcinoma of the uterine cervix: an advanced stage disease despite superficial location: report of two cases and review of the literature. Eur J Gynaecol Oncol. 1998; 19:455–7.
3. Koenig C, Turnicky RP, Kankam CF, Tavassoli FA. Papillary squamotransitional cell carcinoma of the cervix: a report of 32 cases. Am J Surg Pathol. 1997; 21:915–21.
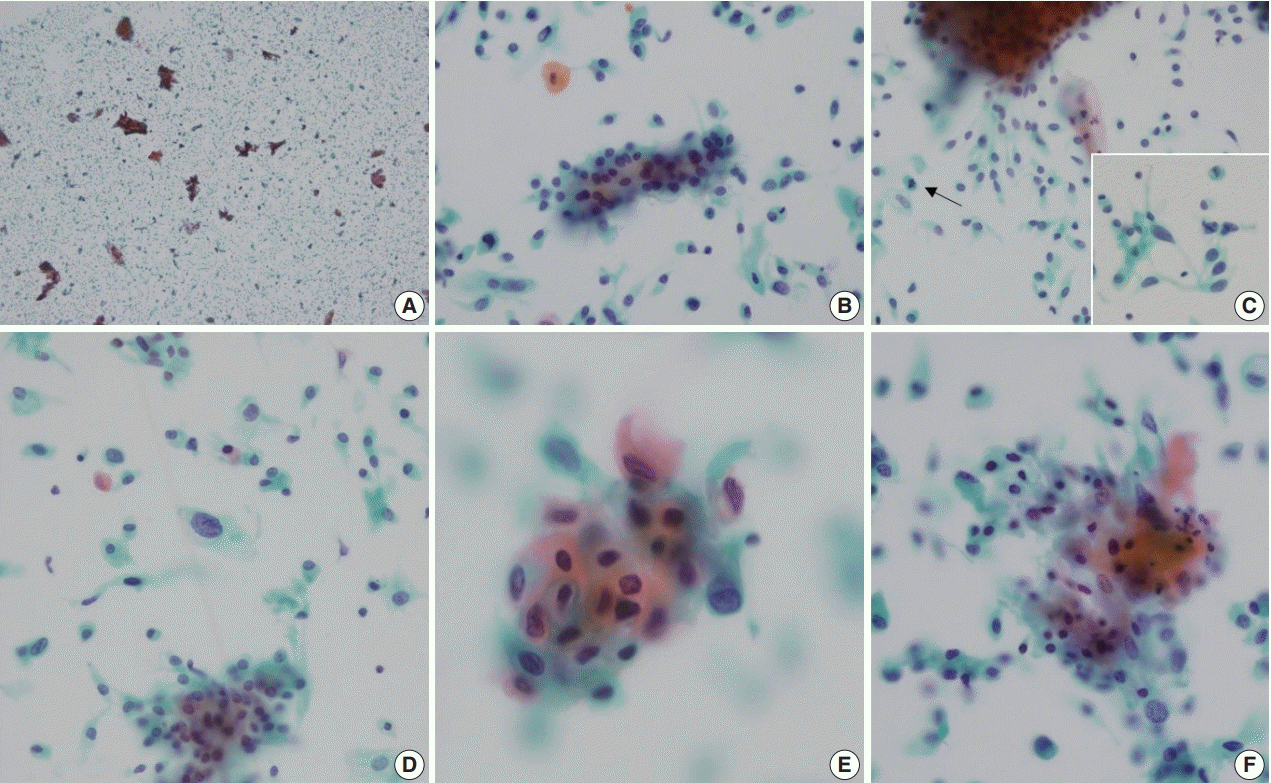
4. Ng WK. Thin-layer (liquid-based) cytologic findings of papillary squamotransitional cell carcinoma of the cervix. Review of cases over a 4-year period with emphasis on potential diagnostic pitfalls. Acta Cytol. 2003; 47:141–8.
5. Vesoulis Z, Erhardt CA. Cytologic diagnosis of vaginal papillary squamotransitional cell carcinoma. A case report. Acta Cytol. 2001; 45:465–9.
6. Kokka F, Verma M, Singh N, Faruqi A, Yoon J, Reynolds K. Papillary squamotransitional cell carcinoma of the uterine cervix: report of three cases and review of the literature. Pathology. 2006; 38:584–6.

7. Kim HS, Seon MI, Kim YJ, Kim HS. Cytologic features of papillary immature metplasia of uterine cervix. Korean J Cytopathol. 2002; 13:21–7.
8. Chung YR, Won JK, Park IA, et al. Cytomorphological characteristics of low-grade papillary urothelial carcinoma for differential diagnosis from benign papillary urothelial lesions: logistic regression analysis in SurePath(TM) liquid-based voided urine cytology. Cytopathology. 2016; 27:83–90.
9. Fujimoto T, Sakuragi N, Shimizu M, et al. Papillary squamous cell carcinoma of the uterine cervix: a report of two cases with human papillomavirus 16 DNA. Acta Obstet Gynecol Scand. 2002; 81:176–8.

10. Maitra A, Gibbons D, Gazdar AF, Albores-Saavedra J. Allelic losses at chromosome 3p are seen in human papilloma virus 16 associated transitional cell carcinoma of the cervix. Gynecol Oncol. 1999; 74:361–8.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download