Abstract
Probe-based confocal microscopy (pCLE) is actively being investigated for applications in the esophagus and stomach. The use of pCLE allows real-time in vivo microscopy to evaluate the microarchitecture of the mucosal epithelium. pCLE appears to be particularly useful in identifying mucosal dysplasia and early malignancies that cannot be clearly distinguished using high-definition white light endoscopy, chromoendoscopy, or magnification endoscopy. In addition, the ability to detect dysplastic tissue in real-time may shift the current screening practice from random biopsy to targeted biopsy of esophageal and gastric cancers and their precursor lesions. We will review the use of pCLE for detection and surveillance of upper gastrointestinal early luminal malignancy.
Go to : 
The endoscopic use of confocal microscopy for in vivo diagnosis is a relatively new application of a previously ex vivo technology.1 Probe catheters range in size from 0.9- to 2.5-mm allowing their use through any endoscope with a 10 Fr working channel. With a video frame rate of 12 frames per second, probe-based confocal microscopy (pCLE) provides in vivo imaging of capillary flow and real-time microscopic scanning ability. The resolution is approximately 1 µm and 240 to 600 µm field of view.2,3 A contrast agent, typically intravenous fluorescein, is administered to highlight the connective tissue of the lamina propria and the vascular rich epithelium. Mucin containing goblet cells remain dark, as does neoplastic tissue.3,4 This contrast effect is present 10 minutes after administration and lasts up to 45 minutes.5 Fluorescein has long been used for ophthalmologic indications and its safety has recently been confirmed for use as a contrast agent in the gastrointestinal tract.6
Due to the small field of view and high magnification, optimal image acquisition is highly dependent on proper positioning. The best images are obtained with the probe perpendicular to the mucosa. Some operators have found benefit from the use of a clear 4-mm cap placed on the tip of the endoscope, as this allows gentle suctioning to keep the probe in place.3 Imaging is best done before biopsy or resection, as bleeding can obscure imaging due to fluorescein leakage.
Go to : 
The current method for surveillance of Barrett esophagus (BE) is both time consuming and expensive. Targeted biopsies of endoscopically visible lesions are then followed by four-quadrant biopsies-every 1 cm for dysplastic BE and every 2 cm for nondysplastic BE. Society guidelines recommend endoscopic surveillance of all patients with established BE.7,8 However, adherence rates to surveillance biopsy guidelines have been reported to be as low as 51% in the community setting.9 Therefore, a method that improves the efficiency (cost and time) of performing endoscopic surveillance in patients with BE would have a positive impact on the management of patients with BE from both a clinical and economic standpoints.
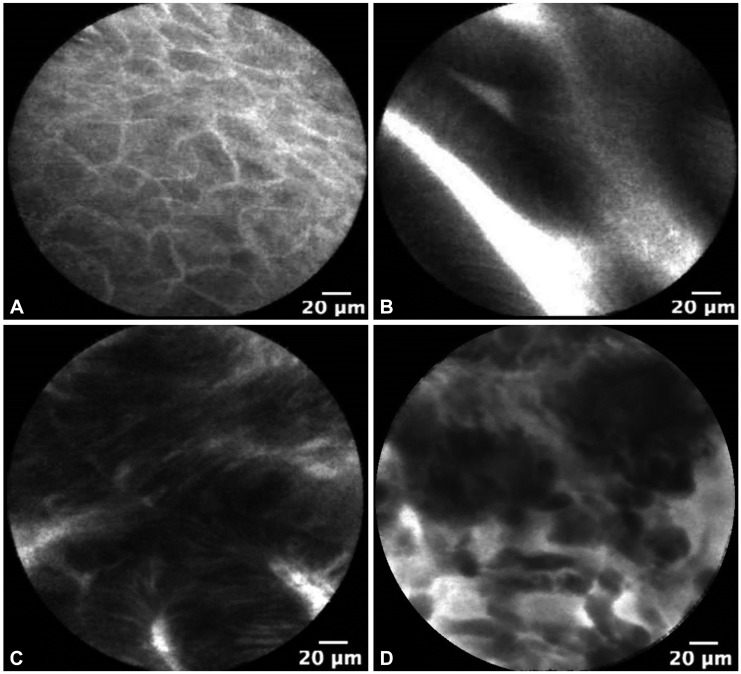
Probe-based confocal laser endomicroscopy is currently used in clinical trials as a tool for BE imaging. The Miami criteria for differentiating nondysplastic BE from dysplastic BE and adenocarcinoma was proposed in 2011.3 Normal squamous epithelium is defined as flat cells without crypts or villi, with bright vessels found within papillae (Fig. 1A). Nondysplastic BE has a uniform villiform architecture and columnar cells interspersed with dark goblet cells (Fig. 1B). High-grade dysplasia (HGD) retains a villiform structure but has dark, irregularly thickened epithelial borders, and dilated irregular vessels (Fig. 1C). In contrast, adenocarcinoma has disorganized or absent villiform structure, multiple dark columnar cells, and dilated irregular vessels (Fig. 1D). This classification was recently validated and notably, after a short formal training session, there was high interobserver agreement and accuracy between experts and recent trainees.10
Current screening and surveillance protocols rely on biopsy to differentiate BE, dysplastic BE, and adenocarcinoma. This requires endoscopists to carefully document biopsy location and, once the pathology returns dysplastic, the patient must return for endoscopic ablation. The use of pCLE theoretically will allow surveillance of a greater number of locations, reduce the total number of biopsies taken, and increase the yield of biopsy for dysplasia or cancer. Two studies have recently demonstrated the potential feasibility of this approach (Table 1). Bajbouj et al.11 reported their experience with 68 patients undergoing BE surveillance. Six hundred and seventy biopsies were performed and 11 patients (16%) had HGD or esophageal cancer (EC) found in 58 biopsies. In this study, onsite analysis and blinded review of pCLE images were performed. The specificity and negative predictive value of pCLE in excluding neoplasia was 0.97 (90% confidence interval [CI], 0.95 to 0.98) and 0.93 (90% CI, 0.91 to 0.95), which had little change in blinded evaluation with specificity of 0.95 (90% CI, 0.90 to 0.98) and a negative predictive value of 0.97 (90% CI, 0.95 to 0.98). A randomized multicenter trial using pCLE in BE demonstrated pCLE to be superior to high-definition white light (HDWL) endoscopy for detection of BE HGD. The investigators looked at the use of pCLE, HDWL, and narrow band imaging (NBI) in 101 patients, 31 of whom had EC (31%), who had a total of 874 biopsies performed. If biopsies were only taken in patients with abnormalities detected on any one of the three imaging modalities, 39% of patients could forgo biopsy without missing HGD or EC.12 These studies indicate that pCLE could decrease biopsy use, and given the high negative predictive value, provide increased reassurance for patients with negative tests.
Another area that pCLE has potential to impact management of patients with BE is in patients undergoing ablative therapy. Ablative therapy is now recommended for patients with BE with flat HGD.1 Patients often require between 2 and 10 ablation sessions to obtain remission.13,14 Probe-based CLE may play a role guiding therapy before ablation, during ablation sessions, and in the lifelong screening following these sessions. A multicenter randomized clinical trial comparing HDWL endoscopy to HDWL+pCLE in the detection of residual BE in patients who have had ablative therapy was recently performed, however was halted early due to a higher rate of residual Barrett's in both treatment arms.15 Further study is needed to determine the utility of pCLE in differentiating mucosa after ablative therapy. If feasible, given the rise in ablative therapy for BE, pCLE may develop a greater role in guiding and assessing therapy.
Go to : 
The potential applications for using pCLE in the stomach are similar to that of the esophagus, especially in populations with a high incidence of gastric cancer. The current method for endoscopic gastric cancer screening typically involves a careful visual examination with white light endoscopy, targeted biopsies of endoscopically visible lesions, and then random biopsies in various regions of the stomach (antrum, body, and incisura). As with Barrett's screening and surveillence, this approach is both time consuming and expensive. Furthermore, the large surface area of the stomach means this approach is even more prone to undersampling of abnormal tissue and oversampling of normal tissue. The use of chromoendoscopy, NBI, or other digital enhancement techniques has increased the accuracy of detecting gastric intestinal metaplasia (GIM) and early gastric cancer (EGC); however, these techniques are limited by significant interobserver variability.12-19 Moreover, while these modalities have increased the sensitivity and specificity of biopsy screening, no study has demonstrated the ability to accurately diagnose dysplasia or GIM-the known precursor lesions for gastric adenocarcinoma. As such, suspicious areas still require biopsy to confirm the presence of GIM or EGC and no studies have demonstrated the ability to accurately diagnose dysplasia.
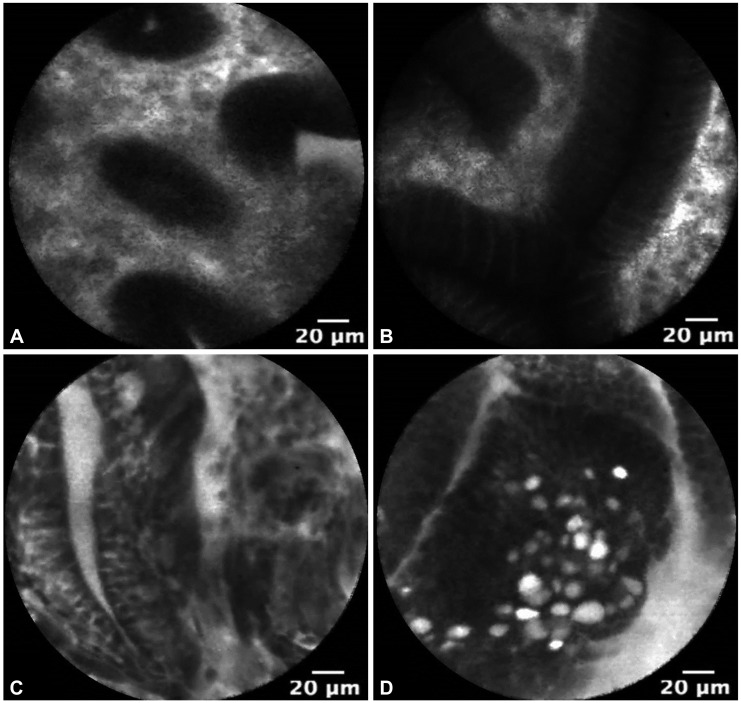
The use of pCLE is currently under investigation for the in vivo diagnosis of GIM, dysplasia, and EGC. Fig. 2 demonstrates pCLE images of normal gastric mucosa, GIM, gastric HGD, and EGC. In a recent study, pCLE performed before endoscopic resection of superficial gastric neoplasia was compared to conventional endoscopic biopsy. pCLE had significantly improved agreement over conventional biopsy for the diagnosis of adenocarcinoma (κ=0.824 vs. κ=0.617; p<0.001) and an accuracy of 91.7% compared to 85.2% for conventional biopsy (p=0.065).20 Guo et al.19 additionally demonstrated 98% sensitivity and 95% specificity for GIM using confocal microscopy. Notably, the learning curve for pCLE detection of GIM is short, typically obtaining acceptable intraobserver agreement with 1 to 2 hours of training.21 Yet to be explored is whether pCLE can improve the diagnosis of dysplasia following gastric endoscopic mucosal resection or endoscopic submucosal resection.
Similar to the applications in BE, pCLE has the potential to identify dysplasia and EGC in vivo to increase the yield of biopsies and to identify lesions that may warrant endoscopic resection. Further studies are needed to fully evaluate the potentially valuable application of pCLE for gastric malignancy.
Go to : 
Probe-based CLE is a promising technology with several potential applications in the management of patients with BE and GIM. One of the limitations to pCLE guided biopsy has been ensuring the visualized mucosa is sampled. Currently in development are biopsy forceps that open around the tip of the probe and extend out to directly capture the visualized mucosa. Also in development are novel contrast agents and other molecular probes for improving diagnostic accuracy.22
Studies have demonstrated the ability of pCLE to diagnose dysplasia while reducing the total number of biopsies.6 However, in order to achieve wider adoption of this technology, further studies will need to be performed to demonstrate that the strategy of targeted biopsies based on pCLE findings is as accurate as conventional biopsy and cost-effective in community practice. In addition, the role of pCLE in guiding therapy warrants further investigation.
Go to : 
References
1. Kiesslich R, Burg J, Vieth M, et al. Confocal laser endoscopy for diagnosing intraepithelial neoplasias and colorectal cancer in vivo. Gastroenterology. 2004; 127:706–713. PMID: 15362025.

2. Wallace MB, Fockens P. Probe-based confocal laser endomicroscopy. Gastroenterology. 2009; 136:1509–1513. PMID: 19328799.

3. Wallace M, Lauwers GY, Chen Y, et al. Miami classification for probe-based confocal laser endomicroscopy. Endoscopy. 2011; 43:882–891. PMID: 21818734.

4. Wang TD, Friedland S, Sahbaie P, et al. Functional imaging of colonic mucosa with a fibered confocal microscope for real-time in vivo pathology. Clin Gastroenterol Hepatol. 2007; 5:1300–1305. PMID: 17936692.

5. Polglase AL, McLaren WJ, Skinner SA, Kiesslich R, Neurath MF, Delaney PM. A fluorescence confocal endomicroscope for in vivo microscopy of the upper- and the lower-GI tract. Gastrointest Endosc. 2005; 62:686–695. PMID: 16246680.

6. Wallace MB, Meining A, Canto MI, et al. The safety of intravenous fluorescein for confocal laser endomicroscopy in the gastrointestinal tract. Aliment Pharmacol Ther. 2010; 31:548–552. PMID: 20002025.

7. American Gastroenterological Association. Spechler SJ, Sharma P, Souza RF, Inadomi JM, Shaheen NJ. American Gastroenterological Association medical position statement on the management of Barrett's esophagus. Gastroenterology. 2011; 140:1084–1091. PMID: 21376940.

8. ASGE Standards of Practice Committee. Evans JA, Early DS, et al. The role of endoscopy in Barrett's esophagus and other premalignant conditions of the esophagus. Gastrointest Endosc. 2012; 76:1087–1094. PMID: 23164510.

9. Abrams JA, Kapel RC, Lindberg GM, et al. Adherence to biopsy guidelines for Barrett's esophagus surveillance in the community setting in the United States. Clin Gastroenterol Hepatol. 2009; 7:736–742. PMID: 19268726.

10. Gaddam S, Mathur SC, Singh M, et al. Novel probe-based confocal laser endomicroscopy criteria and interobserver agreement for the detection of dysplasia in Barrett's esophagus. Am J Gastroenterol. 2011; 106:1961–1969. PMID: 21946283.

11. Bajbouj M, Vieth M, Rösch T, et al. Probe-based confocal laser endomicroscopy compared with standard four-quadrant biopsy for evaluation of neoplasia in Barrett's esophagus. Endoscopy. 2010; 42:435–440. PMID: 20506064.

12. Sharma P, Meining AR, Coron E, et al. Real-time increased detection of neoplastic tissue in Barrett's esophagus with probe-based confocal laser endomicroscopy: final results of an international multicenter, prospective, randomized, controlled trial. Gastrointest Endosc. 2011; 74:465–472. PMID: 21741642.

13. Shaheen NJ, Overholt BF, Sampliner RE, et al. Durability of radiofrequency ablation in Barrett's esophagus with dysplasia. Gastroenterology. 2011; 141:460–468. PMID: 21679712.
14. Gupta M, Iyer PG, Lutzke L, et al. Recurrence of esophageal intestinal metaplasia after endoscopic mucosal resection and radiofrequency ablation of Barrett's esophagus: results from a US Multicenter Consortium. Gastroenterology. 2013; 145:79–86. PMID: 23499759.

15. Wallace MB, Crook JE, Saunders M, et al. Multicenter, randomized, controlled trial of confocal laser endomicroscopy assessment of residual metaplasia after mucosal ablation or resection of GI neoplasia in Barrett's esophagus. Gastrointest Endosc. 2012; 76:539–547. PMID: 22749368.

16. Uedo N, Iishi H, Tatsuta M, et al. Longterm outcomes after endoscopic mucosal resection for early gastric cancer. Gastric Cancer. 2006; 9:88–92. PMID: 16767363.

17. Capelle LG, Haringsma J, de Vries AC, et al. Narrow band imaging for the detection of gastric intestinal metaplasia and dysplasia during surveillance endoscopy. Dig Dis Sci. 2010; 55:3442–3448. PMID: 20393882.

18. Kato M, Kaise M, Yonezawa J, et al. Magnifying endoscopy with narrow-band imaging achieves superior accuracy in the differential diagnosis of superficial gastric lesions identified with white-light endoscopy: a prospective study. Gastrointest Endosc. 2010; 72:523–529. PMID: 20598685.

19. Guo YT, Li YQ, Yu T, et al. Diagnosis of gastric intestinal metaplasia with confocal laser endomicroscopy in vivo: a prospective study. Endoscopy. 2008; 40:547–553. PMID: 18618938.

20. Bok GH, Jeon SR, Cho JY, et al. The accuracy of probe-based confocal endomicroscopy versus conventional endoscopic biopsies for the diagnosis of superficial gastric neoplasia (with videos). Gastrointest Endosc. 2013; 77:899–908. PMID: 23473002.

21. Pittayanon R, Rerknimitr R, Wisedopas N, et al. The learning curve of gastric intestinal metaplasia interpretation on the images obtained by probe-based confocal laser endomicroscopy. Diagn Ther Endosc. 2012; 2012:278045. PMID: 23243385.

22. Gorospe EC, Leggett CL, Sun G, et al. Diagnostic performance of two confocal endomicroscopy systems in detecting Barrett's dysplasia: a pilot study using a novel bioprobe in ex vivo tissue. Gastrointest Endosc. 2012; 76:933–938. PMID: 22980290.

Go to : 




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download