INTRODUCTION
Gastric mesenchymal tumors are usually found incidentally during endoscopy for unrelated conditions, and appear as firm, protruding subepithelial masses. The entities that are responsible for mesenchymal tumors include leiomyomas, gastrointestinal stromal tumors (GISTs), and schwannomas. Among these entities, GISTs are the most common gastric mesenchymal tumors that have a malignant potential [
1,
2].
Schwannomas are tumors of spindle cells that originate from any nerve having a Schwann cell sheath, which are also known as neurinomas or neurilemmomas. Schwannomas in the gastrointestinal tract are rare (approximately 3% of all gastrointestinal mesenchymal tumors), and typically occur in the stomach [
3,
4]. Gastric schwannomas arise from the sheath of Auerbach’s plexus or, less frequently, from Meissner’s plexus [
5]. Schwannomas are generally benign, usually asymptomatic, slow-growing mesenchymal tumors with a very low malignant potential, and they have an excellent prognosis after surgical resection [
3]. However, these tumors are often misdiagnosed as GISTs. It is therefore important to accurately differentiate schwannomas from GISTs.
Endoscopic ultrasonography (EUS) is a valuable imaging modality for diagnosing and evaluating gastric mesenchymal tumors, because they appear as hypoechoic masses contiguous with the proper muscle layer of the normal gut wall on EUS [
6]. Although a definitive diagnosis of a gastric mesenchymal tumor is made via microscopic examination and immunohistochemical staining [
3], the evaluation of EUS findings regarding tumor size or pattern of echogenicity can be helpful in the differential diagnosis of mesenchymal tumors. However, only a few studies have examined the characteristic EUS findings of gastric schwannomas [
7-
9]. Thus, it is important to develop a better understanding of the EUS features of gastric schwannomas; differentiating them from other mesenchymal tumors (especially GISTs) can prevent inappropriate surgery and facilitate the use of minimal invasive treatment. Therefore, the present study aimed to evaluate the characteristic EUS findings of gastric schwannoma.
Go to :

DISCUSSION
In clinical practice, it is difficult to preoperatively differentiate between schwannomas and GISTs, even if EUS-guided performed [
12-
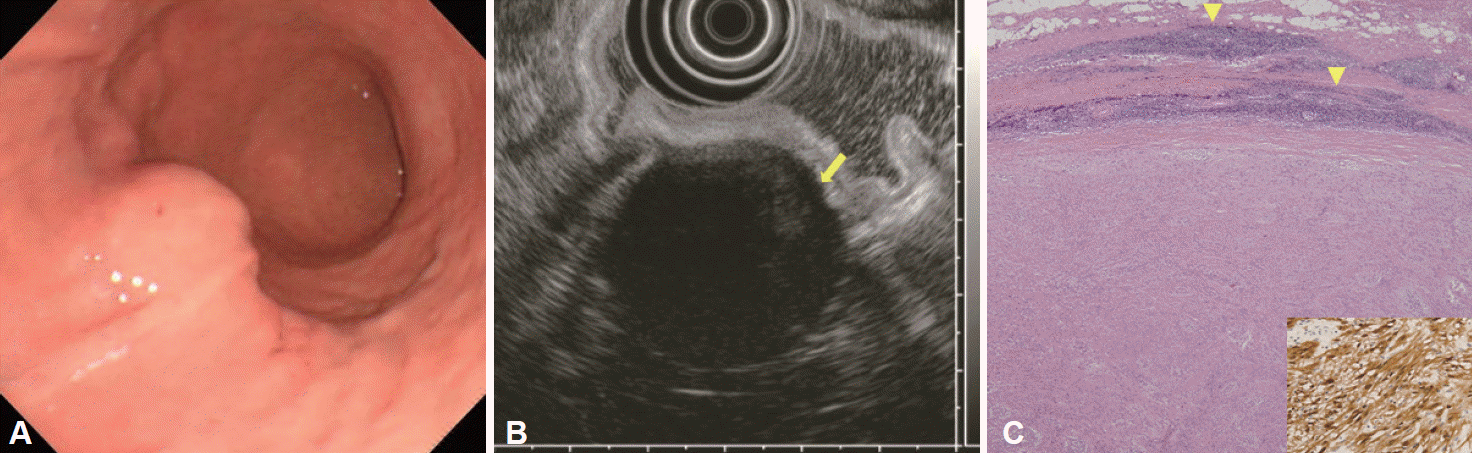
14]. Thus, identifying characteristic findings for schwannomas via non-invasive imaging techniques would be helpful for treating patients with gastric mesenchymal tumors. Here, we investigated the characteristic EUS features of gastric schwannomas using EUS, which is useful for diagnosing subepithelial lesions. Our findings indicate that gastric schwannomas were heterogeneously hypoechoic lesions with distinct borders, marginal haloes, and decreased echogenicity compared to that of the surrounding normal proper muscle layer.
Gastric schwannomas occur most frequently during the fourth and fifth decades of life, and predominantly in women [
15]. Because most patients with gastric schwannoma are asymptomatic, these lesions are usually diagnosed incidentally during upper endoscopy for unrelated symptoms. However, large tumors or tumors with mucosal ulcerations can occasionally cause bleeding [
11]. Similarly, we observed a female:male ratio of 1.5:1 and a mean patient age of 53 years. Furthermore, only four of our patients (15%) were symptomatic, and their symptoms were epigastric pain or dyspepsia.
Computed tomography (CT), magnetic resonance imaging (MRI), and positron emission tomography (PET) provide limited information for differentiating schwannomas from other mesenchymal tumors in the stomach. For example, previous CT studies have demonstrated that gastric schwannomas were well-circumscribed masses showing homogeneous or heterogeneous contrast enhancement [
16,
17]. In addition, MRI studies have revealed that gastric schwannomas were well demarcated and strongly enhanced tumors, with low-to-medium signal intensity on T1-weighted images and high signal intensity on T2-weighted images [
18]. However, these radiological findings for gastric schwannomas are not specific, and are very similar to those of GISTs. Furthermore, several recent reports have showed gastric schwannomas with increased fluorodeoxyglucose uptake during PET, which is similar to the findings for gastric GISTs [
18,
19].
Although EUS is a useful imaging technique for diagnosing and evaluating gastric mesenchymal tumors, only a few reports have been published regarding the EUS characteristics of gastric schwannomas because of the rarity of these tumors. For example, EUS studies have described gastric schwannomas as round homogenous masses with marginal haloes and without internal echogenic foci [
7,
20]. In addition, the echogenicity of gastric schwannomas was lower than that of the surrounding normal muscle layers. In other studies, EUS evaluation of gastric schwannomas revealed heterogeneous and hypoechoic masses with some hyperechogenic foci [
8,
9]. In the present study, most of the gastric schwannomas exhibited heterogeneous and hypoechoic echogenicity with internal hyperechogenic foci and marginal haloes.
In the present study, we paid careful attention to the tumor echogenicity because almost all gastric mesenchymal tumors appear as hypoechoic lesions during EUS. We therefore compared the echogenicity of the tumor to that of the surrounding normal proper muscle layer. In our previous study, although >50% of the GISTs exhibited increased echogenicity, compared to the surrounding normal proper muscle layer, the echogenicity of leiomyomas was similar to that of the surrounding normal proper muscle layer [
6]. In contrast, our present study revealed that two-thirds of the schwannoma exhibited lower echogenicity, compared to the surrounding normal proper muscle layer. This finding is consistent with the findings of our previous study regarding digital image analysis after standardized EUS images for gastric mesenchymal tumors, which revealed that the intensity values for schwannomas were lower than those for GISTs [
1].
Interestingly, it has also been reported that EUS findings reflect the pathologic features of gastric schwannoma [
7,
20]. For example, the trend towards a lower echogenicity for schwannomas in the present study may be related to a reduction in acoustic impedance because of the dense composition of spindle cells [
21]. In addition, approximately 90% of the gastric schwannomas in the present study had marginal haloes, which may correspond to the peripheral lymphoid cuff in our histological analysis [
20]. Nevertheless, despite marginal haloes being extremely common among schwannomas, this feature is not specific to schwannomas, and is frequently observed in GISTs [
6].
Previous EUS studies have also suggested that exogastric growth, cystic changes within the tumor, hyperechogenic foci in the tumor, and lobulation of the tumor margins favor the diagnosis of malignant mesenchymal tumors, such as GISTs [
22-
24]. Our results indicated that these features might also be useful in the identification of gastric schwannomas, as extraluminal growth and hyperechogenic foci within the tumor were common for these lesions, although cystic changes and lobulated margins were uncommon.
A definitive diagnosis of a gastric mesenchymal tumor is typically confirmed via microscopic histopathological examination and immunohistochemical staining. Tissue acquisition can be obtained via EUS-guided tissue sampling, endoscopic resection, or surgical resection. These tumors are typically negative for c-kit, desmin, and SMA; are typically positive for S-100; and stain variably for CD34 [
25]. In the present study, all cases were negative for c-kit and positive for S-100, and most cases (25 of 27) were negative for desmin, SMA, and CD34. However, the diagnostic accuracy of EUS-guided tissue sampling was only ~50% to 60%, due to technical failure and inadequate sampling [
26]. Therefore, we believe that our results regarding the EUS features of gastric schwannoma may be useful for managing patients with gastric mesenchymal tumors.
This study had several limitations. Firstly, we used a retrospective design to identify the EUS features of gastric schwannomas, and there might have been bias during the retrospective review of the EUS images. However, at least 10 images were obtained during EUS examination, and this may partially compensate for any bias that was related to the retrospective design. Secondly, although EUS examinations were performed, the patients were only selected for surgery based on the clinical opinion and decision of their physician. Finally, the number of gastric schwannomas included in the present study is somewhat small, because schwannomas are relatively rare lesions in the stomach compared to GISTs. Therefore, prospective multi-center studies are needed to validate our findings regarding the EUS features of gastric schwannoma.
In conclusion, our examination of EUS images revealed that gastric schwannomas were heterogeneously hypoechoic lesions with distinct margins and marginal haloes that originate in the proper muscle layer. The echogenicity of these lesions was lower than that of the surrounding normal proper muscle layer, and cystic changes and calcifications were rare. Although more invasive procedures may be needed to obtain tissue samples in some cases, these EUS features may be helpful for differentiating gastric schwannomas from other mesenchymal tumors, especially GISTs.
Go to :





 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download