Schwannomas are uncommon neoplasms arising from Schwann cells, which cover the peripheral nerves, and are difficult to distinguish from other mesenchymal tumors. The most common GI site of schwannomas is the stomach, and the duodenum is extremely rare [
4]. Duodenal schwannomas are mostly located in the second or third portion of the duodenum [
5]. Nilsson et al. reported that, of 43 small intestinal schwannomas, eight were located in the duodenum [
6]. There was no difference in the incidence between men and women, and most occurred in the fifth to sixth decades of life [
3]. Our patient was in the sixth decade and the tumor was located in the duodenal bulb, which differs from that which has been reported in the literature. Duodenal schwannomas are usually asymptomatic and can be discovered incidentally. If symptoms are present, the most common symptom is intestinal hemorrhage and abdominal discomfort [
5]. Duodenal schwannomas are usually benign, and malignant transformation is very rare. There are no histologic criteria to determine the grade of schwannoma and the metastatic potential is not yet known. However, malignant changes are reported in peripheral schwannomas [
7]. Therefore, careful observation after surgery is sometimes needed.
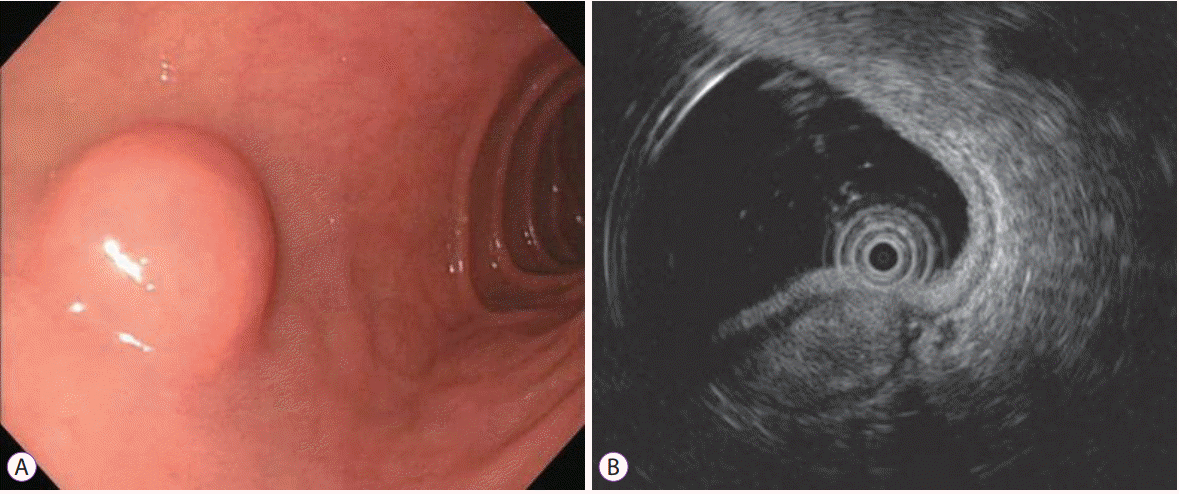
For diagnosis of schwannomas, endoscopy, EUS, abdominal CT, and abdominal magnetic resonance imaging (MRI) are useful to determine localization, relationship of surrounding organs, tumor multiplicity, or metastasis. Since the growth of the schwannomas is subepithelial, endoscopic evaluation is difficult to distinguish schwannomas from other mesenchymal tumors, such as GISTs, leiomyomas, or leiomyosarcomas. Furthermore, endoscopic biopsy is not suitable for definitive diagnosis because the schwannoma is a subepithelial tumor and mucosal abnormalities are rarely seen [
8]. EUS and EUS-guided fine-needle biopsy (EUS-FNB) are very valuable techniques for performing directed biopsies when necessary [
9]. However, any typical EUS features of duodenal schwannomas have not been reported due to the rarity of these tumors. In the case of gastric schwannomas, several typical EUS features, such as heterogeneously or homogenously hypoechoic lesions with marginal haloes, and few internal echogenic foci are reported [
10-
12]. CT is the most common method for evaluating neurogenic tumors and is useful for distinguishing benign and malignant neoplasms and predicting preoperative stage. On CT, the schwannomas are seen as homogeneously enhancing masses with a round or oval shape, which project into the lumen of the small intestine [
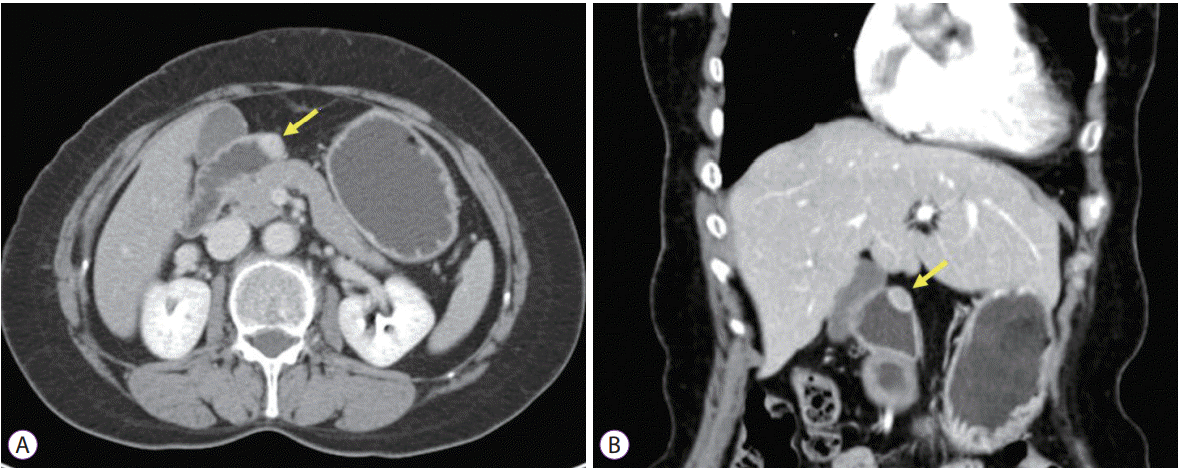
13]. In MRI, GI schwannomas are sharply demarcated and strongly enhancing tumors, with low to medium signal intensity on T1-weighted images and high signal intensity on T2-weighted images [
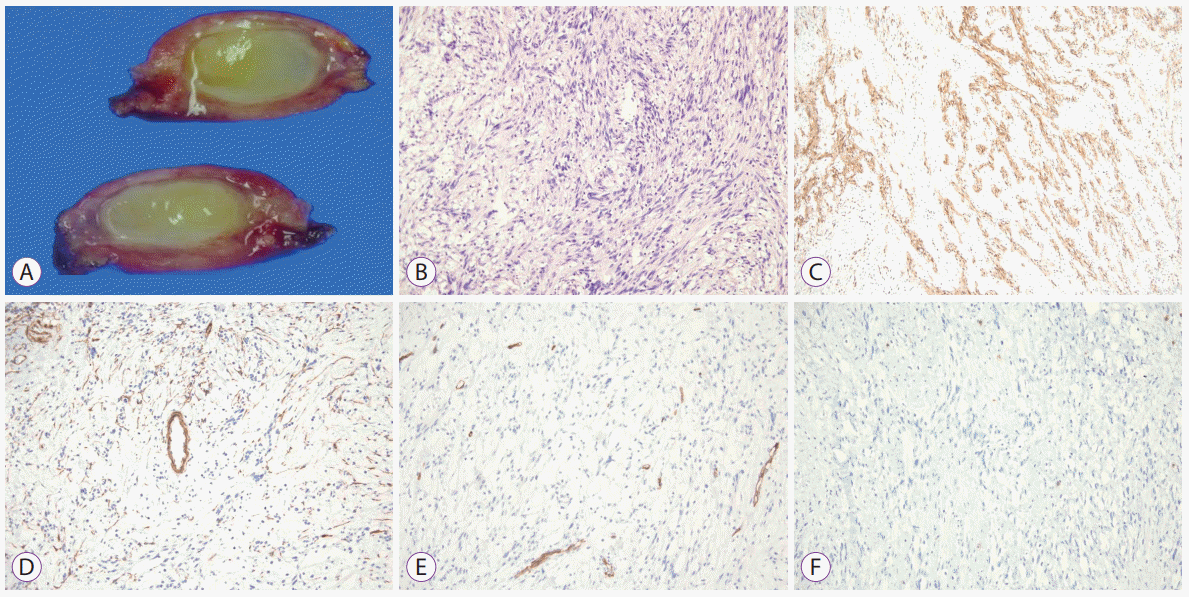
14]. However, schwannomas cannot be distinguished from malignant neurogenic tumors by radiologic images alone. Therefore, histological examination is essential for definitive diagnosis. Schwannomas are encased by the intact mucosa and usually involve the submucosa and muscularis propria. These tumors are composed of spindle-shaped nuclei with high and low cellularity regions called Antoni A and Antoni B areas. Conventional hematoxylin and eosin staining dose not distinguish neurogenic and myogenic tumors. Immunohistochemistry is essential to distinguishing between schwannomas and GISTs or leiomyomas. Cells of schwannoma are 100% immunoreactive with S-100 protein. GISTs are usually positive for c-kit and CD34, but negative for S-100 protein, whereas leiomyomas are positive for smooth muscle actin and desmin, and negative for S-100 protein [
1]. The treatment of choice is complete surgical resection, with an approach that depends on tumor size, localization, and histological features. The optimal treatment for a malignant schwannoma has not been fully established [
3]. The role of chemotherapy and radiotherapy remains unclear. Incomplete resection can be associated with local recurrence. Thus, surgical margins have been regarded as the most important prognostic factor.
In conclusion, we report a very rare case of schwannoma that arises in the duodenal bulb. Since schwannomas are difficult to distinguish from other mesenchymal tumors, diagnosis and treatment may be delayed. A definitive diagnosis is difficult before surgical resection. Imaging studies with abdominal CT, EUS and MRI and recently EUS-FNB can increase the diagnosis rate before operation.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download