1. Yamamoto H, Sekine Y, Sato Y, et al. Total enteroscopy with a nonsurgical steerable double-balloon method. Gastrointest Endosc. 2001; 53:216–220.

2. Ell C, May A. Mid-gastrointestinal bleeding: capsule endoscopy and push-and-pull enteroscopy give rise to a new medical term. Endoscopy. 2006; 38:73–75.

3. Gerson LB, Fidler JL, Cave DR, Leighton JA. ACG clinical guideline: diagnosis and management of small bowel bleeding. Am J Gastroenterol. 2015; 110:1265–1287. quiz 1288.

4. Ohmiya N. Management of obscure gastrointestinal bleeding: comparison of guidelines between Japan and other countries. Dig Endosc. 2020; 32:204–218.

5. Xin L, Liao Z, Jiang YP, Li ZS. Indications, detectability, positive findings, total enteroscopy, and complications of diagnostic double-balloon endoscopy: a systematic review of data over the first decade of use. Gastrointest Endosc. 2011; 74:563–570.

6. Ohmiya N, Nakamura M, Osaki H, et al. Development of a comorbidity index to identify patients with small bowel bleeding at risk for rebleeding and small bowel vascular diseases. Clin Gastroenterol Hepatol. 2019; 17:896–904.e4.

7. Ohmiya N, Nakagawa Y, Nagasaka M, et al. Obscure gastrointestinal bleeding: diagnosis and treatment. Dig Endosc. 2015; 27:285–294.
8. Yamamoto H, Ogata H, Matsumoto T, et al. Clinical practice guideline for enteroscopy. Dig Endosc. 2017; 29:519–546.

9. Murphy B, Winter DC, Kavanagh DO. Small bowel gastrointestinal bleeding diagnosis and management-a narrative review. Front Surg. 2019; 6:25.

10. Wang Z, Chen JQ, Liu JL, Qin XG, Huang Y. CT enterography in obscure gastrointestinal bleeding: a systematic review and meta-analysis. J Med Imaging Radiat Oncol. 2013; 57:263–273.

11. Arieira C, Monteiro S, Dias de Castro F, et al. Capsule endoscopy: is the software TOP 100 a reliable tool in suspected small bowel bleeding? Dig Liver Dis. 2019; 51:1661–1664.

12. Liao Z, Gao R, Xu C, Li ZS. Indications and detection, completion, and retention rates of small-bowel capsule endoscopy: a systematic review. Gastrointest Endosc. 2010; 71:280–286.

13. Hong SN, Kim ER, Ye BD, et al. Indications, diagnostic yield, and complication rate of balloon-assisted enteroscopy (BAE) during the first decade of its use in Korea. Dig Endosc. 2016; 28:443–449.
14. Wadhwa V, Sethi S, Tewani S, et al. A meta-analysis on efficacy and safety: single-balloon vs. double-balloon enteroscopy. Gastroenterol Rep (Oxf). 2015; 3:148–155.

15. Chu Y, Wu S, Qian Y, et al. Complimentary imaging modalities for investigating obscure gastrointestinal bleeding: capsule endoscopy, double-balloon enteroscopy, and computed tomographic enterography. Gastroenterol Res Pract. 2016; 2016:8367519.

16. Chen X, Ran ZH, Tong JL. A meta-analysis of the yield of capsule endoscopy compared to double-balloon enteroscopy in patients with small bowel diseases. World J Gastroenterol. 2007; 13:4372–4378.

17. Ribeiro I, Pinho R, Rodrigues A, Marqués J, Fernandes C, Carvalho J. Obscure gastrointestinal bleeding: which factors are associated with positive capsule endoscopy findings? Rev Esp Enferm Dig. 2015; 107:334–339.
18. Liu Y, Jiang W, Chen G, Li Y. Diagnostic value and safety of emergency single-balloon enteroscopy for obscure gastrointestinal bleeding. Gastroenterol Res Pract. 2019; 2019:9026278.

19. Shinozaki S, Yamamoto H, Yano T, et al. Long-term outcome of patients with obscure gastrointestinal bleeding investigated by double-balloon endoscopy. Clin Gastroenterol Hepatol. 2010; 8:151–158.

20. Otani K, Watanabe T, Shimada S, et al. Usefulness of small bowel reexamination in obscure gastrointestinal bleeding patients with negative capsule endoscopy findings: comparison of repeat capsule endoscopy and double-balloon enteroscopy. United European Gastroenterol J. 2018; 6:879–887.

21. Shinozaki S, Yano T, Sakamoto H, et al. Long-term outcomes in patients with overt obscure gastrointestinal bleeding after negative double-balloon endoscopy. Dig Dis Sci. 2015; 60:3691–3696.

22. Leung WK, Ho SS, Suen BY, et al. Capsule endoscopy or angiography in patients with acute overt obscure gastrointestinal bleeding: a prospective randomized study with long-term follow-up. Am J Gastroenterol. 2012; 107:1370–1376.
23. Alavi A, Dann RW, Baum S, Biery DN. Scintigraphic detection of acute gastrointestinal bleeding. Radiology. 1977; 124:753–756.

24. Bonnet S, Douard R, Malamut G, Cellier C, Wind P. Intraoperative enteroscopy in the management of obscure gastrointestinal bleeding. Dig Liver Dis. 2013; 45:277–284.

25. Hayashi Y, Yamamoto H, Yano T, Sugano K. Diagnosis and management of mid-gastrointestinal bleeding by double-balloon endoscopy. Therap Adv Gastroenterol. 2009; 2:109–117.
26. Wallace JL, Syer S, Denou E, et al. Proton pump inhibitors exacerbate NSAID-induced small intestinal injury by inducing dysbiosis. Gastroenterology. 2011; 141:1314–1322, 1322.e1-e5.

27. Baptista V, Marya N, Singh A, Rupawala A, Gondal B, Cave D. Continuing challenges in the diagnosis and management of obscure gastrointestinal bleeding. World J Gastrointest Pathophysiol. 2014; 5:523–533.

28. Becq A, Rahmi G, Perrod G, Cellier C. Hemorrhagic angiodysplasia of the digestive tract: pathogenesis, diagnosis, and management. Gastrointest Endosc. 2017; 86:792–806.

29. Albitar HAH, Almodallal Y, Papadakis KA, Rajan E, Kamath PS, Iyer VN. Intravenous bevacizumab reduces transfusion requirements and endoscopic interventions in patients with gastric antral vascular ectasia and small bowel angioectasia. Gastroenterology. 2020; 158:1162–1163.e4.

30. Tsuchiya S, Matsumoto Y, Doman T, et al. Disappearance of angiodysplasia following transcatheter aortic valve implantation in a patient with Heyde’s syndrome: a case report and review of the literature. J Atheroscler Thromb. 2020; 27:271–277.

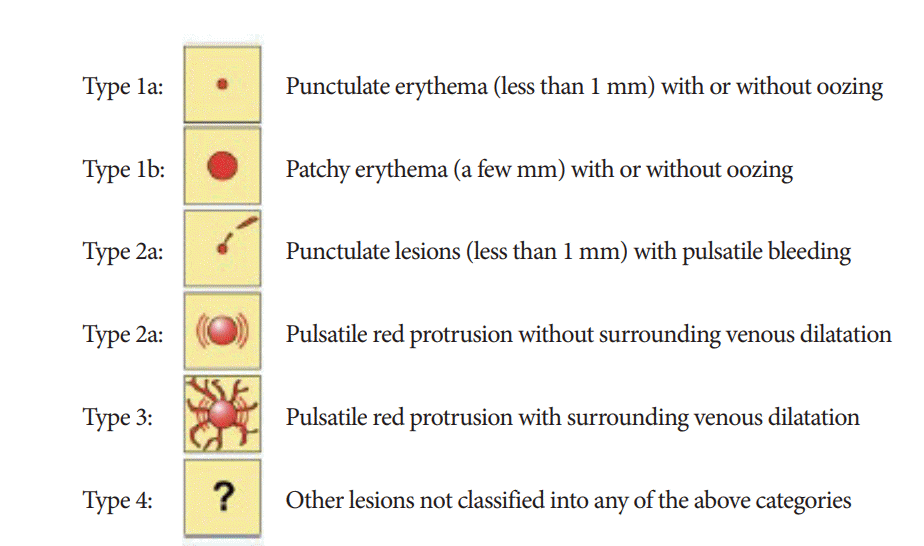
31. Yano T, Yamamoto H, Sunada K, et al. Endoscopic classification of vascular lesions of the small intestine (with videos). Gastrointest Endosc. 2008; 67:169–172.

32. Kushnir VM, Tang M, Goodwin J, et al. Long-term outcomes after single-balloon enteroscopy in patients with obscure gastrointestinal bleeding. Dig Dis Sci. 2013; 58:2572–2579.

33. Yano T, Nemoto D, Ono K, et al. Gel immersion endoscopy: a novel method to secure the visual field during endoscopy in bleeding patients (with videos). Gastrointest Endosc. 2016; 83:809–811.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download