1. Pemberton M, Wells AD. The Mirizzi syndrome. Postgrad Med J. 1997; 73:487–490.

2. Chen H, Siwo EA, Khu M, Tian Y. Current trends in the management of Mirizzi Syndrome: a review of literature. Medicine (Baltimore). 2018; 97:e9691.
3. Yuan H, Yuan T, Sun X, Zheng M. A minimally invasive strategy for Mirizzi syndrome type II: combined endoscopic with laparoscopic approach. Surg Laparosc Endosc Percutan Tech. 2016; 26:248–252.

4. Zheng M, Cai W, Qin M. Combined laparoscopic and endoscopic treatment for Mirizzi syndrome. Hepatogastroenterology. 2011; 58:1099–1105.
5. Ishida Y, Itoi T, Okabe Y. Current status and future perspective in cholangiopancreatoscopy. Curr Treat Options Gastroenterol. 2019; 17:327–341.

6. Csendes A, Díaz JC, Burdiles P, Maluenda F, Nava O. Mirizzi syndrome and cholecystobiliary fistula: a unifying classification. Br J Surg. 1989; 76:1139–1143.

7. Zhao J, Fan Y, Wu S. Safety and feasibility of laparoscopic approaches for the management of Mirizzi syndrome: a systematic review. Surg Endosc. 2020; 34:4717–4726.

8. Beltrán MA. Mirizzi syndrome: history, current knowledge and proposal of a simplified classification. World J Gastroenterol. 2012; 18:4639–4650.

9. Williamson JB, Draganov PV. The usefulness of SpyGlass™ choledochoscopy in the diagnosis and treatment of biliary disorders. Curr Gastroenterol Rep. 2012; 14:534–541.

10. Binmoeller KF, Thonke F, Soehendra N. Endoscopic treatment of Mirizzi’s syndrome. Gastrointest Endosc. 1993; 39:532–536.

11. Tsuyuguchi T, Sakai Y, Sugiyama H, Ishihara T, Yokosuka O. Long-term follow-up after peroral cholangioscopy-directed lithotripsy in patients with difficult bile duct stones, including Mirizzi syndrome: an analysis of risk factors predicting stone recurrence. Surg Endosc. 2011; 25:2179–2185.

12. Issa H, Bseiso B, Al-Salem AH. Successful laser lithotripsy using peroral SpyGlass cholangioscopy in a patient with Mirizzi syndrome. Endoscopy. 2011; 43(Suppl 2):E166–E167.

13. Bhandari S, Bathini R, Sharma A, Maydeo A. Usefulness of single-operator cholangioscopy-guided laser lithotripsy in patients with Mirizzi syndrome and cystic duct stones: experience at a tertiary care center. Gastrointest Endosc. 2016; 84:56–61.

14. Yasuda I, Itoi T. Recent advances in endoscopic management of difficult bile duct stones. Dig Endosc. 2013; 25:376–385.

15. Itoi T, Coelho-Prabhu N, Baron TH. Endoscopic gallbladder drainage for management of acute cholecystitis. Gastrointest Endosc. 2010; 71:1038–1045.

16. Ridtitid W, Piyachaturawat P, Teeratorn N, Angsuwatcharakon P, Kongkam P, Rerknimitr R. Single-operator peroral cholangioscopy cystic duct cannulation for transpapillary gallbladder stent placement in patients with acute cholecystitis at moderate to high surgical risk (with videos). Gastrointest Endosc. 2020; 92:634–644.
17. Shin JU, Lee JK, Kim KM, Lee KH, Lee KT. Endoscopic naso-gallbladder drainage with the Spyglass cholangioscopy system. The Korean Journal of Pancreas and Biliary Tract. 2011; 16:139–144.
18. Antoniou SA, Antoniou GA, Makridis C. Laparoscopic treatment of Mirizzi syndrome: a systematic review. Surg Endosc. 2010; 24:33–39.

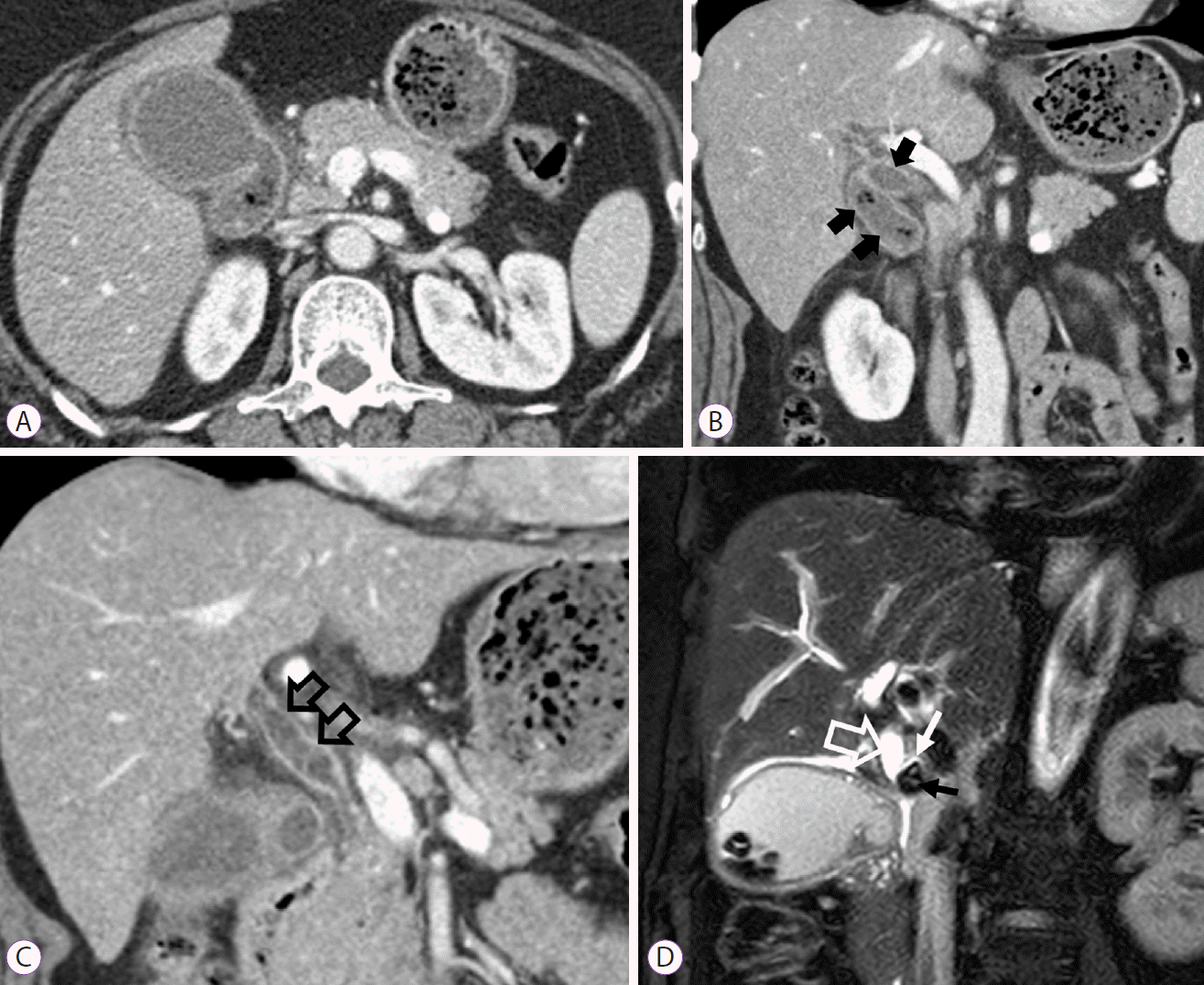
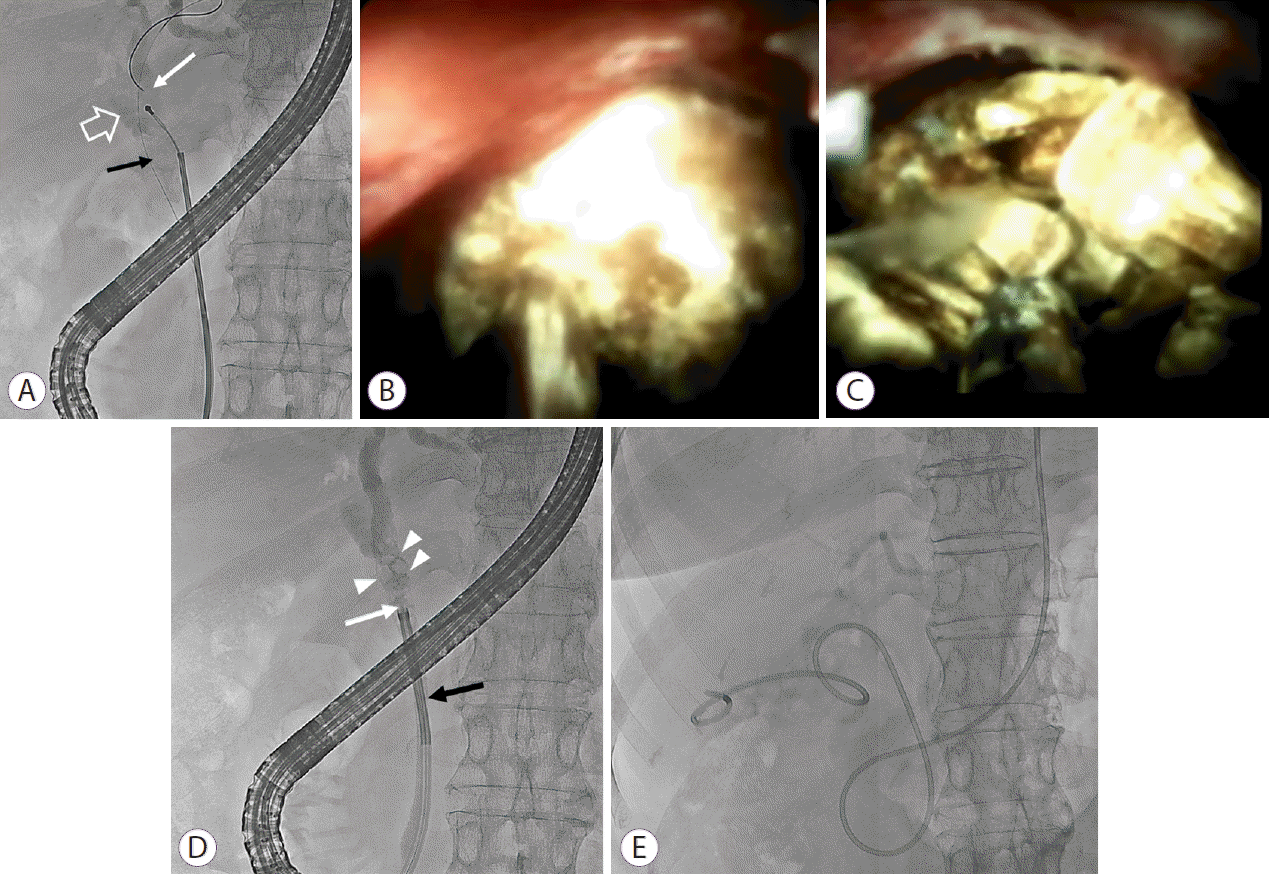




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download