INTRODUCTION
Since first described by Isaacson and Wright in 1983 [
1], mucosa-associated lymphoid tissue (MALT) lymphoma was classified as extranodal marginal zone B-cell lymphoma of the MALT type by the World Health Organization in 2008 [
2]. MALT lymphoma accounts for approximately 9% of non-Hodgkin lymphomas [
3]. Although the stomach is the most frequent site of involvement, MALT lymphoma can occur in various organs, including the lungs, lacrimal gland, salivary glands, and thyroid [
3]. Among the gastrointestinal tract, colorectal MALT lymphoma is rarely found [
3].
As most gastric MALT lymphomas are closely related to
Helicobacter pylori infection, antimicrobial therapy against
H. pylori is the first-line therapy. Different from gastric MALT lymphoma,
H. pylori infection might play a minor role in the treatment of colonic MALT lymphoma. Considering the rarity of colonic MALT lymphoma, its definitive treatment has not been established. If colonic MALT lymphoma is localized or low stage, various treatment approaches have been described, including careful waiting, chemotherapy, surgical resection, radiation, endoscopic resection, or combined surgery and radiation [
4].
We present a case of residual colonic MALT lymphoma after endoscopic mucosal resection, which was a 1.5-cm-sized tumor confined to the superficial wall of the rectum. The lesion was successfully treated using the endoscopic submucosal dissection (ESD) technique. Finally, we recommend the treatment option for localized colorectal MALT lymphoma.
Go to :

CASE REPORT
A 54-year-old man was referred to our hospital for an incomplete polypectomy during screening colonoscopy. He underwent endoscopic mucosal resection for a 3-cm-sized colonic elongated, pedunculated polyp from a primary clinic. Pathology revealed MALT lymphoma with positive resection margin and with incomplete resection. He had no family history and no medical history. His physical examination was normal without evidence of peripheral adenopathy or splenomegaly. Urea breath test was negative for
H. pylori infection. On colonoscopy, a 1.5-cm residual, nodular sessile tumor (type Is from Paris classification) [
5] with scar tissue was identified in the upper rectum, 15 cm from the anal verge (
Fig. 1A).
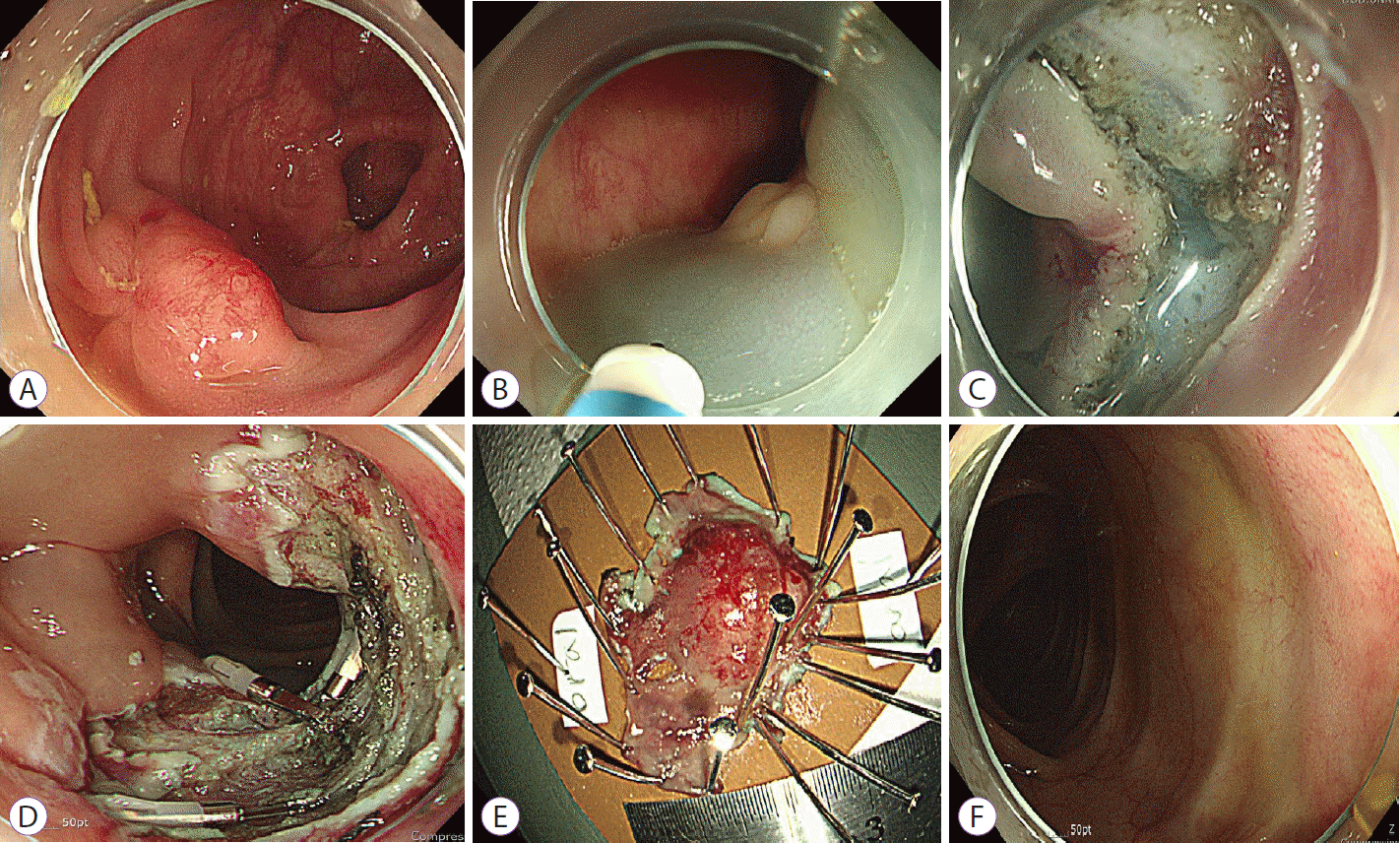 | Fig. 1.Endoscopic submucosal dissection for mucosa-associated lymphoid tissue lymphoma. (A) Colonoscopy revealed a 1.5-cm residual nodular sessile tumor with scar tissue. Multiple red spots and capillaries were noted on the tumor surface. (B) Normal saline mixed with indigo carmine was injected into the submucosal layer. (C) Circumferential mucosal incision and submucosal dissection were performed with a Dual knife. (D) Complete submucosal dissection was performed. (E) En bloc resected tumor. (F) At the 1-year follow-up, colonoscopy revealed a white scar without recurrence. 
|
Chest computed tomography (CT) and abdominal CT revealed no lymphadenopathy or hepatosplenomegaly. Bone marrow examination showed no involvement of MALT lymphoma. Positron emission tomography revealed no abnormal fluorodeoxyglucose uptake in the abdomen. As the initial polyp had a pedunculated shape, invasion depth would be till the mucosa or minute submucosal layer. Therefore, he had a colonic MALT lymphoma localized to the rectum, classified as stage IE (Lugano modification of Ann Arbor non-Hodgkin lymphoma stage) [
6]. After a discussion with oncologist and surgeon, we decided an ESD treatment as a curative therapy.
Under conscious sedation with intravenous midazolam, a transparent hood and carbon dioxide insufflation were used for the procedure. Normal saline mixed with diluted epinephrine (1:100,000) and indigo carmine was injected into the submucosal layer to facilitate submucosal dissection (
Fig. 1B). Circumferential mucosal incision and submucosal dissection were performed using a Dual knife (Olympus Korea, Seoul, Korea) (
Fig. 1C). During submucosal dissection, moderate submucosal fibrosis was identified where the scar was formed. Muscle injury was also identified, and prophylactic clips were placed. Complete submucosal dissection was performed, and the tumor was resected
en bloc (
Fig. 1D,
E). The total operation time was 60 minutes. The patient was discharged 2 days after the procedure, without any complications.
Histological examination revealed marked and dense infiltration of lymphocytes in the lamina propria and superficial submucosa (
Fig. 2A). Polymorphous lymphoid infiltrates including small lymphocytes, centroblast-like cells, and centrocyte-like cells were observed (
Fig. 2B). Immunohistochemical staining showed positive staining for CD20 and negative staining for CD3, compatible with MALT lymphoma. Resection margins for lateral and deep resections were clear. At the 1-year follow-up, surveillance colonoscopy and abdominal CT revealed no recurrence (
Fig. 1F). The patient remained disease-free for 4 years.
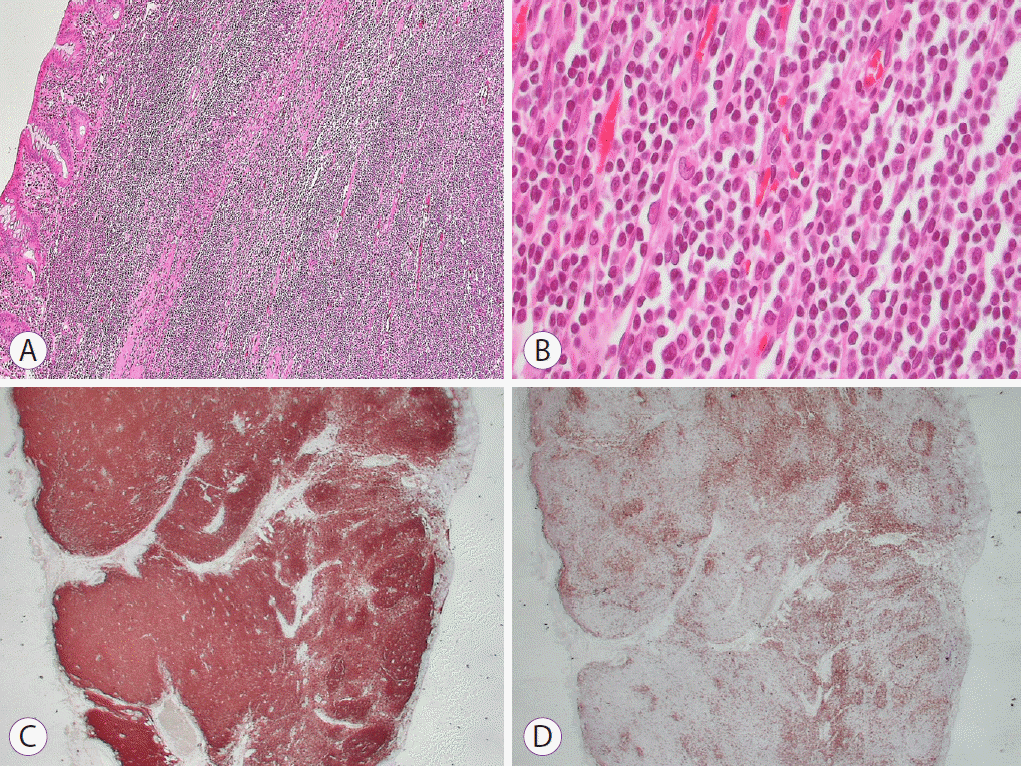 | Fig. 2.Pathologic findings. (A) Diffuse dense infiltration of atypical small lymphocytes (hematoxylin and eosin [H&E] stain, ×100). (B) Polymorphous lymphoid infiltrates including small lymphocytes, centroblast-like cells, and centrocyte-like cells were observed (H&E stain, ×400). (C, D) These lymphocytes immunohistochemically showed diffusely positive staining for CD20 (C) and negative staining for CD3 (D). 
|
Go to :

DISCUSSION
We treated successfully residual colonic MALT lymphoma using the ESD technique. ESD results in higher
en bloc resection rate and lower recurrence rate compared to endoscopic mucosal resection. ESD is indicated in large tumors over 2 cm, which requires piecemeal resection when endoscopic mucosal resection is used [
7]. In case of a recurrent/residual tumor after endoscopic mucosal resection, ESD may be a therapeutic option for curative or salvage therapy [
8,
9]. Nevertheless, colorectal ESD for residual tumors remains challenging because there are technical difficulties in dissecting severe submucosal fibrosis. Submucosal fibrosis has low
en bloc resection rate and high perforation rate [
10]. Fortunately, in our case, complete dissection was achieved because the scar tissue was small and submucosal fibrosis was in moderate degree. Certainly, our MALT lymphoma case treated with ESD cannot be generalized to all endoscopists. In Western countries, ESD technique still failed to achieve an acceptable level of performance in a systematic review [
11]. Proper training and expertise in perforation management are critical for a successful ESD procedure.
In the present case, the primary physician performed an incomplete colon polypectomy. A 3-cm-sized pedunculated polyp can be resected by snare polypectomy, not by the ESD technique. The success of endoscopic resection depends largely on the skill of the endoscopist. It is important to ensure sufficient resection margin free of tumor to prevent recurrence after polypectomy [
12]. A study showed that remnant tumor after polypectomy was related to endoscopist’s inexperience and increasing tumor size [
13]. An inexperienced endoscopist might be unfamiliar with this unusual type of polyp. In this study, MALT lymphoma had red spots and capillaries on the surface of the tumor, which can make it difficult to identify the tumor extent. Eventually, the initial attempt to complete resection had failed, and the tumor was partially resected.
Partial snare resection causes submucosal fibrosis, interfering with future endoscopic treatment [
7]. The ESD technique is required to resect residual tumors with a fibrous lesion [
8]. An endoscopy specialist should educate the referring physician to avoid such practice that may lead to scar formation and fibrosis.
We used a transparent cap attached to the end of the endoscope. Transparent caps are used to visualize the operative field and stabilize the ESD procedure [
14].
In addition to endoscopic therapy, trans-anal endoscopic microsurgery (TEM) and laparoscopic surgery may be treatment options to resect large rectal tumors. In a comparative systematic review, TEM and ESD have similar rates of resection, adverse events, and recurrence in large rectal tumors [
15]. ESD has more advantages compared to TEM or surgery, namely, ESD has a shorter procedure time and shorter hospital stay than TEM or surgery [
15]. For large rectal tumors, treatment options may be selected based on the level of expertise of the endoscopist and institutional resource.
In general, gastric MALT lymphomas are closely related to
H. pylori infection, and 80% of low-grade gastric MALT lymphomas regress in response to anti-
H. pylori eradication therapy [
16]. Different from gastric MALT lymphoma, colorectal MALT lymphoma does not appear to respond to
H. pylori eradication therapy. In a previous case report, rectal MALT lymphoma treated with
H. pylori eradication recurred at the same site 18 months later [
17].
Since colonic MALT lymphoma occurs rarely, there are no standardized guidelines for its treatment. Jeon et al. [
4] reported the largest single-center case-series of 51 patients with colorectal MALT lymphoma. Various therapies have been performed, including surgery, radiotherapy, chemotherapy, combined surgery and radiation, endoscopic resection, and watchful waiting. During the 46-month follow-up period, progression was observed in only 4 patients (8%) [
4]. Colorectal MALT lymphoma had a good prognosis. Its overall 5-year survival rate was 94% [
4].
To date, endoscopically resected colorectal MALT lymphomas were reported in several case studies [
18-
20]. A 2.5-cm broad base nodular polyp with indistinct borders was resected by snare polypectomy [
18]. A 1.0-cm sessile polyp with smooth edges was incidentally discovered and resected using snare cautery [
19]. A 2.0-cm semipedunculated polyp with red spots was resected by endoscopic mucosal resection technique [
20]. Among the various treatment options, endoscopic resection may be recommended because it is effective and less invasive when performed by a skilled hand. A single session of endoscopic therapy can cure the disease compared to chemotherapy or radiation therapy.
In conclusion, we report a case of residual rectal MALT lymphoma after endoscopic mucosal resection, which was successfully treated using the ESD technique. This case provides rationale for ESD as a salvage therapy for residual tumors in properly selected patients with colonic MALT lymphoma.
Go to :







 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download