Abstract
Purpose
Materials and Methods
References
Fig. 1
PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) flow diagram describing selection process for included studies.
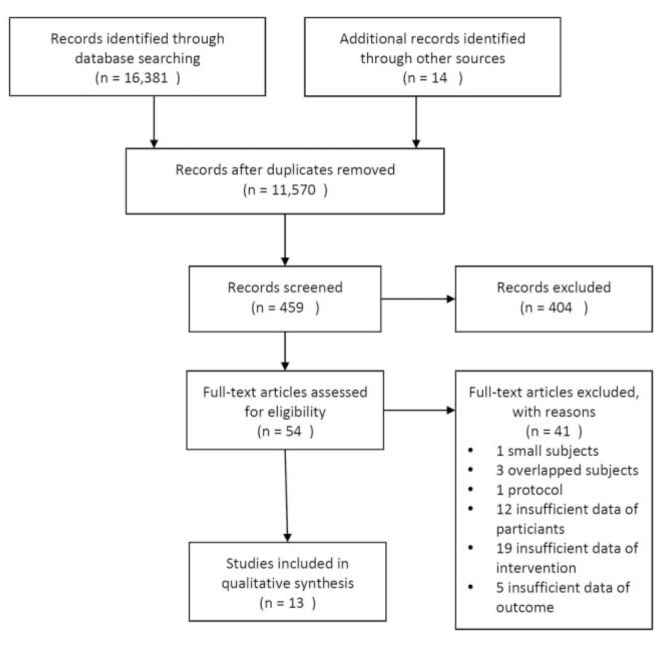
Fig. 2
Clinical outcomes of endovascular treatments in ischemic stroke patients with high ASPECTS vs. low ASPECTS A. Meta-analyses of high ASPECTS (>7) vs. low ASPECTS (≤7) for favorable clinical outcome (modified Rankin scale score 0-2) (A). The relative size of the data markers indicates the weight of each study's sample size. The included trials for functional outcome are divided into subgroups according to study design (prospective randomized controlled or single-arm trials and observational case series). B. A funnel plot with all points evenly distributed on both sides of the solid vertical line indicates no publication bias. C. Subgroup analysis of favorable outcomes of endovascular treatments in ischemic stroke patients with high ASPECTS vs. low ASPECTS. The included trials for functional outcome are divided into subgroups according to the methods of endovascular therapy (dominant intraarterial thrombolysis and dominant intraarterial thrombectomy). One study (23) which did not clearly present the endovascular methods was excluded from subgroup analysis for endovascular methods. D. Scatterplots of the relationship between the rate of ICA occlusion and log odds ratio. The size of the bubbles indicates the weight of each study in the meta-analysis. The trend line indicates the degree to which the log odds ratio decreases as the rate of ICA occlusion in subjects increases.

Fig. 3
Mortality and symptomatic intracranial hemorrhage of endovascular treatments in ischemic stroke patients with high ASPECTS vs. low ASPECTS.

Fig. 4
Comparison among very low, intermediate, and high ASPECTS.

Table 1
Designs of Included Studies in the Meta-analysis and Characteristics of Endovascular Treatment
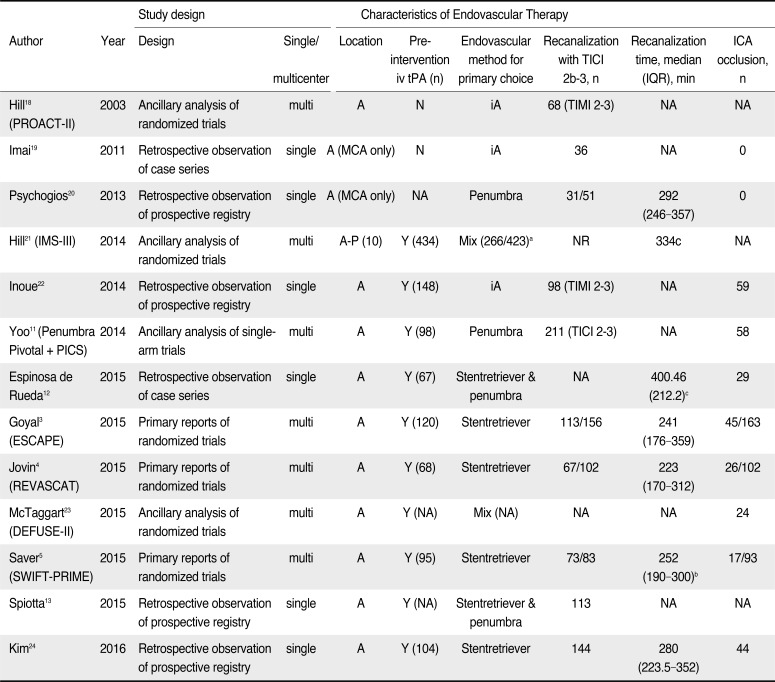
| Author | Year | Study design | Characteristics of Endovascular Therapy | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Design | Single/multicenter | Location | Preintervention iv tPA (n) | Endovascular method for primary choice | Recanalization with TICI 2b–3, n | Recanalization time, median (IQR), min | ICA occlusion, n | ||
| Hill18 (PROACT-II) | 2003 | Ancillary analysis of randomized trials | multi | A | N | iA | 68 (TIMI 2–3) | NA | NA |
| Imai19 | 2011 | Retrospective observation of case series | single | A (MCA only) | N | iA | 36 | NA | 0 |
| Psychogios20 | 2013 | Retrospective observation of prospective registry | single | A (MCA only) | NA | Penumbra | 31/51 | 292 (246–357) | 0 |
| Hill21 (IMS-III) | 2014 | Ancillary analysis of randomized trials | multi | A-P (10) | Y (434) | Mix (266/423)a | NR | 334c | NA |
| Inoue22 | 2014 | Retrospective observation of prospective registry | single | A | Y (148) | iA | 98 (TIMI 2–3) | NA | 59 |
| Yoo11 (Penumbra Pivotal + PICS) | 2014 | Ancillary analysis of single-arm trials | multi | A | Y (98) | Penumbra | 211 (TICI 2–3) | NA | 58 |
| Espinosa de Rueda12 | 2015 | Retrospective observation of case series | single | A | Y (67) | Stentretriever & penumbra | NA | 400.46 (212.2)c | 29 |
| Goyal3 (ESCAPE) | 2015 | Primary reports of randomized trials | multi | A | Y (120) | Stentretriever | 113/156 | 241 (176–359) | 45/163 |
| Jovin4 (REVASCAT) | 2015 | Primary reports of randomized trials | multi | A | Y (68) | Stentretriever | 67/102 | 223 (170–312) | 26/102 |
| McTaggart23 (DEFUSE-II) | 2015 | Ancillary analysis of randomized trials | multi | A | Y (NA) | Mix (NA) | NA | NA | 24 |
| Saver5 (SWIFT-PRIME) | 2015 | Primary reports of randomized trials | multi | A | Y (95) | Stentretriever | 73/83 | 252 (190–300)b | 17/93 |
| Spiotta13 | 2015 | Retrospective observation of prospective registry | single | A | Y (NA) | Stentretriever & penumbra | 113 | NA | NA |
| Kim24 | 2016 | Retrospective observation of prospective registry | single | A | Y (104) | Stentretriever | 144 | 280 (223.5–352) | 44 |
Abbreviations: CT, computed tomography; DEFUSE-II, The Diffusion and Perfusion Imaging Evaluation for Understanding Stroke Evaluation II; ESCAPE, Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness; EVT, endovascular treatment; IMS-III; Interventional Management of Stroke III; MR CLEAN, The Multicenter Randomized Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands; N, No; NIHSS, National Institutes of Health Stroke Scale; NR, not reported; PROACT-II, Prolyse in Acute Cerebral Thromboembolism II; PICS, Penumbra Imaging Collaborative Study; REVASCAT, Randomized Trial of Revascularization With Solitaire FR Device Versus Best Medical Therapy in the Treatment of Acute Stroke Due to Anterior Circulation Large Vessel Occlusion Presenting Within 8 Hours of Symptom Onset; SWIFT, SOLITAIRETM with the intention for thrombectomy; iA, intra-arterial thrombolytic drug infusion through catheter; TICI, Thrombolysis in Cerebral Infarction; TIMI, Thrombolysis In Myocardial Infarction; MCA, middle cerebral artery; ICA, internal carotid artery; Y, yes; N, no; NA, not applicable
a: Combinations of more than one endovascular therapy were used in 128 patients in total 266 patients with intraarterial tPA infusion.
b: The data of ‘recanalization time’ of this study means time interval from symptom onset to first stent deployment.
c. mean (standard deviation)
Table 2
Characteristics of Patients in Endovascular and Medical Treatment Arms
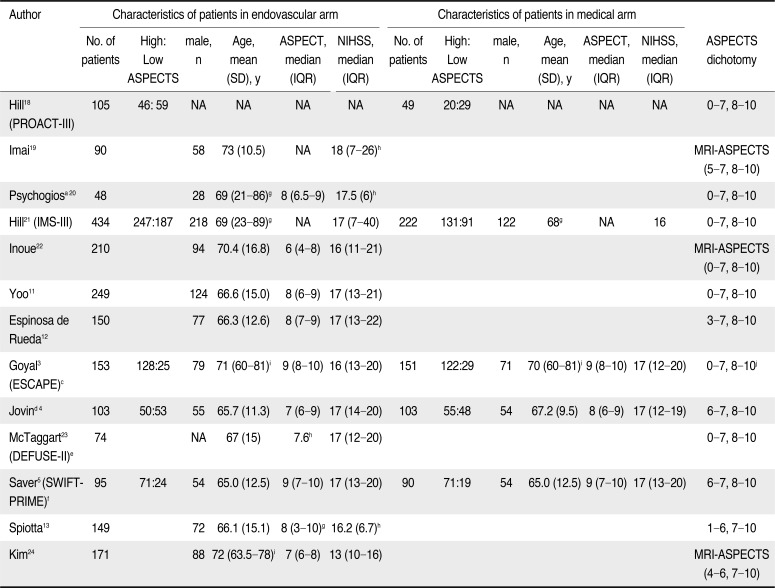
| Author | Characteristics of patients in endovascular arm | Characteristics of patients in medical arm | ASPECTS dichotomy | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No. of patients | High:Low ASPECTS | male, n | Age, mean (SD), y | ASPECT, median (IQR) | NIHSS, median (IQR) | No. of patients | High:Low ASPECTS | male, n | Age, mean (SD), y | ASPECT, median (IQR) | NIHSS, median (IQR) | ||
| Hill18 (PROACT-III) | 105 | 46: 59 | NA | NA | NA | NA | 49 | 20:29 | NA | NA | NA | NA | 0–7, 8–10 |
| Imai19 | 90 | 58 | 73 (10.5) | NA | 18 (7–26)h | MRI-ASPECTS (5–7, 8–10) | |||||||
| Psychogiosa 20 | 48 | 28 | 69 (21–86)g | 8 (6.5–9) | 17.5 (6)h | 0–7, 8–10 | |||||||
| Hill21 (IMS-III) | 434 | 247:187 | 218 | 69 (23–89)g | NA | 17 (7–40) | 222 | 131:91 | 122 | 68g | NA | 16 | 0–7, 8–10 |
| Inoue22 | 210 | 94 | 70.4 (16.8) | 6 (4–8) | 16 (11–21) | MRI-ASPECTS (0–7, 8–10) | |||||||
| Yoo11 | 249 | 124 | 66.6 (15.0) | 8 (6–9) | 17 (13–21) | 0–7, 8–10 | |||||||
| Espinosa de Rueda12 | 150 | 77 | 66.3 (12.6) | 8 (7–9) | 17 (13–22) | 3–7, 8–10 | |||||||
| Goyal3 (ESCAPE)c | 153 | 128:25 | 79 | 71 (60–81)i | 9 (8–10) | 16 (13–20) | 151 | 122:29 | 71 | 70 (60-81)i | 9 (8–10) | 17 (12–20) | 0–7, 8–10j |
| Jovind 4 | 103 | 50:53 | 55 | 65.7 (11.3) | 7 (6–9) | 17 (14–20) | 103 | 55:48 | 54 | 67.2 (9.5) | 8 (6–9) | 17 (12–19) | 6–7, 8–10 |
| McTaggart23 (DEFUSE-II)e | 74 | NA | 67 (15) | 7.6h | 17 (12–20) | 0–7, 8–10 | |||||||
| Saver5 (SWIFT-PRIME)f | 95 | 71:24 | 54 | 65.0 (12.5) | 9 (7–10) | 17 (13–20) | 90 | 71:19 | 54 | 65.0 (12.5) | 9 (7–10) | 17 (13–20) | 6–7, 8–10 |
| Spiotta13 | 149 | 72 | 66.1 (15.1) | 8 (3–10)g | 16.2 (6.7)h | 1–6, 7–10 | |||||||
| Kim24 | 171 | 88 | 72 (63.5–78)i | 7 (6–8) | 13 (10–16) | MRI-ASPECTS (4–6, 7–10) | |||||||
Abbreviations: A, anterior circulation; A-P, anterior and posterior circulation (number of posterior circulation); ASPECTS, Alberta Stroke Program Early CT Scale; CT, computed tomography; iA, intra-arterial thrombolysis; ICA, internal carotid artery; iv tPA; intravenous tissue plasminogen activator infusion; LAO, large artery occlusion; MCA. Middle cerebral artery; N, No; NA, not applicable; NIHSS, National Institutes of Health Stroke Scale; NR, not reported; SD, standard deviation; TICI, Thrombolysis in Cerebral Infarction; TIMI, Thrombolysis in Myocardial Infarction; Y, yes
a: Among 51 patients who were enrolled in this study, follow up mRS were available in 48 patients. The data of ‘characteristics of patients’ was collected from 51 patients.
b: Odds ratio was adjusted.
c: The information of characteristics of subjects were extracted from the per protocol population (165 and 150). Subjects which used in meta-analysis are provided in ancillary analysis including protocol violators and excluding follow-up loss.
d: Among 103 patients who were enrolled in this study, number of successful recanalization and ICA occlusion were available in 102 patients.
e: In DEFUSE 2 trial, Merci device, Penumbra system, and intra-arterial tPA were used to most of patients and only 6 stentrievers were used. Combinations of more than one endovascular therapy were used in 45%.
f. The information of characteristics of subjects were extracted from the per protocol population (98 and 98)
g. median (range)
h. mean (standard deviation)
i. median and interquartile range (IQR)
j. 9 protocol violators (0~5) included.
Table 3
Risk Assessment of Individual Studies Using the National Institute of Health Quality Assessment Tool for Observational Cohort and Cross-sectional Studies
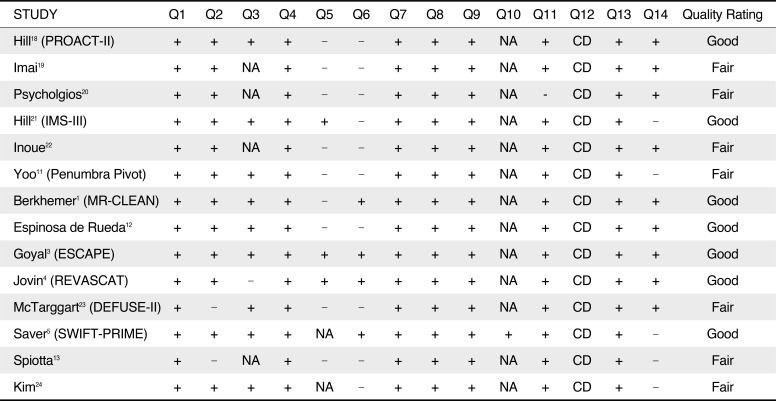
| STUDY | Q1 | Q2 | Q3 | Q4 | Q5 | Q6 | Q7 | Q8 | Q9 | Q10 | Q11 | Q12 | Q13 | Q14 | Quality Rating |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hill18 (PROACT-II) | + | + | + | + | - | - | + | + | + | NA | + | CD | + | + | Good |
| Imai19 | + | + | NA | + | - | - | + | + | + | NA | + | CD | + | + | Fair |
| Psycholgios20 | + | + | NA | + | - | - | + | + | + | NA | - | CD | + | + | Fair |
| Hill21 (IMS-III) | + | + | + | + | + | - | + | + | + | NA | + | CD | + | - | Good |
| Inoue22 | + | + | NA | + | - | - | + | + | + | NA | + | CD | + | + | Fair |
| Yoo11 (Penumbra Pivot) | + | + | + | + | - | - | + | + | + | NA | + | CD | + | - | Fair |
| Berkhemer1 (MR-CLEAN) | + | + | + | + | - | + | + | + | + | NA | + | CD | + | + | Good |
| Espinosa de Rueda12 | + | + | + | + | - | - | + | + | + | NA | + | CD | + | + | Good |
| Goyal3 (ESCAPE) | + | + | + | + | + | + | + | + | + | NA | + | CD | + | + | Good |
| Jovin4 (REVASCAT) | + | + | - | + | + | + | + | + | + | NA | + | CD | + | + | Good |
| McTarggart23 (DEFUSE-II) | + | − | + | + | − | − | + | + | + | NA | + | CD | + | + | Fair |
| Saver5 (SWIFT-PRIME) | + | + | + | + | NA | + | + | + | + | + | + | CD | + | − | Good |
| Spiotta13 | + | − | NA | + | − | − | + | + | + | NA | + | CD | + | − | Fair |
| Kim24 | + | + | + | + | NA | − | + | + | + | NA | + | CD | + | − | Fair |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download