Abstract
Objective
To investigate the immediate effect of a single session of whole body vibration (WBV) on lower extremity spasticity in children with cerebral palsy (CP).
Methods
Seventeen children with spastic CP were included. A single session of WBV was administered: 10-minute WBV, 1-minute rest, and 10-minute WBV. The effects of WBV were clinically assessed with the Modified Ashworth Scale (MAS) and Modified Tardieu Scale (MTS) before and immediately, 30 minutes, 1 hour, 2 hours, 3 hours, and 4 hours after WBV.
Results
Spasticity of the ankle plantarflexor, as assessed by MAS and MTS scores, was reduced after WBV. Post-hoc analysis demonstrated that, compared to baseline, the MAS significantly improved for a period of 1 hour after WBV, and the R1 and R2–R1 of the MTS significantly improved for a period of 2 hours after WBV.
Cerebral palsy (CP), which is a common childhood disability and a well-recognized neurodevelopmental condition, begins in early childhood and persists throughout life [1]. Because spastic CP is the most common type of CP in both preterm and term infants, reducing spasticity is an important goal in the management of these children [2]. Anti-spastic medication, botulinum toxin injection, nerve block, and surgery have been used to reduce spasticity in children with CP [3].
Whole body vibration (WBV) has been introduced recently as a new rehabilitation modality. WBV is typically applied while standing on an oscillating platform that displaces the individual and alters the gravitational forces of the body [4]. WBV has been reported to improve muscle strength [5], bone mineral density [6], balance control [7], and lower extremity function [8]. It is also known that WBV reduces spasticity in the lower extremity of people with central nervous system disorders [9].
Some previous studies reported the clinical effects of WBV on spasticity in children with CP. For instance, 3 months of WBV with physiotherapy reduced spasticity and improved motor function more effectively than physiotherapy alone [10]. Also, 8 weeks of WBV (20 minutes at 20 Hz) improved ambulatory function and decreased spasticity in children with CP [8]. Even a single session of WBV can reduce ankle plantarflexor spasticity in patients with chronic stroke [11]. However, it is not known whether a single session of WBV might relieve spasticity in CP, and how long this anti-spastic effect might be maintained. Therefore, this study examined the immediate and longer-lasting effects of a single session of WBV on spasticity of ankle plantarflexors in children with CP.
This was a prospective intervention study. Ethical approval was granted by the Institutional Review Board and Ethics Committee. Because all children in this study were younger than 18 years, informed consent was obtained from the parents of children who agreed to participate in this study.
This study was conducted in a rehabilitation hospital affiliated with the medical college between December 2015 and April 2016. Subjects were selected from those children with spastic CP who were admitted to the hospital for intensive therapy, and who met the following inclusion criteria: (1) over the age of 3 years old, (2) Modified Ashworth Scale (MAS) score at ankle plantarflexors ≥1+ with knee extension, and (3) able to maintain a standing position with or without holding.
Exclusion criteria were as follows: (1) chemodenervation therapy within 6 months, (2) previous selective rhizotomy or orthopedic surgery, (3) cognitive impairment and inability to comply with the required procedures, and (4) any musculoskeletal problems (arthritis, congenital limb deficiency, muscle disease, etc.).
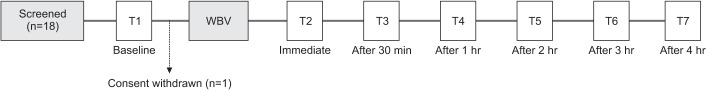
Eighteen children met the criteria, and one eligible participant declined to participate in the study. Thus, 17 children participated in this study (Fig. 1). One child was on anti-spastic medication (baclofen 30 mg/day).
WBV was conducted using a Galileo Med S (Novotech Medical, Pforzheim, Germany), which provides side-to-side, alternating vertical, sinusoidal vibration. The treatment session consisted of the following schedule: 10-minute WBV, 1-minute rest, and 10-minute WBV. The vibration protocol was 20 Hz with 2 mm vertical displacement [8]. Children maintained a semi-squat position on the platform with 30 degrees of knee flexion during the vibration to focus energy on the lower extremities during WBV [12]. Children also grasped the ladder with their hand to maintain a semi-squat position. Four hours of bedrest were done after WBV to exclude other confounding factors.
The muscle tone of the ankle plantarflexor was assessed by the MAS and the Modified Tardieu Scale (MTS) in both knee flexion and extension. The same investigator performed all measurements, including goniometric examinations. Assessments were conducted before, and immediately, 30 minutes, 1 hour, 2 hours, 3 hours, and 4 hours after the intervention. The time points of assessment are shown in Fig. 1. In the case of bilateral CP, muscle tone of the more affected limb was assessed.
The MAS is a 6-point rating system from 0 to 4. For statistical convenience, MAS grade 1+ was converted to grade 2, and MAS grades 2–4 were converted to grades 3–5 [13].
SPSS ver. 20 (IBM SPSS, Armonk, NY, USA) was used for statistical analyses. A linear mixed model was used to analyze data from repeated measurements. An unstructured model was selected as the covariance structure. Scores after WBV were compared to those before WBV to determine whether there was an improvement in spasticity. Statistical significance was set at p<0.05.
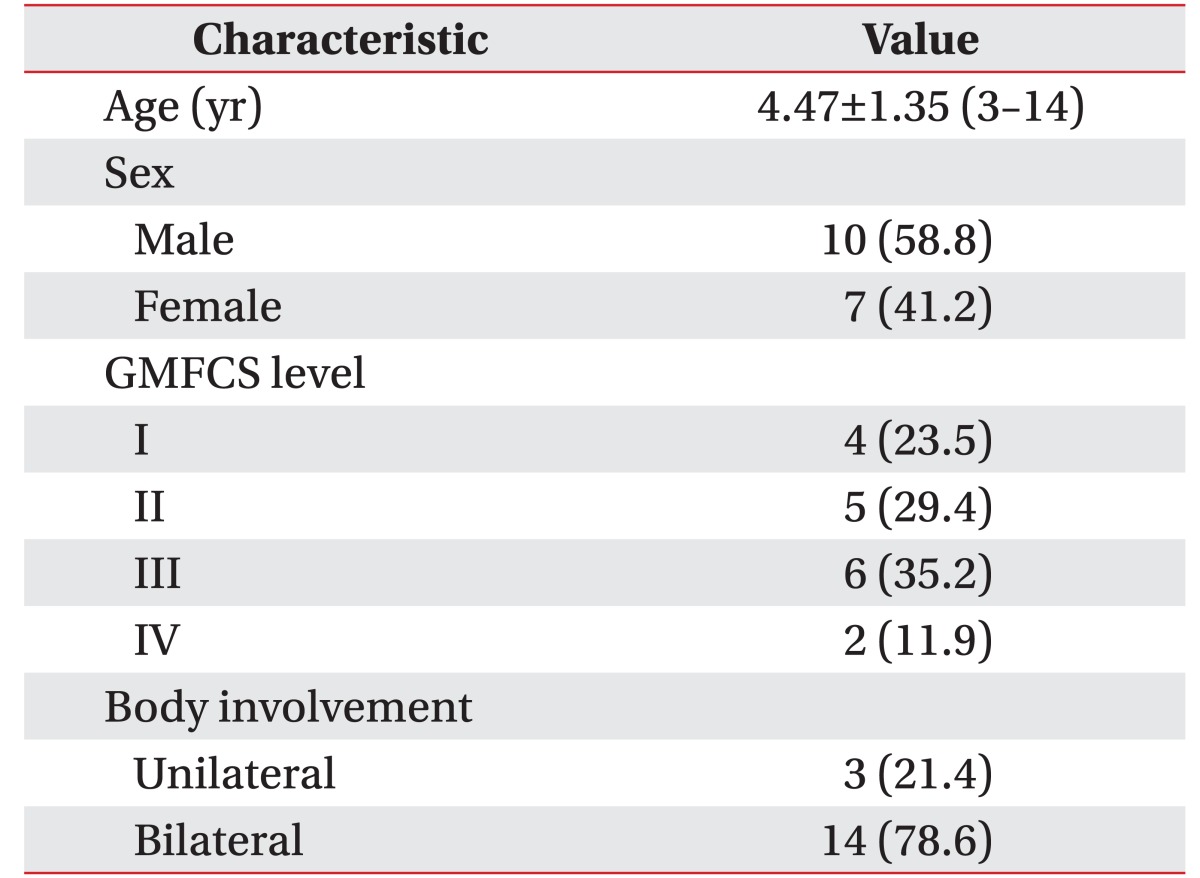
A total of 18 children were recruited, and one child was excluded due to consent withdrawal. Table 1 shows the baseline patient characteristics.
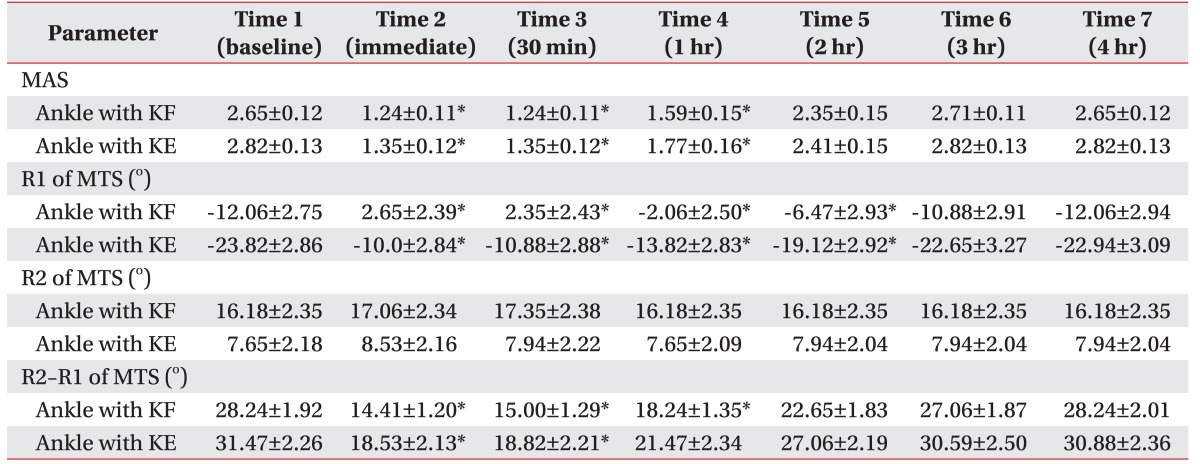
Spasticity of the ankle plantarflexor, as measured by the MAS and MTS, significantly improved after a single session of WBV. Post-hoc analysis demonstrated a significant reduction in MAS score after WBV compared with the baseline score (Fig. 2). The mean difference in MAS score between baseline and immediately after WBV was 1.41 with knee flexion and 1.47 with knee extension (Table 2). The anti-spastic effect, as measured by MAS, lasted for 1 hour after WBV.
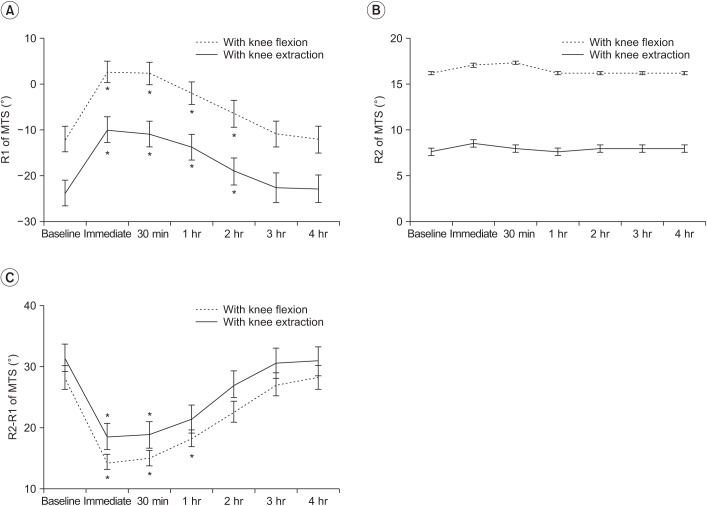
For the MTS, the R1 angle was significantly improved immediately and for up to 2 hours after WBV compared with the measurement before WBV (Fig. 3). The mean difference in R1 angles between baseline and immediately after the intervention was 14.71 with knee flexion and 13.82 with knee extension. The anti-spastic effect measured by the R1 angle lasted for 1–2 hours. There was no significant change in the R2 angle after intervention.
The difference between the two angles (R2–R1) with knee flexion significantly improved compared to the baseline for a period of 1 hour after WBV. The difference between angles with knee extension improved for a period of 30 minutes after WBV. The mean difference in R2– R1 angle between baseline and immediately after WBV was 13.82 with knee flexion and 13.21 with knee extension.
WBV is a noninvasive therapeutic method with no known serious harmful side effects. However, WBV as a therapeutic modality for children with CP is a relatively unexplored area of research. To our knowledge, this is the first study to evaluate the anti-spastic effect of single-session WBV and its duration on ankle plantarflexors in children with CP. The MAS and MTS were used to measure changes in spasticity. We found that spasticity was reduced immediately after a single session of WBV, and this improvement lasted for 1–2 hours.
Children with CP received WBV during supported standing on a vibrating platform. The vibrations are believed to initiate muscle contractions by stimulating muscle spindles and alpha motor neurons [4]. The direct vibratory stimulation of muscles may reduce spasticity by influencing the Ia afferent fibers of the muscle spindle, which inhibit the monosynaptic reflex [1216]. Therefore, the Hmax/Mmax ratio of the H reflex is significantly decreased, together with clinical spasticity associated with the hypersensitive muscle stretch reflex [11]. Although the mechanism by which WBV reduces spasticity is not yet known, the previous finding of a reduction in Hmax/Mmax ratio after WBV implies that the underlying mechanism may be similar to direct muscle vibration [11].
In this study, muscle tone was assessed using the MTS, which is a more reliable and valid method than the MAS. The R1 angle was defined as the point in the joint range at which a velocity-dependent ‘catch’ or clonus was felt during a quick stretch of the muscle, whereas the R2 angle was defined as the passive range of motion. When a muscle is stretched to the R2 position, the viscoelasticity of the soft tissues and the component of contracture become involved. Thus, the difference between the two angles (R2–R1) represents the dynamic component of spasticity [17]. In this study, the R1 angle and R2–R1 improved after WBV, but the R2 angle did not show a significant change. This suggests that WBV is effective for reducing dynamic spasticity, but not for relieving the static components of hypertonicity. This possibility is supported, not only by the electrophysiological changes and reduced Hmax/Mmax ratio observed in previous studies, but also by clinical assessments in the present study.
On the other hand, involuntary tonic reflex contractions of stimulated muscles are induced when the amplitude is as high as 0.2–0.5 mm for direct vibratory stimulation of muscles. Therefore, a low amplitude vibration for direct muscle stimulation has been applied for spasticity management. In addition, high frequency vibration (100 Hz) consistently produces anti-spastic effects [18]. However, relatively high amplitudes and low frequencies (2 mm and 20 Hz in the present study) have been used for WBV to reduce spasticity [8]. Therefore, the mechanisms underlying spasticity reduction may be different between direct muscle vibration and WBV. Future studies are needed to elucidate the mechanism and optimal parameters of WBV to reduce spasticity.
By reducing spasticity in children with CP, a ‘window of opportunity’ can be provided to promote motor development [19]. For example, botulinum toxin for reducing spasticity in children with CP can enhance both motor ability and functional skills, although its anti-spastic effects are temporary [20]. Many physiotherapists spend much time reducing abnormal tone by stretching, handling, and positioning at each therapy session so that the children are ‘prepared’ to move [21]. Our results suggest that WBV before physiotherapy sessions can help therapists manage patients more effectively by reducing spasticity.
Standing for 20 minutes may stretch the calf and Achilles tendons which might influence the MAS and MTS in this study. This could be a limitation and should be considered in further studies.
In conclusion, this study found that a single 20-minute session of 20 Hz WBV reduced spasticity of ankle plantarflexors for 1–2 hours in children with CP. The results suggest that WBV performed just before physiotherapy or occupational therapy sessions may help prepare children with spasticity to learn motor skills. However, future studies are needed to verify the efficacy of WBV for preparing children with spastic CP for physiotherapy and occupational therapy sessions.
ACKNOWLEDGMENTS
This study is supported by a research grant of Research Institute of Rehabilitation Medicine, Yonsei University College of Medicine for 2015.
References
2. Himpens E, Van den Broeck C, Oostra A, Calders P, Vanhaesebrouck P. Prevalence, type, distribution, and severity of cerebral palsy in relation to gestational age: a meta-analytic review. Dev Med Child Neurol. 2008; 50:334–340. PMID: 18355333.

3. Krigger KW. Cerebral palsy: an overview. Am Fam Physician. 2006; 73:91–100. PMID: 16417071.
4. Cochrane DJ. Vibration exercise: the potential benefits. Int J Sports Med. 2011; 32:75–99. PMID: 21165804.

5. Delecluse C, Roelants M, Verschueren S. Strength increase after whole-body vibration compared with resistance training. Med Sci Sports Exerc. 2003; 35:1033–1041. PMID: 12783053.

6. Slatkovska L, Alibhai SM, Beyene J, Cheung AM. Effect of whole-body vibration on BMD: a systematic review and meta-analysis. Osteoporos Int. 2010; 21:1969–1980. PMID: 20407890.

7. Kawanabe K, Kawashima A, Sashimoto I, Takeda T, Sato Y, Iwamoto J. Effect of whole-body vibration exercise and muscle strengthening, balance, and walking exercises on walking ability in the elderly. Keio J Med. 2007; 56:28–33. PMID: 17392595.

8. Cheng HY, Ju YY, Chen CL, Chuang LL, Cheng CH. Effects of whole body vibration on spasticity and lower extremity function in children with cerebral palsy. Hum Mov Sci. 2015; 39:65–72. PMID: 25461434.

9. Huang M, Liao LR, Pang MY. Effects of whole body vibration on muscle spasticity for people with central nervous system disorders: a systematic review. Clin Rehabil. 2017; 31:23–33. PMID: 26658333.

10. Katusic A, Alimovic S, Mejaski-Bosnjak V. The effect of vibration therapy on spasticity and motor function in children with cerebral palsy: a randomized controlled trial. NeuroRehabilitation. 2013; 32:1–8. PMID: 23422453.

11. Chan KS, Liu CW, Chen TW, Weng MC, Huang MH, Chen CH. Effects of a single session of whole body vibration on ankle plantarflexion spasticity and gait performance in patients with chronic stroke: a randomized controlled trial. Clin Rehabil. 2012; 26:1087–1095. PMID: 23035004.

12. Ness LL, Field-Fote EC. Effect of whole-body vibration on quadriceps spasticity in individuals with spastic hypertonia due to spinal cord injury. Restor Neurol Neurosci. 2009; 27:621–631. PMID: 20042786.

13. Bohannon RW, Smith MB. Interrater reliability of a modified Ashworth scale of muscle spasticity. Phys Ther. 1987; 67:206–207. PMID: 3809245.

14. Boyd R, Barwood S, Baillieu C, Graham H. Validity of a clinical measure of spasticity in children with cerebral palsy in a double-blind randomized controlled clinical trial. Dev Med Child Neurol. 1998; 40(Suppl 78):7.
15. Mackey AH, Walt SE, Lobb G, Stott NS. Intraobserver reliability of the modified Tardieu scale in the upper limb of children with hemiplegia. Dev Med Child Neurol. 2004; 46:267–272. PMID: 15077704.

16. Seo HG, Oh BM, Leigh JH, Chun C, Park C, Kim CH. Effect of Focal Muscle Vibration on Calf Muscle Spasticity: A Proof-of-Concept Study. PM R. 2016; 8:1083–1089. PMID: 26994885.

17. Yam WK, Leung MS. Interrater reliability of Modified Ashworth Scale and Modified Tardieu Scale in children with spastic cerebral palsy. J Child Neurol. 2006; 21:1031–1035. PMID: 17156693.

18. Kavounoudias A, Roll R, Roll JP. Specific whole-body shifts induced by frequency-modulated vibrations of human plantar soles. Neurosci Lett. 1999; 266:181–184. PMID: 10465703.

19. Francis HP, Wade DT, Turner-Stokes L, Kingswell RS, Dott CS, Coxon EA. Does reducing spasticity translate into functional benefit? An exploratory meta-analysis. J Neurol Neurosurg Psychiatry. 2004; 75:1547–1551. PMID: 15489384.

20. Friedman BC, Goldman RD. Use of botulinum toxin A in management of children with cerebral palsy. Can Fam Physician. 2011; 57:1006–1073. PMID: 21918142.
21. Levitt S. Treatment of cerebral palsy and motor delay. 5th ed. Hoboken: John Wiley & Sons;2010.
Fig. 2
Changes in Modified Ashworth Scale (MAS) after whole body vibration therapy. *p<0.05 vs. baseline (post-hoc test).
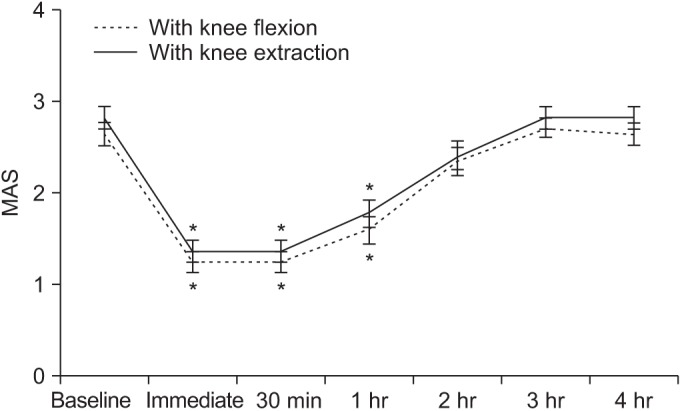
Fig. 3
Changes in Modified Tardieu Scale (MTS) scores after whole body vibration therapy. (A) R1 angle, (B) R2 angle, and (C) R2–R1. *p<0.05 vs. baseline (post-hoc test).




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download