1. Scheer SJ, Radack KL, O'Brien DR Jr. Randomized controlled trials in industrial low back pain relating to return to work. Part 2. Discogenic low back pain. Arch Phys Med Rehabil. 1996; 77:1189–1197. PMID:
8931535.

2. Ferreira ML, Ferreira PH, Latimer J, Herbert RD, Hodges PW, Jennings MD, et al. Comparison of general exercise, motor control exercise and spinal manipulative therapy for chronic low back pain: a randomized trial. Pain. 2007; 131:31–37. PMID:
17250965.

3. Koltyn KF, Garvin AW, Gardiner RL, Nelson TF. Perception of pain following aerobic exercise. Med Sci Sports Exerc. 1996; 28:1418–1421. PMID:
8933493.

4. Sculco AD, Paup DC, Fernhall B, Sculco MJ. Effects of aerobic exercise on low back pain patients in treatment. Spine J. 2001; 1:95–101. PMID:
14588388.

5. Meyer P, Kayser B, Mach F. Stair use for cardiovascular disease prevention. Eur J Cardiovasc Prev Rehabil. 2009; 16(Suppl 2):S17–S18. PMID:
19675430.

6. Borschmann K, Pang MY, Bernhardt J, Iuliano-Burns S. Stepping towards prevention of bone loss after stroke: a systematic review of the skeletal effects of physical activity after stroke. Int J Stroke. 2012; 7:330–335. PMID:
21967614.

7. Granacher U, Muehlbauer T, Gruber M. A qualitative review of balance and strength performance in healthy older adults: impact for testing and training. J Aging Res. 2012; 2012:708905. PMID:
22315687.

8. Marin R, Chang A, Cyhan T, Dinauer P. Rehabilitation implications of stepper exercise technique on exertion and hip extensor muscle activation: a small exploratory study. J Rehabil Res Dev. 2008; 45:125–134. PMID:
18566931.
9. Souza DR, Gross MT. Comparison of vastus medialis obliquus: vastus lateralis muscle integrated electromyographic ratios between healthy subjects and patients with patellofemoral pain. Phys Ther. 1991; 71:310–316. PMID:
2008454.
10. Bergmark A. Stability of the lumbar spine: a study in mechanical engineering. Acta Orthop Scand Suppl. 1989; 230:1–54. PMID:
2658468.
11. Norris CM. Abdominal muscle training in sport. Br J Sports Med. 1993; 27:19–27. PMID:
8457806.

12. Hodges PW. Is there a role for transversus abdominis in lumbo-pelvic stability? Man Ther. 1999; 4:74–86. PMID:
10509061.

13. Souza GM, Baker LL, Powers CM. Electromyographic activity of selected trunk muscles during dynamic spine stabilization exercises. Arch Phys Med Rehabil. 2001; 82:1551–1557. PMID:
11689975.

14. Richardson C, Hodges PW, Hides J. Therapeutic exercise for lumbopelvic stabilization: a motor control approach for the treatment and prevention of low back pain. 2nd ed. Edinburgh: Churchill Livingstone;2004.
15. Henry SM, Westervelt KC. The use of real-time ultrasound feedback in teaching abdominal hollowing exercises to healthy subjects. J Orthop Sports Phys Ther. 2005; 35:338–345. PMID:
16001905.

16. Henry SM, Teyhen DS. Ultrasound imaging as a feedback tool in the rehabilitation of trunk muscle dysfunction for people with low back pain. J Orthop Sports Phys Ther. 2007; 37:627–634. PMID:
17970410.

17. Hodges PW, Richardson CA. Altered trunk muscle recruitment in people with low back pain with upper limb movement at different speeds. Arch Phys Med Rehabil. 1999; 80:1005–1012. PMID:
10489000.

18. Urquhart DM, Hodges PW, Allen TJ, Story IH. Abdominal muscle recruitment during a range of voluntary exercises. Man Ther. 2005; 10:144–153. PMID:
15922235.

19. Costa LO, Costa Lda C, Cancado RL, Oliveira Wde M, Ferreira PH. Short report: intra-tester reliability of two clinical tests of transversus abdominis muscle recruitment. Physiother Res Int. 2006; 11:48–50. PMID:
16594315.
20. Hodges PW, Pengel LH, Herbert RD, Gandevia SC. Measurement of muscle contraction with ultrasound imaging. Muscle Nerve. 2003; 27:682–692. PMID:
12766979.

21. Claus AP, Hides JA, Moseley GL, Hodges PW. Different ways to balance the spine: subtle changes in sagittal spinal curves affect regional muscle activity. Spine (Phila Pa 1976). 2009; 34:E208–E214. PMID:
19282726.
22. Tsao H, Druitt TR, Schollum TM, Hodges PW. Motor training of the lumbar paraspinal muscles induces immediate changes in motor coordination in patients with recurrent low back pain. J Pain. 2010; 11:1120–1128. PMID:
20434958.

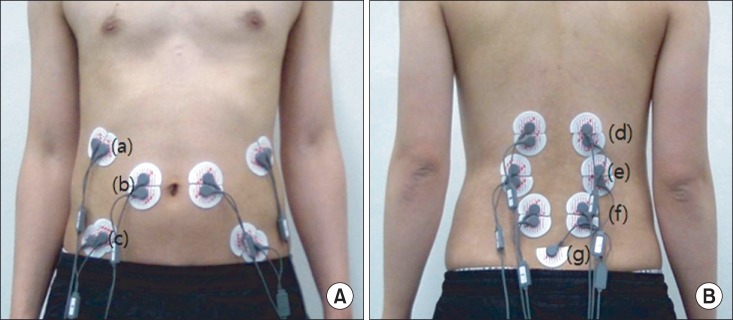
23. de Seze MP, Cazalets JR. Anatomical optimization of skin electrode placement to record electromyographic activity of erector spinae muscles. Surg Radiol Anat. 2008; 30:137–143. PMID:
18183349.

24. Ng JK, Kippers V, Richardson CA. Muscle fibre orientation of abdominal muscles and suggested surface EMG electrode positions. Electromyogr Clin Neurophysiol. 1998; 38:51–58. PMID:
9532434.
25. Bressel E, Dolny DG, Gibbons M. Trunk muscle activity during exercises performed on land and in water. Med Sci Sports Exerc. 2011; 43:1927–1932. PMID:
21448084.

26. Roys MS. Serious stair injuries can be prevented by improved stair design. Appl Ergon. 2001; 32:135–139. PMID:
11277505.

27. Stevens VK, Bouche KG, Mahieu NN, Coorevits PL, Vanderstraeten GG, Danneels LA. Trunk muscle activity in healthy subjects during bridging stabilization exercises. BMC Musculoskelet Disord. 2006; 7:75. PMID:
16987410.

28. Pfingsten M, Hildebrandt J, Leibing E, Franz C, Saur P. Effectiveness of a multimodal treatment program for chronic low-back pain. Pain. 1997; 73:77–85. PMID:
9414059.

29. Hagins M, Adler K, Cash M, Daugherty J, Mitrani G. Effects of practice on the ability to perform lumbar stabilization exercises. J Orthop Sports Phys Ther. 1999; 29:546–555. PMID:
10518297.

30. Nachemson AL. Instability of the lumbar spine: pathology, treatment, and clinical evaluation. Neurosurg Clin N Am. 1991; 2:785–790. PMID:
1821757.
31. Martin PR, Rose MJ, Nichols PJ, Russell PL, Hughes IG. Physiotherapy exercises for low back pain: process and clinical outcome. Int Rehabil Med. 1986; 8:34–38. PMID:
2942511.

32. Hides JA, Jull GA, Richardson CA. Long-term effects of specific stabilizing exercises for first-episode low back pain. Spine (Phila Pa 1976). 2001; 26:E243–E248. PMID:
11389408.

33. Hides JA, Richardson CA, Jull GA. Multifidus muscle recovery is not automatic after resolution of acute, first-episode low back pain. Spine (Phila Pa 1976). 1996; 21:2763–2769. PMID:
8979323.

34. Hodges PW, Richardson CA. Contraction of the abdominal muscles associated with movement of the lower limb. Phys Ther. 1997; 77:132–142. PMID:
9037214.

35. Hodges PW, Richardson CA. Transversus abdominis and the superficial abdominal muscles are controlled independently in a postural task. Neurosci Lett. 1999; 265:91–94. PMID:
10327176.

36. Hodges PW, Richardson CA. Inefficient muscular stabilization of the lumbar spine associated with low back pain: a motor control evaluation of transversus abdominis. Spine (Phila Pa 1976). 1996; 21:2640–2650. PMID:
8961451.
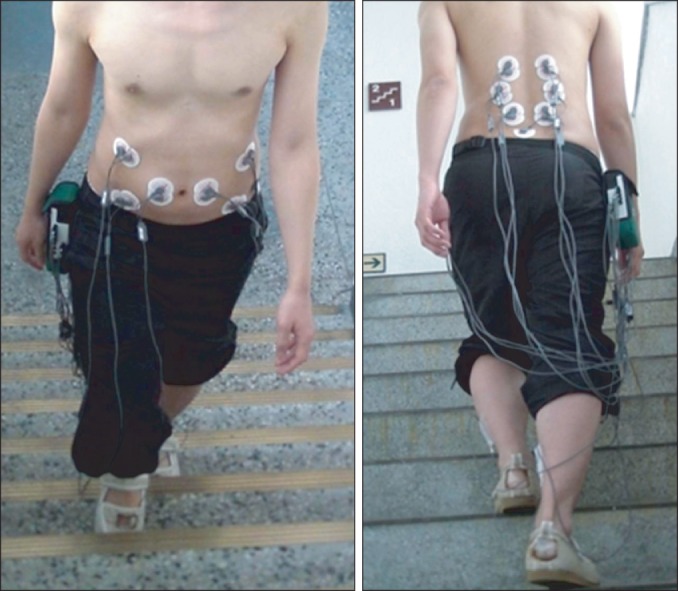
37. Lee AH, Ahn SH, Cho YW, Jang SH, Jo HK, Choi HS, et al. Abdominal hollowing during gait can selectively activate local trunk stabilizing muscles. In : Proceedings of the 40th Annual Meeting of the Korean Academy of Rehabilitation Medicine; 2012 Oct 12-13; Seoul, Korea.
38. Richardson C, Jull G, Toppenberg R, Comerford M. Techniques for active lumbar stabilization for spinal protection: a pilot study. Aust J Physiother. 1992; 38:105–112.
39. Vezina MJ, Hubley-Kozey CL. Muscle activation in therapeutic exercises to improve trunk stability. Arch Phys Med Rehabil. 2000; 81:1370–1379. PMID:
11030503.

40. Duval K, Lam T, Sanderson D. The mechanical relationship between the rearfoot, pelvis and low-back. Gait Posture. 2010; 32:637–640. PMID:
20889344.











 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download