Abstract
The intrathecal chemotherapy with methotrexate and cytarabine arabinoside is used for the treatment and prophylaxis of the primary central nervous system lymphoma. The therapy may induce neurotoxicity including the cauda equina syndrome. We report a case of a 58-year-old man with the diffuse large B-cell lymphoma, who developed the cauda equina syndrome after the administration of intrathecal methotrexate and cytarabine arabinoside, as diagnosed by the electrodiagnostic, urodynamic, and radiologic approaches.
The intrathecal (IT) chemotherapy is an important component of the treatment and prophylaxis of the primary central nervous system (CNS) lymphoma. There have been reports of neurotoxicity attributed to the IT chemotherapy, including arachnoiditis, seizures, encephalopathy, and the spinal cord lesion. The incidence rates range from 4.2% to 61%, showing a large discrepancy between the various reports [1-4].
The spinal cord lesions manifesting as tetraplegia, paraplegia, and the cauda equina syndrome are some of the most frequently described neurotoxicity cases after the IT chemotherapy. Of the 28 reported cases with the spinal cord lesions after the IT chemotherapy, paraplegia has occurred in 23 cases (82%), tetraplegia in three cases (11%), and the cauda equina syndrome in two cases (7%). Most patients have shown poor recovery and are left with residual neurological deficits [2].
Three cases of paraplegia after the IT chemotherapy have been reported in Korea [1,4,5]. Two cases had received IT methotrexate (MTX) only. One case was diagnosed as thoracic myelopathy by the magnetic resonance imaging (MRI). The other case was suspected as the thoracic spinal cord lesion clinically, but not confirmed by a radiologic evaluation. Another case had received IT MTX and cytarabine arabinoside (Ara-C), and was diagnosed as the thoracic myelopathy by MRI. There is no report of the cauda equina syndrome after IT MTX and Ara-C, a standard method for the primary CNS lymphoma, as diagnosed by the electrodiagnostic, urodynamic, and radiologic approaches. This is the first case report of the cauda equina syndrome after the IT chemotherapy in Korea.
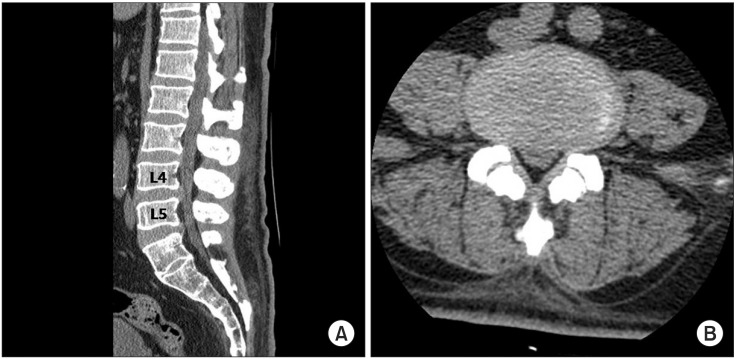
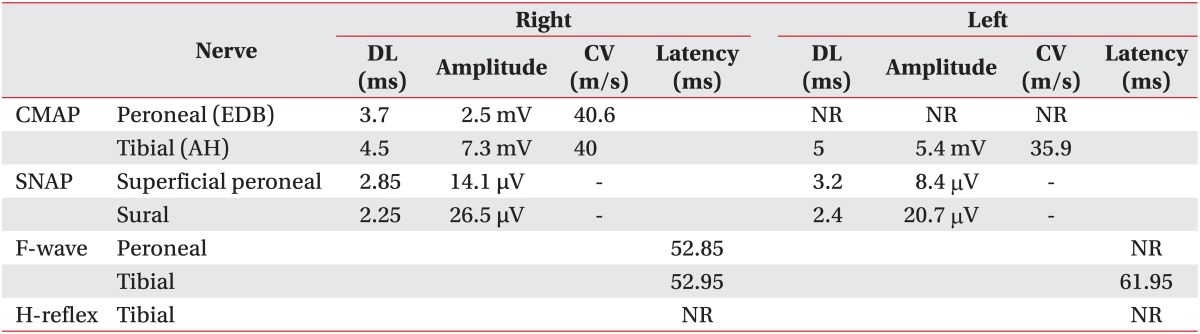
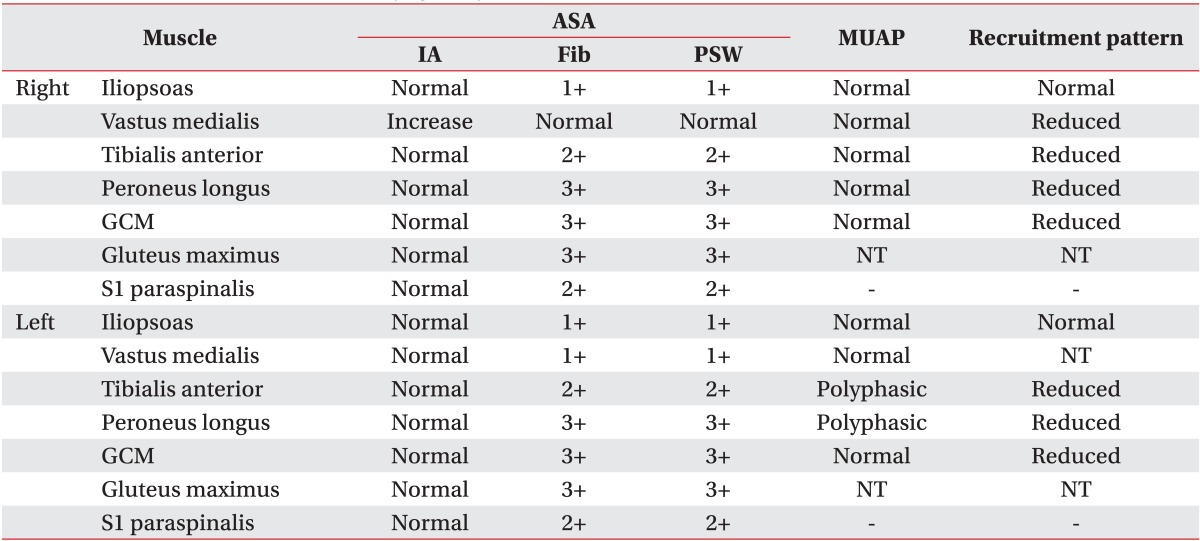
A 58-year-old male patient who had no past medical history presented with fatigue, subjective feeling of the left lower-extremity weakness for 1 month, and the left facial palsy for 3 days. Brain MRI was preformed after admission to the Department of Neurosurgery at the Konkuk University Medical Center. A brain tumor was revealed at corpus callosum and septum pellucidum. On the physical examination was observed was the central type left facial palsy without other neurological abnormalities. He underwent craniotomy and a tumor removal operation, and didn't show any changes in the neurological examination following the surgery. After the diagnosis of the diagnosed as diffuse large B-cell lymphoma on the tissue biopsy, the patient he was transferred to the Department of Internal Medicine for chemotherapy. He received one treatment of the systemic chemotherapy, with MTX, thiopeta, and vincristine, 16 days after the surgery. Five weeks after the surgery, the patient he underwent the IT chemotherapy 9 times, with MTX 15 mg, Ara-C 40 mg, and hydrocortisone 50 mg over a 4-week period, followed by the intracranial radiation therapy with 30 Gy doses. After 4 weeks from the initiation of the IT chemotherapy, he developed a gradual weakness of the bilateral lower extremities, and then dysuria 4 days later. The cerebrospinal fluid (CSF) study was normal with a pressure of 13.0 cm H2O, the protein level 78.1 mg/dL, and the glucose level more than 70% of blood sugar. There were no findings of white blood cells nor malignant cells in the CSF. The patient was transferred to the Department of Rehabilitation Medicine for the evaluation and treatment of the bilateral lower extremities weakness. He demonstrated a Medical Research Council (MRC) grade III weakness of the right hip flexor and knee extensor, MRC grade II weakness of the right hip extensor, knee flexor, ankle dorsiflexor and plantarflexor, and big toe extensor and flexor, and general MRC grade II weakness of the left lower extremity. There was hypesthesia to the pain, temperature, vibration, and proprioception at the dermatome below L2 level bilaterally. There were no responses of the bilateral knee and the ankle deep tendon reflexes, and the pathologic reflexes including Babinski sign and ankle clonus were absent. He received a Foley catheter insertion, as he did not have any urination sense and was not able to void at all. He also could not sense anal mucosa touch or deep anal pressure, and showed weakness of the anal sphincter muscle. There were no responses of the bilateral bulbocavernosus reflexes (BCR). However, the bilateral upper extremities and trunk had normal neurological findings. Electrodiagnostic examination was performed 5 weeks after the onset of the bilateral lower extremities weakness. It revealed a decreased compound muscle action potential (CMAP) amplitude of the right common peroneal nerve, measured from the extensor digitorum brevis (EDB) and the left tibial nerve from the abductor hallucis (AH) muscles. No CMAP response was detected from the left EDB muscle. There were normal sensory findings of the bilateral superficial peroneal and sural nerves. Late response studies showed the delayed F-wave latency of the left tibial nerve, with no F-wave of the left common peroneal nerve. The H-reflex was absent in the bilateral tibial nerves (Table 1). The motor-evoked potentials recorded from the bilateral AH muscles following the stimulation of the bilateral primary motor cortices showed delayed latencies, 46.00 ms in the right and 45.85 ms in the left side. However, the recordings of the bilateral upper extremities were normal. The somatosensory evoked potentials which were recorded on the bilateral somatosensory cortices had no responses when the bilateral tibial nerves were stimulated, but showed normal findings when the bilateral upper extremities were stimulated. The needle electromyogram revealed abnormal spontaneous activities in the bilateral iliopsoas, vastus medialis, tibialis anterior, peroneus longus, gastrocnemius, gluteus maximus, and the 1st sacral paraspinalis muscles, along with the polyphasic motor unit potentials in the left tibialis anterior and the peroneus longus muscles (Table 2). The electrodiagnostic study findings were consistent with the bilateral lumbosacral polyradiculopathy from the 2nd lumbar to the 1st sacral spinal nerve. The urodynamic study revealed the areflexic neurogenic bladder which may present the 2nd to the 4th sacral nerves dysfunction. He also presented a decreased sense of the anus and a weakness of the anal sphincter muscle. He was diagnosed with the cauda equina syndrome based on his clinical presentation and the results of the electrodiagnostic and urodynamic studies. The lumbar computed tomography (CT) did not show other lesions except the central spinal stenosis at the L4-5 intervertebral disc level to a mild degree, which was not significant enough to induce a nerve injury (Fig. 1). He received a rehabilitation program regimen including the wheelchair transfer training, strengthening of the upper extremities, activities of daily living training, and management of neurogenic bladder and bowel. There were no neurological improvements upon discharge and at 14 months follow-up visit after the onset.
The IT chemotherapy is commonly used for the treatment of the hematologic malignancies in the CNS and prophylaxis of the CNS metastasis, especially in the patients with aggressive lymphomas and acute leukemia. The two most commonly used drugs for IT chemotherapy are MTX and Ara-C.
There are several hypotheses on the mechanism of the neurotoxicity of IT MTX. The neurotoxicity can occur when the malignant cells are located at the sites of the CSF transition area, where MTX cannot be absorbed into systemic circulation [4]. The second hypothesis is that a prolonged exposure to a high-dose IT MTX can change the neural tissues [4]. Bleyer et al. [6] reported that a patient who showed neurotoxicity showed large amounts of MTX in the CSF, which was 13.8 times greater than that of an a asymptomatic patient. The third hypothesis is that methyl hydroxybenzoate and propyl hydroxybenzoate, used as the preservatives, can induce the conduction block of the spinal nerve roots and diffuse the degenerative changes of the nerves, respectively [4,5]. If normal saline or distilled water is used as diluents, the chemical irritation to the nerves is increased as a change in acidity or the ionic contents is induced [4,7]. Another theory is that the lack of folic acid in the CSF can induce the lack of protein synthesis in the nerve cells and interfere with the metabolism of the nerve cells. It is reported that the patients who developed myelopathy after the IT chemotherapy have shown showed improvement after the administration of folic acid [4,5,8]. Finally, paraplegia can be induced by the hypersensitive reaction to MTX. There has been one fatal case of allergic urticaria and paralysis following IT MTX [4].
Systemic Ara-C is eliminated rapidly by hepatic cytidine deaminase, with the plasma half-life of less than 20 minutes. However, cytidine deaminase is not present in the CNS, and the half-life of Ara-C in the CSF is typically much longer, reaching to 2-6 hours. The longtime exposure of Ara-C in the CSF can induce neurotoxicity [2,5,9]. Additionally, Ara-C triphosphate, a metabolite of Ara-C, can interfere with the DNA synthesis and cell replication in the CNS. It also forms arabinosyl cytidine choline, which interferes with the synthesis of glycoprotein and glycolipid, the components of the cell membrane [8].
Literature suggests that the concomitant use of IT MTX and IT Ara-C may enhance the neurotoxicity of MTX [5], but there are few studies on the interaction of the two drugs. IT corticosteroids are used to increase cytotoxicity and to reduce the risks of chemical arachnoiditis which may arise in response to other IT drugs. However, no studies have proven the preventive effect of the concomitant use of hydrocortisone to date [2,5,10].
Three cases of paraplegia after the IT chemotherapy have been reported in Korea [1,4,5]. The first case [1], presenting with a neurological abnormality on the trunk and the bilateral lower extremities, was diagnosed as thoracic myelopathy with severe atrophy at multiple levels, which were ranged from T8 to T12 by MRI. There was no improvement at 15 months after the symptom onset. The second case [4] demonstrated similar symptoms as the first case and was diagnosed as multiple polyradiculoneuropathy of the bilateral lower extremities. Because there were abnormal spontaneous activities at the bilateral thoracic 5th to 9th paraspinalis muscles on the needle electromyogram study, the patient was suspected to have thoracic spinal lesions, but was not confirmed by a radiologic evaluation. There was no improvement up to the 3 months follow-up after the onset. The last case [5], where the patient developed the neurological abnormality of the bilateral lower extremities, was diagnosed as thoracic myelopathy with severe thoracic cord atrophy by MRI. This case also had no signs of neurological improvement up to the 18 months follow-up. Our case demonstrated flaccid paralysis, hypesthesia, and a loss of the deep tendon reflexes of the bilateral lower extremities with the neurogenic bladder and bowel symptoms. He was diagnosed as bilateral lumbosacral polyradiculopathy from the 2nd lumbar to the 1st sacral spinal nerves by the electrodiagnostic evaluation. The 2nd to 4th sacral nerves could not be evaluated as the patient did not tolerate the examination, and there was fecal incontinence. The urodynamic study showed areflexic bladder. Based on the above study results and his clinical presentation, including the decreased sense of anus, a weakness of the anal sphincter muscle, and no response of the bilateral BCR, he was diagnosed with the cauda equina syndrome. There were no symptoms suggesting arachnoiditis, seizures, or encephalopathy in the patient. As his brain lesions did not involve the corticospinal tract, they were ruled out as the cause of the neurological abnormalities. We could not have the lumbar MRI after the developed symptoms, because his blood pressure dropped when he received sedatives. However, there were no abnormal lesions in the lumbar spines CT which could induce the cauda equina syndrome. There were no inflammatory findings nor malignant cells in the CSF study. There is a small possibility that the needle used for the CSF tapping could have injured the cauda equina. However, his symptoms were diffuse and gradually manifested, and there was no hematoma seen in the CT. This is the first case report in Korea, where the cauda equina syndrome was occurred by neurotoxicity after the IT chemotherapy with MTX and Ara-C.
Our patient did not show any neurological improvements up to 14 months follow-up after the symptoms onset, same as the two previous cases of paraplegia after the IT chemotherapy reported in Korea. A complete recovery has been exceptional, occurring in only three of the 28 reported cases as reported by Kwong et al. [2]. Variable degrees of motor power recovery may be achieved, but the neurogenic bladder and bowels rarely improve [2]. There is a case of the paraplegia occurred after the IT chemotherapy, where there were neurological improvements after the administration of folic acid [8]. However, there are no randomized controlled trials with a large subject size at this time. There is no standard treatment for the spinal cord lesions occurred after the IT chemotherapy, but if the symptoms of neurotoxicity are doubted, the IT chemotherapy should be stopped immediately and the folic acid administration should be considered.
We have reported the case of the cauda equina syndrome after IT MTX and Ara-C for the treatment of the primary CNS lymphoma, which was diagnosed by the electrodiagnostic, urodynamic and radiologic approaches.
References
1. Lee CH, Oh MK, Cho JH, Lee ES, Shin HS. A case of paraplegia associated with intrathecal methotrexate: a case report. J Korean Acad Rehabil Med. 2006; 30:188–190.
2. Kwong YL, Yeung DY, Chan JC. Intrathecal chemotherapy for hematologic malignancies: drugs and toxicities. Ann Hematol. 2009; 88:193–201. PMID: 19050889.

3. Jabbour E, O'Brien S, Kantarjian H, Garcia-Manero G, Ferrajoli A, Ravandi F, et al. Neurologic complications associated with intrathecal liposomal cytarabine given prophylactically in combination with high-dose methotrexate and cytarabine to patients with acute lymphocytic leukemia. Blood. 2007; 109:3214–3218. PMID: 17209054.

4. Kim JI, Roh JK, Myung H, Kim JM. A case of paraplegia following intrathecal methotrexate instillation. J Korean Neurol Assoc. 1990; 8:145–150.
5. Lee HY, Im SI, Kang MH, Kim KM, Kim SH, Kim HG, et al. Irreversible paraplegia following one time prophylactic intrathecal chemotherapy in an adult patient with acute lymphoblastic leukemia. Yonsei Med J. 2008; 49:151–154. PMID: 18306482.

6. Bleyer WA, Drake JC, Chabner BA. Neurotoxicity and elevated cerebrospinal-fluid methotrexate concentration in meningeal leukemia. N Engl J Med. 1973; 289:770–773. PMID: 4517004.

7. Geiser CF, Bishop Y, Jaffe N, Furman L, Traggis D, Frei E 3rd. Adverse effects of intrathecal methotrexate in children with acute leukemia in remission. Blood. 1975; 45:189–195. PMID: 1091308.
8. Park SY, Park HR, Kim JE, Sung JJ. Intrathecal chemotherapy related myelopathy improved with folate and cyanocobalamin. J Korean Neurol Assoc. 2011; 29:224–226.
9. Slevin ML, Piall EM, Aherne GW, Harvey VJ, Johnston A, Lister TA. Effect of dose and schedule on pharmacokinetics of high-dose cytosine arabinoside in plasma and cerebrospinal fluid. J Clin Oncol. 1983; 1:546–551. PMID: 6583325.

10. Ferreri AJ, Marturano E. Primary CNS lymphoma. Best Pract Res Clin Haematol. 2012; 25:119–130. PMID: 22409828.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download