Abstract
Early diagnosis and treatment of heterotopic ossification (HO) is essential to the prevention of complications. It is difficult to diagnose HO in its initial phase because non-specific clinical manifestations, laboratory findings and imaging findings of immature HO may mimic other diseases such as cellulitis, osteomyelitis, thrombophlebitis, deep vein thrombosis and local infection with abscess. We experienced two cases of HO, which were misdiagnosed as pyomyositis at first by clinical signs and MRI findings indicating the deep infection; the extensive intramuscular ossification appeared later on. We observed an increase of C-reactive protein and creatine kinase followed by the elevation of alkaline phosphatase with abnormal triphasic bone scan. The trajectory of these biomarkers was analyzed to get more insight into the early stages of HO along with the imaging findings. Although our cases cannot be generalized as typical of immature HO, they clearly demonstrate that the change of specific biomarkers with a careful history taking and physical examination should be noted to detect HO as early as possible while avoiding confusion with other mimicking conditions.
Heterotopic ossification (HO) is the formation of mature, lamellar bone in soft tissues. HO has been frequently noted after neurogenic and traumatic insults. Neurogenic insults include brain injury and spinal cord injury (SCI). Traumatic HO occurs occasionally in patients with fractures, dislocation, and total hip arthroplasty. The clinical signs and symptoms of HO may appear as early as 3 weeks, or as late as 12 weeks after neurogenic and/or traumatic insult. Complications of HO include peripheral nerve entrapment, pressure ulcers, and functional impairment if joint ankylosis develops.1 Early diagnosis and treatment of HO is critical to preventing complications of HO. However early diagnosis remains problematic due to non-specific presenting signs and symptoms. Fever, swelling, erythema, and decreased joint motion typically seen in early HO may closely mimic symptoms of cellulitis, osteomyelitis, thrombophlebitis, or deep vein thrombosis. In addition, MRI signal characteristics of immature HO formation are equivalent to those of local infection with abscess.2 Particularly, it is more difficult to diagnose in the early stage of HO around ulcerative lesions or postoperative areas. We experienced two cases of HO, with one neurogenic and other traumatic cause, both of which were initially misdiagnosed as pyomyositis by MRI. The details of early findings of HO are described here to help physicians pay closer attention to early diagnosis and treatment for HO.
A 50-year-old male presented to the emergency room following a motor vehicle accident. He was diagnosed with a C5-C6 fracture- dislocation and immediately underwent a fixation surgery of C5 and C6 vertebral body. After an unremarkable postoperative course, he was transferred to inpatient rehabilitation with an C6/C6 American Spinal Injury Association (ASIA) A tetraplegia. Twenty days after the operation, he began to have intermittent high fever with a temperature of 38.4℃. On postoperative day 35, his right thigh was noted to be edematous. The circumferential difference between the lower extremities measured at 15 cm above the knee was 6.6 cm (right 39.6/left 33.0 cm). His white blood cell count was 11,230 cells/uL, erythrocyte sedimentation rate (ESR) was 31 mm/hr and C-reactive protein (CRP) was 6.24 mg/dl. Our differential diagnosis included deep infection including osteomyelitis, heterotopic ossification, and deep vein thrombosis (DVT) at the time because of high fever and increased inflammatory markers such as CRP, ESR and leukocytosis. Plain film of the pelvis showed no abnormalities (Fig. 1-A). CT angiography of lower extremities demonstrated no evidence suggesting deep vein thrombosis. Three phase bone scintigraphy showed mildly increased blood pooling and bone uptake in both proximal femurs. A thigh contrast MRI showed intramuscular fluid collections that appeared hyperintense on T2-weighted imaging (T2WI) and hypointense with a hyperintense rim on T1-weighed imaging (T1WI) while T1WI after fat saturation and gadolinium enhancement showed peripheral enhancement. Fluid collection was also noted in both hip joints. Diffusely increased signal intensity on T2WI was noted in both buttocks and proximal thighs involving both iliopsoas, vastus, gluteus, adductor and right sartorius muscle groups. Diffusely increased thickness and signal intensity in the subcutaneous tissues of both buttocks and thighs were observed (Fig. 2). This finding suggested pyomyositis. Sono-guided aspiration of the collections was cultured for antibiotic sensitivities. There was no growth of microorganism and empiric antibiotics (ampicillin/sulbactam) were used for two weeks.
Although his fever subsided and inflammatory markers normalized with antibiotics, the swelling persisted, while limitation of motion, especially in hip external rotation and flexion contracture, worsened. In follow-up, simple radiographic imaging after ten days showed overt ossified lesions (Fig. 1-B). Indomethacin was prescribed and gentle stretching and range of motion exercises were performed. In addition, he was given radiotherapy with 2000 cGy in 10 fractions on the area. Range of motion was significantly improved. On follow-up MRI 40 days after the initial MRI, the extent of high signal intensity on T1WIs was decreased. Multiple hypointense amorphous materials consistent with calcification were observed around both lesser trochanteric areas. Loculated fluid collections from the previous study had resolved.
A 55-year-old man complained of left hip pain after slipping. He was diagnosed with an intertrochanteric fracture of left femur and underwent closed reduction and internal fixation. His past medical history is significant for a left basal ganglia lacunar infarct two years prior, right intertrochanteric femoral fracture treated by closed reduction and internal fixation. On postoperative day 29, CRP was elevated and he had a mild fever with a temperature of 37.9℃. The focus of inflammation was uncertain. On postoperative day 34, he presented with swelling around his left hip and upper thigh. As the hemoglobin level persistently decreased, CRP level remained elevated and thigh swelling worsened, we strongly suspected hemorrhage or infection around the operation site rather than deep vein thrombosis or HO. Simple X-ray showed no abnormal findings. A very small amount of fluid was detected on the left thigh by ultrasound but aspiration was not carried out due to insufficient volume. Bone scintigraphy obtained to exclude the possibility of HO showed no definite abnormal findings. On the MR scan, diffuse enhancement and increased signal intensity in the T2 weighted images were noted in the left proximal femur and hip muscles. These findings suggested pyomyositis (Fig. 3) and empiric antibiotic therapy (cefazolin) was administered for one week. A soft tissue mass with cloudy ossification around the left femoral neck was observed in simple X-ray taken one week later. Finally, the diagnosis of HO was made and overly aggressive passive range of motion exercise was avoided to prevent any trauma. Active-assistive and gentle passive range of motion were carefully initiated to maintain available joint motion and to avoid progressive contractures.
We encountered two cases of HO, both of which were mimicking pyomyositis from laboratory and imaging findings. The two cases presented here highlight many similarities and differences between the immature HO and pyomyositis in laboratory, imaging and clinical findings. When patients present with numerous medical comorbidities predisposing them to infectious conditions, immature HO can be extremely difficult to distinguish from deep infections such as pyomyositis, cellulitis and osteomyelitis. We investigated the longitudinal change (trajectory) of biomarkers along with basal examinations and imaging findings. For prevention and early diagnosis, it is necessary to differentiate from other mimicking conditions and to track the emergence and maturation of HO. Therefore, our two cases can be used as a detailed clinical template which has been interpreted with intercorrelation and the comparison of each trajectory.
Previous studies found that elevated alkaline phosphatase (ALP), ESR and CK levels are associated with HO.3,4 Elevation of ALP is a hallmark of HO and is considered a sensitive and convenient tool for early detection.3,5 ALP level may be elevated in the early stages of bone formation and returns to normal as the bone becomes mature. The change of serum ALP in our two cases was found only after confirmation of heterotopic ossification radiographically (Fig. 4). CK level was significantly increased along with CRP in case 1 and then normalized before the elevation of ALP (Fig. 4-A). In case 2, the CK level was not checked frequently to make the trajectory for its change, but we can also notice the increase of CRP and CK followed by the elevation of ALP (Fig. 4-B). It is known that elevated serum levels of CK have value in predicting the HO,6 and in anticipating a more severe state of HO7 Therefore, CK level could be a more sensitive marker to diagnose early HO than ALP level.
Conventional radiographs usually show the evolution of HO lesions with a detectable calcific density present 4-6 weeks after the results of the 3-phase bone scintigraphy have become positive.1 Three-phase bone scintigraphy is the most sensitive imaging modality for early detection of HO. Specifically, flow studies and blood-pool images detect incipient HO approximately 2.5 weeks after injury, with findings on delayed scan becoming positive approximately 1 week later.1 Scintigraphic findings may not be specific between the soft tissue infection and early HO. Both simple radiographic study and three-phase bone scintigraphy for our two cases were not helpful for early diagnosis when performed initially and only follow-up studies demonstrated evidence of HO. With maturation of HO, T2 signal intensity and contrast enhancement were decreased, and fat and cortical bone-equivalent signal intensity was increased. These findings are consistent with the literature.8,9 On MR imaging of our cases, the affected muscles appeared enlarged with heterogeneous areas of low signal intensity on T1-weighted images. On T2-weighted images, high signal intensity within the muscle was poorly marginated and hyperintense signal along the fascia was shown. Also, diffusely increased thickness and high signal intensity was noted in subcutaneous tissues. After intravenous administration of gadolinium contrast, we could see heterogenous and feature-like enhancement revealing intramuscular fluid collections with hypointensity and peripheral rim enhancement. Immature ossification lesions with central hyperintense signal intensity and marked rim enhancement may be confused with septic bursitis or abscess and immature HO with diffuse enhancement can be mistaken for soft-tissue infection.10 Therefore, correct recognition of HO is hindered by the insensitivity of MRI in the detection of matrix calcification in immature HO.
The principal findings upon examination were similar to those seen in the deep infection of the hip. The clinical findings were characterized by pain in the hip and features of systemic infection. Lab findings which reveal an elevated ESR, CRP and leukocytosis can be seen in both conditions.6 Nonetheless, we need to pay attention to a number of distinguishing features. Pyomyositis does not reduce hip movement so dramatically, and is associated with more discrete areas of tenderness.6 It may not be related to the elevation of ALP and CK. Therefore, the diagnosis of HO should be made with thorough information including a careful history, physical examination, specific biomarkers and imaging findings. Furthermore, initially negative examinations should be repeated if clinical suspicion remains. Even without any increase of ALP level, other biomarkers such as CK level could help to diagnose early HO.
One must keep in mind that increasing number of patients undergoing acute care and rehabilitation treatment are aging and thus have more medical comorbidities and chronic underlying conditions accompanying the neurologic and orthopedic problems. In clinical circumstances, it is practical to prescribe empirical antibiotics before identifying the exact microorganism. The current study suggest that we should predict the possibility of HO development later based on findings from thorough history taking, physical examination and serial biomarkers check-up as well as imaging studies and therefore, take preventive measures against HO by gentle remobilization and use of anti-inflammatory agents. A regular serum CK and ALP screening test for HO in prolonged immobilized patients may be an effective way to identify the early development of HO. Controversy remains as to which of the available therapy options are better for the prophylaxis.
The two cases described here cannot be used to generalize the characteristics of immature HO. More intensive studies including serial biomarkers and imaging studies will enhance our understandings and help to differentiate HO from other conditions.
References
1. Shehab D, Elgazzar AH, Collier BD. Heterotopic ossification. J Nucl Med. 2002; 43:346–353. PMID: 11884494.
2. Shirkhoda A, Armin AR, Bis KG, Makris J, Irwin RB, Shetty AN. MR imaging of myositis ossificans: variable patterns at different stages. J Magn Reson Imaging. 1995; 5:287–292. PMID: 7633105.

3. Singh RS, Craig MC, Katholi CR, Jackson AB, Mountz JM. The predictive value of creatine phosphokinase and alkaline phosphatase in identification of heterotopic ossification in patients after spinal cord injury. Arch Phys Med Rehabil. 2003; 84:1584–1588. PMID: 14639555.
4. Taly AB, Nair KP, Jayakumar PN, Ravishankar D, Kalaivani PL, Indiradevi B, Murali T. Neurogenic heterotopic ossification: a diagnostic and therapeutic challenge in neurorehabilitation. Neurol India. 2001; 49:37–40. PMID: 11303239.
5. McCarthy EF, Sundaram M. Heterotopic ossification: a review. Skeletal Radiol. 2005; 34:609–619. PMID: 16132978.

6. Al-Najar M, Obeidat F, Ajlouni J, Mithqal A, Hadidy A. Primary extensive pyomyositis in an immunocompetent patient: case report and literature review. Clin Rheumatol. 2010; 29:1469–1472. PMID: 20857159.

7. Sherman AL, Williams J, Patrick L, Banovac K. The value of serum creatine kinase in early diagnosis of heterotopic ossification. J Spinal Cord Med. 2003; 26:227–230. PMID: 14997963.

8. Wick L, Berger M, Knecht H, Glucker T, Ledermann HP. Magnetic resonance signal alterations in the acute onset of heterotopic ossification in patients with spinal cord injury. Eur Radiol. 2005; 15:1867–1875. PMID: 15856244.

9. Ledermann HP, Schweitzer ME, Morrison WB. Pelvic heterotopic ossification: MR imaging characteristics. Radiology. 2002; 222:189–195. PMID: 11756725.

10. Yu CW, Hsiao JK, Hsu CY, Shih TT. Bacterial pyomyositis: MRI and clinical correlation. Magn Reson Imaging. 2004; 22:1233–1241. PMID: 15607094.

Fig. 1
Changes in x-ray images in a 50-year-old tetraplegic man who had a heterotopic ossification after spinal cord injury; (A) Initial x-ray does not show any definite abnormal findings. (B) Periarticular calcifications are observed around both proximal femurs especially around right lesser trochanteric area (arrowheads). (C) Follow-up X-ray performed one month later shows increased calcifications (arrowheads).

Fig. 2
MRI findings in a 50-year-old tetraplegic man who had a heterotopic ossification after spinal cord injury; Axial (A) and Coronal (B) T2-weighted MR images display hyperintense swelling of muscles (thin arrows) and diffusely increased signal intensity in both buttocks and proximal thighs affecting both iliopsoas, vastus, gluteus and adductor muscle groups (thick arrows). Diffusely increased thickness and signal intensity in the subcutaneous tissues are noted (arrowheads). Coronal (C), axial (D) and sagittal (E) gadolinum-enhanced T1-weighted with fat saturation MR images show heterogenous and feather-like enhancement of the affected muscles such as gluteus maximus, vastus lateralis, vastus medialis and iliopsoas (thin arrows). Also, it reveals intramuscular fluid collections with hypointensity with peripheral rim enhancement (thick arrows).

Fig. 3
MRI findings in a 55-year-old man who had a heterotopic ossification after operation for intertrochanteric fracture of left femur; (A) Axial T2-weighted non-enhanced MR image shows diffusely increased signal intensity in left hip muscles (thin arrows) and hyperintense signal along the fascia (thick arrow). (B) Coronal T2-weighted non-enhanced MR image reveals hyperintense infiltration in hip girdle muscles (thin arrows).
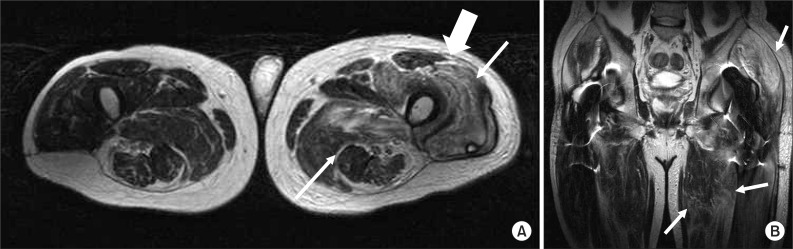
Fig. 4
Chronologic changes in the levels of parameters in Case 1 (A) and Case 2 (B). (A) Serum CRP and CK levels increased ahead of the development of swelling followed by the change of serum ALP after confirmation of heterotopic ossification radiographically. (B) CK level was not checked frequently to make the trajectory for its change, but we can notice the increase of CRP and CK followed by the elevation ALP after confirmation of heterotopic ossification radiographically.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download