Abstract
Purpose
Although metastasis occurs in 1 or 2 sentinel lymph nodes (SLNs), axillary lymph node dissection (ALND) has been widely not performed. For axillary staging and management, the necessity of intraoperative frozen section analysis of SLN has been controversial. The aim of this study is to evaluate the validity and benefit of SLN analysis by permanent section alone in clinically negative lymph node breast cancer patients.
Methods
We conducted a retrospective study of 283 cases with negative node clinical findings between July 2018 and August 2019 in Samsung Medical Center. Clinical nodal stage was evaluated by physical examination, breast ultrasonography, breast magnetic resonance imaging, and chest computerized tomography. The cases were divided into 2 groups; the permanent group had 151 cases (53.4%) and the frozen group had 132 cases (46.6%). We retrospectively analyzed the differences in the number of metastatic lymph nodes and rates of performed ALND between the 2 groups.
Results
Baseline and clinicopathologic characteristics between the 2 groups were well balanced. Three cases in the permanent group and 6 cases in the frozen group underwent additional or immediate ALND. The rates of ALND between the 2 groups were not significantly different (P = 0.312). The cased of 78.9% and 89.5% with metastatic lymph nodes in permanent and frozen groups were in the pathologic N1 stage, respectively.
Go to : 
Sentinel lymph node biopsy (SLNB) has been proposed as a less invasive alternative to axillary lymph node dissection (ALND) for axillary staging in early-stage breast cancer patients [1]. Intraoperative sentinel lymph node (SLN) analysis by frozen section can determine whether or not ALND should be performed [2]. However, ALND has been widely not performed although metastasis occurs in 1 or 2 SLNs. The need for ALND in patients with small metastases has been recently called into question and recent clinical trials such as ACOSOG Z0011 (American College of Surgeons Oncology Group Z0011), AMAROS (After Mapping of the Axilla: Radiotherapy or Surgery), and IBCSG 23-01 (International Breast Cancer Study Group 23-01) have addressed these issues [3456].
In Korea, all breast cancer patients are given equal healthcare in accordance with national insurance policies, resulting in the performance of intraoperative frozen section examination in most cases. However, some previous studies reported that the value of intraoperative frozen section examination of SLNs is questionable [27]. In addition to being incorrect, intraoperative frozen section examination can result in permanent tissue loss in patients with small-sized lymph nodes [8]. Permanent section examination alone may be sufficient in selected patients with low possibility of metastasis of SLNs; however, there are no studies concerning permanent section analysis of SLNs in Korea.
The aim of this study is to compare the number of metastatic lymph nodes and rates of performed ALND for clinically negative node breast cancer cases in which permanent section examination alone was performed to those in which both frozen and permanent section examination were performed. This comparison allows for the evaluation of the benefit of performing permanent section examination alone in clinically negative node breast cancer patients.
Go to : 
We retrospectively reviewed electronic medical records of 652 cases who underwent breast cancer surgery by the same surgeon between July 2018 and August 2019 at Samsung Medical Center, Seoul, Korea. Patients who were diagnosed with bilateral breast cancer were considered to be 2 cases. In this study, patients who were diagnosed with bilateral breast cancer were 7. This study was approved by the Institutional Review Board of Samsung Medical Center (No. 2019-11-021). Signed informed consent from the patients was not required.
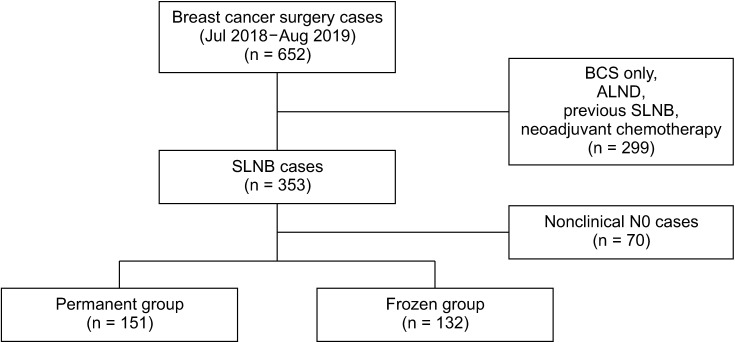
Inclusion criteria included cases of SLNB with clinically negative lymph node findings. Cases of patients who underwent breast surgery only, had planned ALND, or had previous SLNB or neoadjuvant chemotherapy for breast cancer were excluded. Although SLNB would have replaced ALND in clinically node-negative or -positive patients after neoadjuvant chemotherapy, the use of SLNB in patients receiving neoadjuvant chemotherapy remains controversial. The patients with neoadjuvant chemotherapy were excluded because neoadjuvant chemotherapy is associated with a high number of false-negative findings resulting in missed opportunities of immediate ALND [9101112]. Clinical nodal stage was evaluated by physical examination, breast ultrasonography, breast MRI, and chest CT preoperatively in accordance with South Korean health insurance policies. Cases with suspicious node findings on any one of these examinations were excluded. When suspicious or enlarged lymph nodes were detected intraoperatively, they were excised and analyzed by frozen biopsy. In this study, suspicious or enlarged lymph nodes were found in 13 cases, and they were analyzed by frozen section as SLNs. A total of 283 cases were selected and retrospectively reviewed and the cases were divided into 2 groups based on the analysis of lymph nodes by permanent section alone (permanent group) or frozen and permanent section (frozen group) (Fig. 1).
SLNs were detected by lymphoscintigraphy with technetium-99m sulfur-colloid diluted in normal saline solution or indigo carmine blue dye. Blue dye was performed when lymphoscintigraphy was not available or when SLN mapping had failed. Radioactive isotope or blue dye was injected at a subareolar or peritumoral site. Lymph nodes registering a 10% or higher count than the highest ex vivo count via gamma probe were regarded as the SLNs.
SLNs analysis were initially performed by frozen or permanent section. Lymph nodes for frozen section were sliced into several blocks at 2-mm perpendicular intervals along the long axis of lymph node. The 80-µm blocks with a minimum of 2 levels were embedded at −20℃, and samples were taken with a microtome setting of 4 µm. Analysis of frozen slides was performed by a pathologist specialized in breast pathology. We fixed each block of remaining tissue, taken with a microtome setting of 4 µm, in 10% formalin and embedded these tissues in paraffin. Serial sections were stained with H&E for pathological analysis. The other side of each node was stained for immunohistochemistry.
Statistical analyses were performed using IBM SPSS Statistics ver. 25.0 (IBM Corp., Armonk, NY, USA). Continuous variables were described as the mean and standard deviation with the range. Categorical variables were described as the number and percentage. Continuous variables of the 2 groups were compared with the Student t-test and Mann-Whitney tests. Chi-square test and Fisher exact test were conducted to compare categorical variables. Statistical significance was defined as P < 0.05 in all tests.
Go to : 
A total of 652 breast cancer surgeries were performed between July 2018 and August 2019 at Samsung Medical Center. Of these, 299 cases of patients who underwent breastconserving surgery only, planned ALND, previous SLNB, or neoadjuvant chemotherapy for breast cancer, and 70 cases in which clinically positive lymph nodes were excluded. The remaining 283 cases were divided based on the method of lymph node analysis. Permanent section alone group (permanent group) had 151 cases (53.4%) and the frozen and permanent section group (frozen group) had 132 cases (46.6%) (Fig. 1).
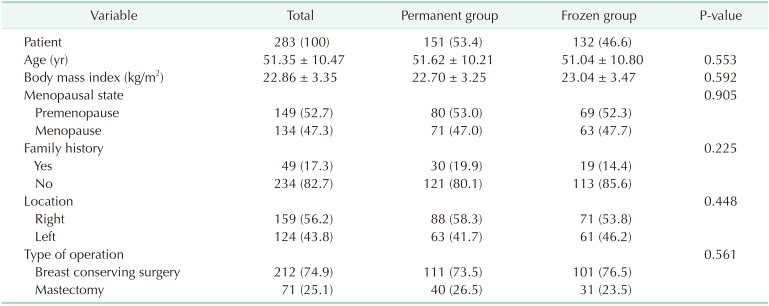
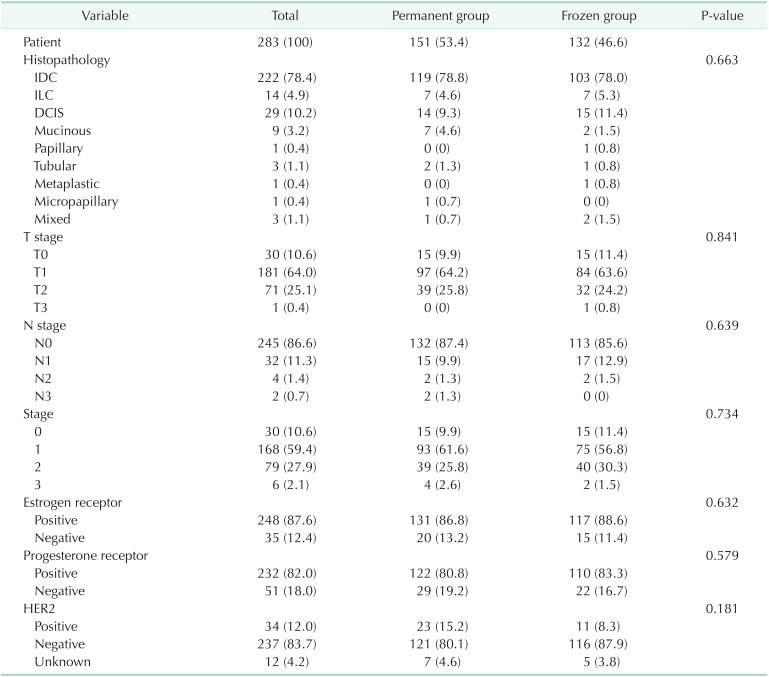
The characteristics of the cases between the 2 groups were well balanced. Breast-conserving surgery was performed 73.5% and 76.5% and pathologic T1 stage was 64.2% and 63.6% in the permanent and frozen groups, respectively. The cases with human epidermal growth factor receptor 2 (HER2)-negative were 83.7% in 2 groups; neoadjuvant systemic treatment is the standard of care in most breast cancer patients with HER2-positive or triple negative subtype. All of the other variables of clinicopathologic characteristics were not significantly different between the 2 groups (Tables 1, 2).
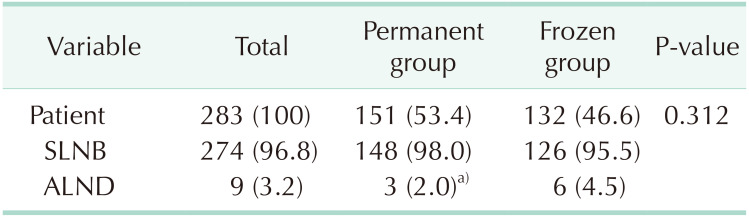
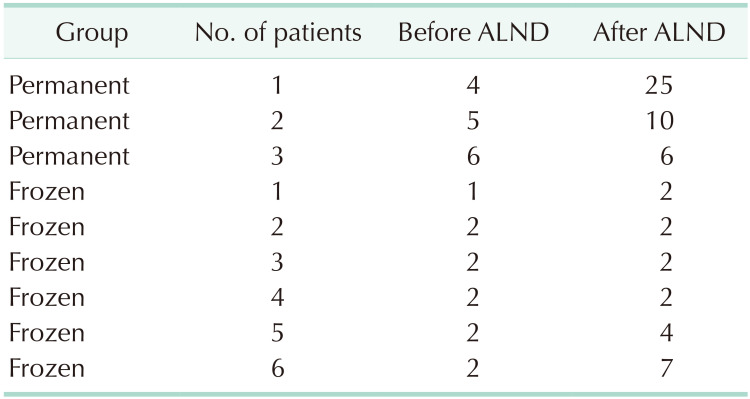
Three patients in the permanent group and 6 patients in the frozen group underwent additional or immediate ALND depending on the final or intraoperative pathologic results, and the rates of ALND between permanent and frozen groups were not significantly different (P = 0.312). Two of 3 patients in permanent group and 3 of 6 patients in frozen group had additional metastatic lymph nodes after ALND. Four patients in the permanent group and 2 patients in the frozen group were diagnosed with pathologic N2 or higher stage. One patient with pathologic N2 stage in permanent group strongly refused additional ALND and decided to have adjuvant systemic chemotherapy and radiotherapy. The number of patients who had additional ALND was not significantly different whether patients had breast-conserving surgery or mastectomy (P = 0.575) (Tables 2, 3, 4).
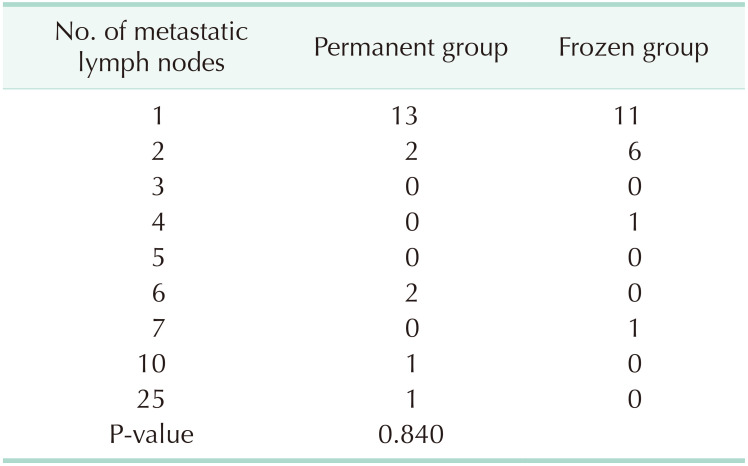
There were significantly more retrieved lymph nodes in the permanent group than in the frozen group (4.2 ± 2.2 vs. 3.5 ± 2.0, P = 0.009). The rates of cases with 1 or 2 metastatic lymph nodes were 78.9% and 89.5% between 2 groups in the cases with metastatic lymph nodes, respectively. Three false-negative cases were found in the frozen group; however, additional ALND was not performed because these cases were N1 (Table 5).
Operation time of cases who underwent mastectomy with SLNB showed significantly shorter in permanent group than frozen group (P = 0.008), whereas operation time of cases who underwent breast-conserving surgery with SLNB showed no significant difference (P = 0.105) (Table 6).
Go to : 
Before publication of the initial ACOSOG Z0011 trial results, there was a general consensus that ALND was necessary for better cancer control when metastases were identified in SLNs. The results of this trial demonstrated that morbidity associated with ALND can be avoided without affecting cancer control [3]. Currently, SLNB has replaced ALND for the assessment of axillary staging in clinically negative node patients.
Health insurance does not apply to all patients universally in the United States, so there is inequality among populations concerning examinations and treatment [13]. For this reason, intraoperative SLN examination by frozen section has been performed in only some patients scheduled for SLNB. Additionally, there has been a significant decline in utilization of intraoperative frozen section examination of SLNs after publication of Z0011 [1415]. In Korea, all patients with breast cancer are under the same insurance coverage; therefore, intraoperative frozen section examination of SLNs has been usually performed. The base cost of frozen biopsy was 37,674 won in Korean currency, and a maximum cost was 263,744 Korean won in our institution. If intraoperative frozen section examination of SLNs in clinically negative node breast cancer patients was routinely analyzed, it is not cost-effective because this couldn't be allocated elsewhere.
Intraoperative frozen section analysis of SLNs may avoid the need for additional ALND in positive axillary node patients. However, intraoperative SLNs analysis in axillary management has been still controversial. Reported false-negative rates for intraoperative frozen section examination in breast cancer range from 9% to 59%, and artifacts that occur in the frozen section process can affect permanent analysis [3161718]. In addition, intraoperative frozen section examination can result in permanent tissue loss in patients with small-sized lymph nodes [8]. After the ACOSOG Z0011 study, the criteria for performing ALND have been relaxed for patients who had 1 or 2 metastatic SLNs and were scheduled for breast-conserving surgery, adjuvant chemotherapy, and adjuvant radiation therapy [3]. The results of the AMAROS trial confirmed that the type of axillary management (axillary radiotherapy or ALND) in patients with a positive SLNs does not have an effect on survival [4]. Moreover, the results of the IBCSG 23-01 trial indicated that further axillary surgery was not necessary when 1 or more SLNs with micrometastases were seen [6]. Therefore, ALND have been routinely not indicated in patients with metastatic SLNs.
In our study, the patients who were not indicated for SLNB in ACOSOG Z0011 or AMAROS trials received additional or immediate ALND. Patients with pathologic N2 or higher stages accounted for 2.6% and 1.5% of the patients in the permanent and frozen groups, respectively. In the permanent group, out of the 4 patients who were pathologically confirmed N2 or higher stage, 3 patients received additional ALND. Two of these patients had additional metastatic lymph nodes after ALND, even though there were no suspicious lymph node findings in imaging studies preoperatively. The other 1 patient strongly refused ALND, instead deciding to undergo adjuvant systemic chemotherapy and radiotherapy. In the frozen group, 2 patients were confirmed as pathologic N2 or higher stage, but 6 patients received ALND. Four of these patients were expected to be higher than pathologic T3 and N1 stage perioperatively. The other 2 patients were upstaged from N1 to N2 stage. Three of 6 patients had additional metastatic lymph nodes after immediate ALND. Therefore, only 4.5% of frozen group cases received immediate ALND. This was not significantly different from the 2.0% of permanent group patients receiving additional ALND (Tables 2, 3, 4).
In the frozen group, suspicious or enlarged lymph nodes were detected in 13 patients, and they were analyzed as SLNs. Five of the 13 patients had 1 or more metastatic lymph nodes, and 2 of 5 patients had 1 metastatic lymph node each. Three of 5 patients had 2 metastatic lymph nodes each and they received immediate ALND because these patients were expected to be higher than pathologic T3 stage. Therefore, 3 of 13 patients who had suspicious or enlarged lymph nodes were received immediate ALND, and 1 patient was upstaged from N1 to N2. Even though rates of ALND were not significantly different between 2 groups in this study, metastatic lymph nodes were found in about 23% of patients who had suspicious or enlarged lymph nodes and it may have affected the low rate of ALND in the permanent group.
In addition, we investigated operation time which spent on analyzing intraoperative frozen section of SLNs by type of operation. In patients who underwent mastectomy with SLNB by frozen section analysis, mean operation time took about 47 more minutes (Table 6). However, operation time of patients who underwent breast-conserving surgery with SLNB showed no significant difference regardless of method of SLN analysis. The result would have been affected because time for intraoperative frozen section analysis of margin of cancer was included. However, this study couldn't be analyzed in consideration of that time for margin of cancer.
Some researchers have questioned whether frozen section examination of SLNs may be routinely indicated in any breast cancer patients and some previous studies have mentioned the necessity of intraoperative frozen section analysis of SLNs. In some studies, frozen section examination of SLN have low sensitivity and high false-negative rates in detecting micrometastases [81819]. In other studies, the benefit of routine intraoperative frozen section analysis has tendency to increase with tumor size; therefore, routine intraoperative frozen section analysis may not be indicated in patients with small breast cancer [161820]. Arora et al. [21] reported that there is a higher rate of SLNs positivity in specimens evaluated by both frozen and permanent section; intraoperative frozen section analysis offers the advantage of decreased delay over axillary dissections. Geertsema et al. [7] reported that when metastases are only detected by permanent section examination in clinically staged T1–2N0 breast cancer, ALND will rarely lead to postsurgical treatment changes although discordance between intraoperative frozen and permanent section analysis of SLNs is common and substantial proportions of patients had additional lymph node metastases in any case.
Our study showed that, although permanent section analysis alone was performed, a small number of patients underwent additional ALND. Therefore, permanent section SLNs analysis alone can be indicated in clinically negative node patients. In addition, intraoperative frozen section analysis has a tendency to be more time-consuming and costly. However, this study had a few limitations. This study was investigated in a single institution by 1 surgeon and number of cases was relatively small. Also, it was retrospectively reviewed; therefore, there has been a possibility of selection bias. In addition, out of patients who had suspicious or enlarged lymph nodes which were pathologically confirmed metastatic lymph nodes in frozen biopsy, about 23% of patients underwent immediate ALND and it may have influenced the low rate of ALND in permanent group. Despite these limitations, however, this study is meaningful because this is the first report related validity and benefit of SLNs analysis by permanent section alone in clinically negative lymph node breast cancer patients in Korea.
In conclusion, ALND was not indicated for most patients with clinically negative nodes in our study. Even when ALND was indicated following permanent section SLNs examination alone, the method of SLNs analysis did not have a significant impact on avoiding additional surgery in clinically negative node patients; intraoperative frozen section examination of SLNs is not increasingly beneficial. Therefore, permanent section SLNs examination alone may be performed in clinically negative axillary node breast cancer patients. Our findings can help to avoid unnecessary intraoperative frozen section analysis.
Go to : 
References
1. Kelley MC, Hansen N, McMasters KM. Lymphatic mapping and sentinel lymphadenectomy for breast cancer. Am J Surg. 2004; 188:49–61. PMID: 15219485.
2. van der Noordaa ME, Vrancken Peeters MT, Rutgers EJ. The intraoperative assessment of sentinel nodes: standards and controversies. Breast. 2017; 34 Suppl 1:S64–S69. PMID: 28673534.
3. Giuliano AE, Ballman KV, McCall L, Beitsch PD, Brennan MB, Kelemen PR, et al. Effect of axillary dissection vs no axillary dissection on 10-year overall survival among women with invasive breast cancer and sentinel node metastasis: the ACOSOG Z0011 (Alliance) randomized clinical trial. JAMA. 2017; 318:918–926. PMID: 28898379.
4. Donker M, van Tienhoven G, Straver ME, Meijnen P, van de Velde CJ, Mansel RE, et al. Radiotherapy or surgery of the axilla after a positive sentinel node in breast cancer (EORTC 10981-22023 AMAROS): a randomised, multicentre, open-label, phase 3 non-inferiority trial. Lancet Oncol. 2014; 15:1303–1310. PMID: 25439688.
5. Poling JS, Tsangaris TN, Argani P, Cimino-Mathews A. Frozen section evaluation of breast carcinoma sentinel lymph nodes: a retrospective review of 1,940 cases. Breast Cancer Res Treat. 2014; 148:355–361. PMID: 25318925.
6. Galimberti V, Cole BF, Viale G, Veronesi P, Vicini E, Intra M, et al. Axillary dissection versus no axillary dissection in patients with breast cancer and sentinel-node micrometastases (IBCSG 23-01): 10-year follow-up of a randomised, controlled phase 3 trial. Lancet Oncol. 2018; 19:1385–1393. PMID: 30196031.
7. Geertsema D, Gobardhan PD, Madsen EV, Albregts M, van Gorp J, de Hooge P, et al. Discordance of intraoperative frozen section analysis with definitive histology of sentinel lymph nodes in breast cancer surgery: complementary axillary lymph node dissection is irrelevant for subsequent systemic therapy. Ann Surg Oncol. 2010; 17:2690–2695. PMID: 20422461.
8. Yoon KH, Park S, Kim JY, Park HS, Kim SI, Cho YU, et al. Is the frozen section examination for sentinel lymph node necessary in early breast cancer patients? Ann Surg Treat Res. 2019; 97:49–57. PMID: 31388507.
9. Park S, Lee JE, Paik HJ, Ryu JM, Bae SY, Lee SK, et al. Feasibility and prognostic effect of sentinel lymph node biopsy after neoadjuvant chemotherapy in cytologyproven, node-positive breast cancer. Clin Breast Cancer. 2017; 17:e19–e29. PMID: 27495997.
10. Takahashi M, Jinno H, Hayashida T, Sakata M, Asakura K, Kitagawa Y. Correlation between clinical nodal status and sentinel lymph node biopsy false negative rate after neoadjuvant chemotherapy. World J Surg. 2012; 36:2847–2852. PMID: 22806206.
11. Kuehn T, Bauerfeind I, Fehm T, Fleige B, Hausschild M, Helms G, et al. Sentinellymph-node biopsy in patients with breast cancer before and after neoadjuvant chemotherapy (SENTINA): a prospective, multicentre cohort study. Lancet Oncol. 2013; 14:609–618. PMID: 23683750.
12. Boughey JC, Suman VJ, Mittendorf EA, Ahrendt GM, Wilke LG, Taback B, et al. Sentinel lymph node surgery after neoadjuvant chemotherapy in patients with node-positive breast cancer: the ACOSOG Z1071 (Alliance) clinical trial. JAMA. 2013; 310:1455–1461. PMID: 24101169.
13. Yabroff KR, Gansler T, Wender RC, Cullen KJ, Brawley OW. Minimizing the burden of cancer in the United States: goals for a high-performing health care system. CA Cancer J Clin. 2019; 69:166–183. PMID: 30786025.
14. Jorns JM, Kidwell KM. Sentinel lymph node frozen-section utilization declines after publication of American College of Surgeons Oncology Group Z0011 trial results with no change in subsequent surger y for axi l lar y lymph node dissection. Am J Clin Pathol. 2016; 146:57–66. PMID: 27373347.
15. Bishop JA, Sun J, Ajkay N, Sanders MA. Decline in frozen section diagnosis for axillary sentinel lymph nodes as a result of the American College of Surgeons Oncology Group Z0011 trial. Arch Pathol Lab Med. 2016; 140:830–835. PMID: 26716950.
16. Francissen CM, van la Parra RF, Mulder AH, Bosch AM, de Roos WK. Evaluation of the benefit of routine intraoperative frozen section analysis of sentinel lymph nodes in breast cancer. ISRN Oncol. 2013; 2013:843793. PMID: 24167745.
17. Akay CL, Albarracin C, Torstenson T, Bassett R, Mittendorf EA, Yi M, et al. Factors impacting the accuracy of intra-operative evaluation of sentinel lymph nodes in breast cancer. Breast J. 2018; 24:28–34. PMID: 28608612.
18. Weiser MR, Montgomery LL, Susnik B, Tan LK, Borgen PI, Cody HS. Is routine intraoperative frozen-section examination of sentinel lymph nodes in breast cancer worthwhile? Ann Surg Oncol. 2000; 7:651–655. PMID: 11034241.
19. Lombardi A, Nigri G, Maggi S, Stanzani G, Vitale V, Vecchione A, et al. Role of frozen section in sentinel lymph node biopsy for breast cancer in the era of the ACOSOG Z0011 and IBCSG 23-10 trials. Surgeon. 2018; 16:232–236. PMID: 29329752.
20. Wada N, Imoto S, Hasebe T, Ochiai A, Ebihara S, Moriyama N. Evaluation of intraoperative frozen section diagnosis of sentinel lymph nodes in breast cancer. Jpn J Clin Oncol. 2004; 34:113–117. PMID: 15078905.
21. Arora N, Martins D, Huston TL, Christos P, Hoda S, Osborne MP, et al. Sentinel node positivity rates with and without frozen section for breast cancer. Ann Surg Oncol. 2008; 15:256–261. PMID: 17879116.
Go to : 




 PDF
PDF Citation
Citation Print
Print




 XML Download
XML Download