DISCUSSION
Since GBC manifests as vague or with no symptoms, it cannot be diagnosed by clinical symptoms alone and objective and noninvasive tools are necessary to accurately diagnose GBC. Therefore, novel diagnostic biomarkers with high sensitivity that can detect the disease in a screening population without symptoms [
8], and can detect the disease specifically and noninvasively are needed [
9]. Many tumor biomarkers have been utilized to detect malignant disease; for example, CA 125 for ovarian cancer [
10], or prothrombin in vitamin K absence-II and alpha-fetoprotein for hepatocellular carcinoma [
11]. Although some biomarkers, such as cytokeratin 19 fragments, matrix metalloproteinase-7, or osteopontin were proposed as potential diagnostic biomarkers for BTC, these biomarkers were investigated with small-scaled studies, and were only utilized in limited situations [
12131415]. Although serum CA 19-9 and CEA levels have been considered as diagnostic biomarkers for BTC, their diagnostic performance for BTC remains controversial. Some previous studies have investigated the diagnostic performance of serum CA 19-9 and CEA in terms of BTC, but they were small-scale and not specific to GBC [
16]. Although the incidence of BTC is relatively high in Chile, South America, India, and Eastern Asia, few studies on BTC can be found in these countries [
17]. This study evaluated the diagnostic performance of these 2 biomarkers with a large number of GBC patients and a control group in a high-incidence country and demonstrated their clinical usefulness as a screening program for GBC.
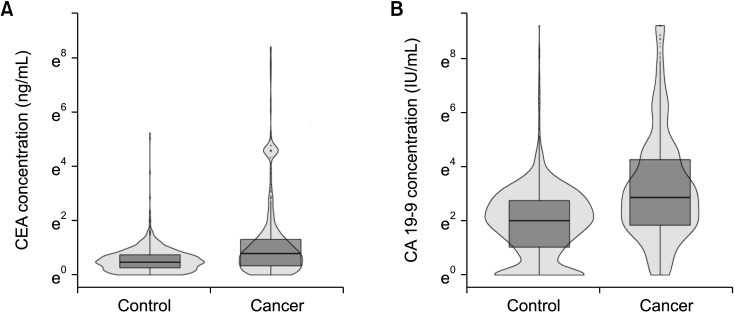
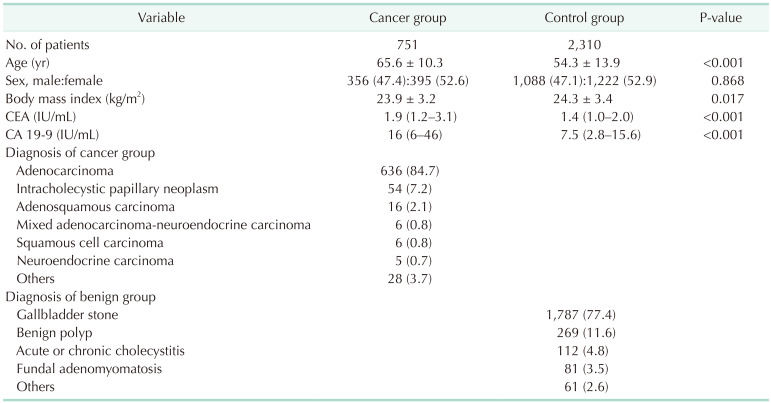
Age, body mass index (BMI), or sex could affect preoperative CEA and CA 19-9 level. Therefore, the propensity score matching analysis was performed and the ranges of preoperative CEA and CA 19-9 level were compared before and after propensity score matching. Because age (65.3 years vs. 54.3 years, P < 0.001) and BMI (23.9 kg/m2
vs. 24.3 kg/m2, P = 0.017) were significantly different between the GBC and normal control group, the propensity score matching with age and sex was performed and the median and IQR of CEA and CA 19-9 were similar before and after propensity score matching. Therefore, age, sex, and BMI might not affect the preoperative serum CEA and CA 19-9 level. Finally, we analyzed all the data collected.
Conventionally, the cutoff value for determining benign and malignant disease at diagnosis is 5 ng/mL of serum CEA and 37 IU/mL of serum CA 19-9. However, these cutoff values were based on the results from colorectal cancer (CEA, 5 ng/mL) and pancreatic cancer (CA 19-9, 37 IU/mL) [
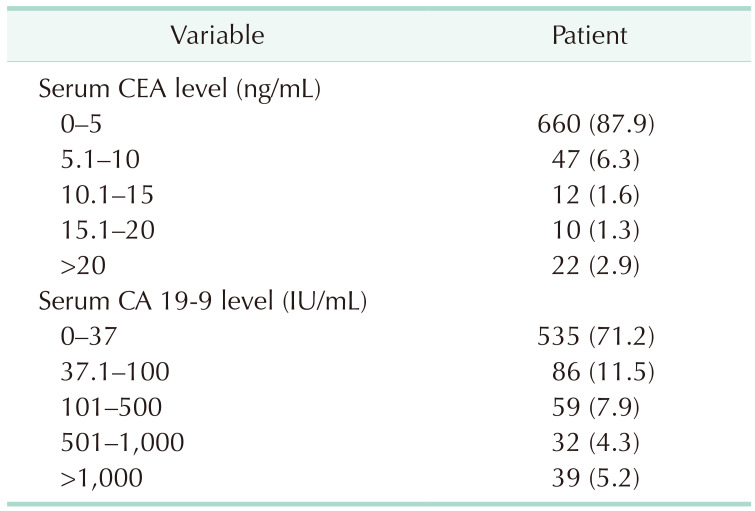
18], and no studies existed in regards to the GBC. In the present study, 87.9% of GBC patients had CEA levels of ≤5 ng/mL and 71.2% of patients had CA 19-9 levels of ≤37 IU/mL (
Table 2). Most patients with GBC had these 2 biomarkers within the conventional cutoff values. Therefore, these cutoff values were not suitable for diagnosing GBC and new criteria would be needed to screen for GBC.
CA 19-9 is a tumor-associated antigen, initially isolated as a colorectal cancer antigen, but also presented in epithelial cells of the gallbladder, biliary tract, pancreas, and stomach [
19]. Serum CA 19-9 level is elevated in patients with not only malignant disease, but also liver cirrhosis, jaundice, or inflammatory conditions [
1920]. In contrast, serum CA 19-9 was undetectable in some patients with Lewis-antigen negative phenotype [
21]. The median sensitivity of CA 19-9 for the diagnosis of pancreatic cancer was reported as 79% [
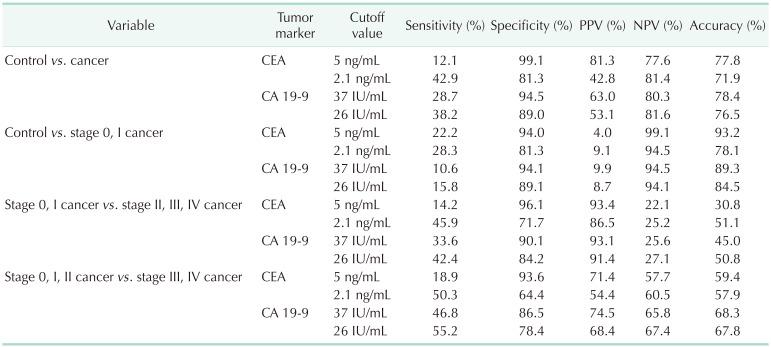
22]. However, in the present study, the sensitivity of CA 19-9 for the diagnosis of GBC did not exceed 30% at the conventional cutoff values (37 IU/mL) (
Table 3). Therefore, the 37 IU/mL is not a reliable cutoff value of serum CA 19-9 for the diagnosis of GBC without reliable sensitivity as the screening test.
CEA is a glycoprotein involved in cell adhesion and is normally produced during fetal development; however, its production stops before birth [
23]. While serum CEA is elevated in patients with colorectal cancer, it can also be elevated in smokers, and in cases of ulcerative colitis, liver cirrhosis, and chronic bronchitis [
24]. The sensitivity of CEA for diagnosis was reported to be low, with a sensitivity of 50% in pancreatic cancer [
25], and 33% in BTC [
6]. In this study, the sensitivity of CEA was 12.1% for diagnosing GBC and 22.2% for diagnosing early GBC when the cutoff value was 5 ng/mL (
Table 3). Current cutoff values of CEA and CA 19-9 are not suitable for the diagnosis of entire GBC and that of early-stage GBC. Therefore, new cutoff values suitable for the diagnosis of GBC are needed.
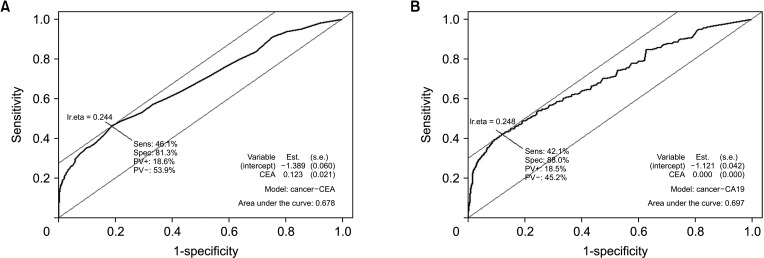
In this study, the ROC curves of serum CEA and CA 19-9 levels were evaluated using the database of 751 GBC patients and 2,310 normal controls (
Fig. 3A). The optimal cutoff value of CEA was set to 2.1 ng/mL (
Fig. 3A) and that of CA 19-9 was set to 26 IU/mL (
Fig. 3B). The sensitivity of the proposed cutoff value was higher than that with the previous cutoff value that the sensitivity of CEA and CA 19-9 for the diagnosis of GBC rose to about 40% (
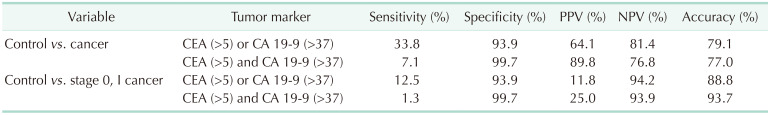
Table 3). However, a sensitivity of 40% is not sufficient for a novel diagnostic tool. Furthermore, the sensitivity of CEA and CA 19-9 did not exceed 30% when these biomarkers were utilized to diagnose early GBC, and that of the composition of 2 biomarkers remained less than 30%. Therefore, these 2 biomarkers would not be suitable for detecting early GBC.
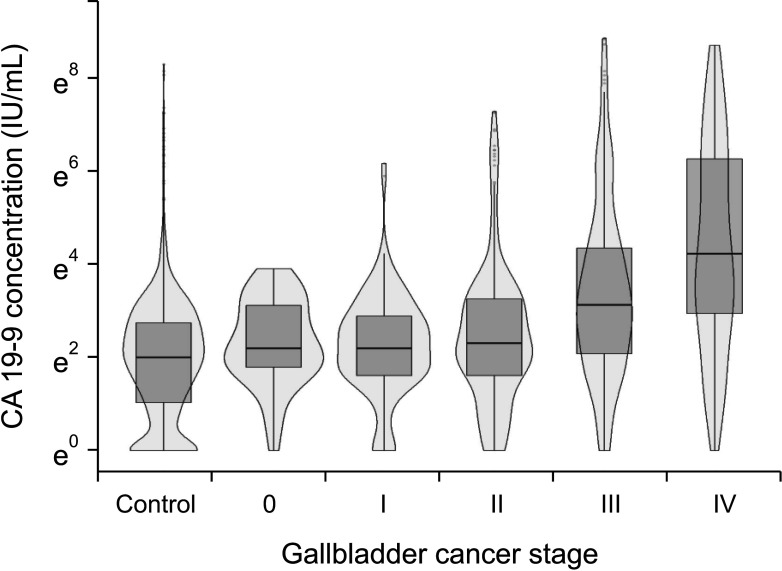
In regards to the disease severity, the sensitivity of the 2 biomarkers exceeded 50% when they were utilized to differentiate stage III and IV cancers from stage 0, I, and II cancers. Furthermore, the stage III and IV groups showed a higher distribution of serum CA 19-9 levels than did the stage I and II groups (median, 34 IU/mL
vs. 11 IU/mL;
Supplementary Fig. 1A). Considering that both biomarkers had high specificity for diagnosis and reliable sensitivity for differentiating cancer stage, these 2 biomarkers would be useful for the evaluation of disease severity, but not as a screening tool.
Radiologic modalities are the gold standard tools for diagnosing GBC. Ultrasonography is widely utilized for screening due to its noninvasiveness and real-time evaluation. Although ultrasonography has good performance for diagnosing gallbladder polyps, it is still difficult to differentiate neoplastic polyps from non-neoplastic polyps [
26]. In particular, the sensitivity of CT images was reported to be 54% when the gallbladder lesion was T1 with only a thickened wall [
27]. Because the ultrasonography results depend on the practitioner, new diagnostic biomarkers with higher sensitivity and objectivity should be included in the screening programs for GBC. If GBC can be diagnosed at an early stage, minimally invasive surgery such as laparoscopic or robotic cholecystectomy can be the appropriate treatment with comparable perioperative outcomes [
28].
Although this study had some limitations related to a retrospective cohort study, this study evaluated more than 700 surgically resected GBC patients and over 2,000 normal controls. Considering that most previous studies on GBC analyzed a small number of patients in Western countries, this study was conducted in the Republic of Korea, in which the prevalence of GBC is much higher than that in other Western countries [
29]. One study reported a high incidence of specific genomic mutations in Korea, which was not frequently found in other Western countries [
30]. Therefore, if these researchers investigate new biomarkers for GBC, they should consider the difference in prevalence related to geographic variation.
In conclusion, serum CEA and CA 19-9 had low sensitivity and high specificity for diagnosing GBC, especially for the early GBC, thereby making them unsuitable for screening GBC patients from the normal control group. New biomarkers with high sensitivity for the diagnosis of GBC should be explored.




 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download