1. Tei M, Otsuka M, Suzuki Y, Kishi K, Tanemura M, Akamatsu H. Safety and feasibility of single-port laparoscopic low anterior resection for upper rectal cancer. Am J Surg. 2018; 216:1101–1106. PMID:
29631909.

2. Bademler S, Koza KB, Ucuncu MZ, Tokmak H, Bakir B, Oral EN, et al. Standardized laparoscopic sphincter-preserving total mesorectal excision for rectal cancer: median of 10 years' long-term oncologic outcome in 217 unselected consecutive patients. Surg Laparosc Endosc Percutan Tech. 2019; 29:354–361. PMID:
31107850.

3. Wu Z, Zhou W, Chen F, Wang W, Feng Y. Short-term outcomes of transanal versus laparoscopic total mesorectal excision: a systematic review and meta-analysis of cohort studies. J Cancer. 2019; 10:341–354. PMID:
30719128.

4. Zhang X, Gao Y, Dai X, Zhang H, Shang Z, Cai X, et al. Short- and long-term outcomes of transanal versus laparoscopic total mesorectal excision for mid-to-low rectal cancer: a meta-analysis. Surg Endosc. 2019; 33:972–985. PMID:
30374790.

5. Detering R, Roodbeen SX, van Oostendorp SE, Dekker JT, Sietses C, Bemelman WA, et al. Three-year nationwide experience with transanal total mesorectal excision for rectal cancer in the Netherlands: a propensity score-matched comparison with conventional laparoscopic total mesorectal excision. J Am Coll Surg. 2019; 228:235–244. PMID:
30639298.

6. Chen YT, Kiu KT, Yen MH, Chang TC. Comparison of the short-term outcomes in lower rectal cancer using three different surgical techniques: Transanal total mesorectal excision (TME), laparoscopic TME, and open TME. Asian J Surg. 2019; 42:674–680. PMID:
30318319.

7. Holmer C, Benz S, Fichtner-Feigl S, Jehle EC, Kienle P, Post S, et al. Transanal total mesorectal excision-a critical appraisal. Chirurg. 2019; 90:478–486. PMID:
30911795.
8. Daabiss M. American Society of Anaesthesiologists physical status classification. Indian J Anaesth. 2011; 55:111–115. PMID:
21712864.

9. Palys KE, Berger VW. A note on the Jadad score as an efficient tool for measuring trial quality. J Gastrointest Surg. 2013; 17:1170–1171. PMID:
23233271.

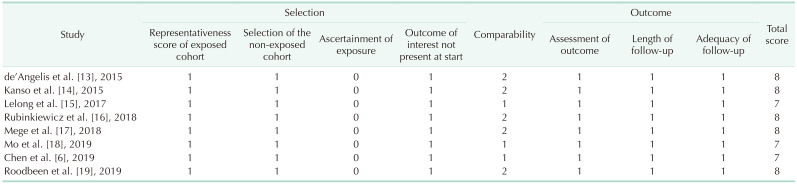
10. Wells GA, Shea B, O'Connell D, Peterson J, Welch V, Losos M, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses [Internet]. Ottawa: Ottawa Hospital Research Institute;2012. cited 2020 Dec 30. Available from:
www.ohri.ca/programs/clinical_epidemiology/oxford.asp.
11. Denost Q, Adam JP, Rullier A, Buscail E, Laurent C, Rullier E. Perineal transanal approach: a new standard for laparoscopic sphincter-saving resection in low rectal cancer, a randomized trial. Ann Surg. 2014; 260:993–999. PMID:
24950270.
12. Ren J, Liu S, Luo H, Wang B, Wu F. Comparison of short-term efficacy of transanal total mesorectal excision and laparoscopic total mesorectal excision in low rectal cancer. Asian J Surg. 2020; 05. DOI:
10.1016/j.asjsur.2020.05.007. [Epub].

13. de'Angelis N, Portigliotti L, Azoulay D, Brunetti F. Transanal total mesorectal excision for rectal cancer: a single center experience and systematic review of the literature. Langenbecks Arch Surg. 2015; 400:945–959. PMID:
26497544.
14. Kanso F, Maggiori L, Debove C, Chau A, Ferron M, Panis Y. Perineal or abdominal approach first during intersphincteric resection for low rectal cancer: which is the best strategy? Dis Colon Rectum. 2015; 58:637–644. PMID:
26200677.
15. Lelong B, Meillat H, Zemmour C, Poizat F, Ewald J, Mege D, et al. Short- and mid-term outcomes after endoscopic transanal or laparoscopic transabdominal total mesorectal excision for low rectal cancer: a single institutional case-control study. J Am Coll Surg. 2017; 224:917–925. PMID:
28024946.

16. Rubinkiewicz M, Nowakowski M, Wierdak M, Mizera M, Dembiński M, Pisarska M, et al. Transanal total mesorectal excision for low rectal cancer: a case-matched study comparing TaTME versus standard laparoscopic TME. Cancer Manag Res. 2018; 10:5239–5245. PMID:
30464621.

17. Mege D, Hain E, Lakkis Z, Maggiori L, Prost À, Panis Y. Is trans-anal total mesorectal excision really safe and better than laparoscopic total mesorectal excision with a perineal approach first in patients with low rectal cancer?: a learning curve with case-matched study in 68 patients. Colorectal Dis. 2018; 20:O143–O151. PMID:
29693307.

18. Mo SP, Yang P, Chen YJ. Clinical outcome and safety analysis of laparoscopic assisted TaTME for patients with low rectal cancer. Chin J Oper Proc Gen Surg (electronic edition). 2019; 13:155–158.
19. Roodbeen SX, Penna M, Mackenzie H, Kusters M, Slater A, Jones OM, et al. Transanal total mesorectal excision (TaTME) versus laparoscopic TME for MRI-defined low rectal cancer: a propensity score-matched analysis of oncological outcomes. Surg Endosc. 2019; 33:2459–2467. PMID:
30350103.

20. Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg. 2009; 250:187–196. PMID:
19638912.
21. Sohn DK, Park SC, Kim MJ, Chang HJ, Han KS, Oh JH. Feasibility of transanal total mesorectal excision in cases with challenging patient and tumor characteristics. Ann Surg Treat Res. 2019; 96:123–130. PMID:
30838184.

22. Sylla P, Rattner DW, Delgado S, Lacy AM. NOTES transanal rectal cancer resection using transanal endoscopic microsurgery and laparoscopic assistance. Surg Endosc. 2010; 24:1205–1210. PMID:
20186432.

23. Zheng M, Ma J. Transanal total mesorectal excision in laparoscopic surgery for lower rectal cancer: leading actor or supporting actor. Zhonghua Wei Chang Wai Ke Za Zhi. 2019; 22:215–219. PMID:
30919371.
24. Perdawood SK, Warnecke M, Bjoern MX, Eiholm S. The pattern of defects in mesorectal specimens: is there a difference between transanal and laparoscopic approaches? Scand J Surg. 2019; 108:49–54. PMID:
29966503.

25. Veltcamp Helbach M, Koedam TW, Knol JJ, Diederik A, Spaargaren GJ, Bonjer HJ, et al. Residual mesorectum on postoperative magnetic resonance imaging following transanal total mesorectal excision (TaTME) and laparoscopic total mesorectal excision (LapTME) in rectal cancer. Surg Endosc. 2019; 33:94–102. PMID:
29967990.

26. Wasmuth HH, Faerden AE, Myklebust TÅ, Pfeffer F, Norderval S, Riis R, et al. Transanal total mesorectal excision for rectal cancer has been suspended in Norway. Br J Surg. 2020; 107:121–130. PMID:
31802481.

27. Lacy AM, Nogueira ST, de Lacy FB. Comment on: Norwegian moratorium on transanal total mesorectal excision. Br J Surg. 2019; 106:1855.

28. Bolshinsky V, Shawki S, Steele S. CO2 embolus during transanal total mesorectal excision: thoughts on aetiology. Colorectal Dis. 2019; 21:6–7.
29. Sammour T, Chang GJ. Lateral pelvic lymph node dissection and radiation treatment for rectal cancer: mutually exclusive or mutually beneficial? Ann Gastroenterol Surg. 2018; 2:348–350. PMID:
30238075.

30. Hashiguchi Y, Muro K, Saito Y, Ito Y, Ajioka Y, Hamaguchi T, et al. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2019 for the treatment of colorectal cancer. Int J Clin Oncol. 2020; 25:1–42. PMID:
31203527.





 PDF
PDF Citation
Citation Print
Print



 XML Download
XML Download