INTRODUCTION
Glanzmann thrombasthenia (GT) is a rare platelet aggregation disorder associated with abnormalities in glycoprotein IIb/IIIa (alpha IIb/beta3 integrin). This disorder is inherited as an autosomal recessive trait and occurs equally in both sexes. GT has an incidence of approximately 1 per 1, 000, 000 individuals worldwide [
1]. The genes for both GPIIb and GPIIIa are located in chromosome 17q21-23. The GPIIb gene (ITGA2B) has 30 exons, while the GPIIIa gene (ITGB3) has 15 exons [
2]. More than 400 mutations or deletions in the ITGA2B and ITGB3 genes have been identified [
2]. GT has an acquired form, which occurs secondary to malignancy, lymphoproliferative disorders, autoimmune diseases, and drugs. Acquired GT is attributed to the presence of autoantibodies, alloantibodies, or myeloma paraproteins specific for the platelet glycoprotein IIb/IIIa complex [
3]. Glycoprotein IIb/IIIa is imperative for facilitating the fibrinogen bridging of platelets to other platelets [
4]. As a result of the absence of fibrinogen bridging in GT, some patients experience significant bleeding such as menorrhagia, epistaxis, hemoptysis, and gastrointestinal hemorrhage. The platelet count and morphology are frequently normal, with a characteristic pattern observed in the platelet aggregation tests. Platelet flow cytometry is useful for evaluating glycoproteins such as GPIa/IIa (CD31 and CD49b), GPIV (CD36), and GPVI to identify GT and its variants [
5]. Platelets showed no activity with GPIIb (CD41) or GPIIIa (CD61) on flow cytometry. Crossed immunoelectrophoresis of platelet membrane proteins or electron microscopy utilizing immuno-gold-labeled fibrinogen is also a method for diagnosing GT [
5]. Many patients with this disorder experience injudicious use of blood products due to the occurrence of bleeding symptoms, as many clinicians are not familiar with non-transfusion blood management alternatives. Unnecessary allogeneic blood transfusion increases the risks of adverse transfusion reactions, transfusion-associated dyspnea, anaphylaxis, and alloimmunization [
6]. Patient blood management (PBM) is a multidisciplinary approach for optimizing anemia in patients who are at risk of blood transfusion. The term PBM was first coined in 2005 by an Australian hematologist, Professor James Isbister [
7]. The Society for the Advancement of Blood Management defines PBM as a timely application of evidence-based medical and surgical concepts designed to maintain hemoglobin concentration, optimize hemostasis, and minimize blood loss in an effort to improve patient’s outcomes. PBM is based on three main pillars: optimizing the patient’s endogenous red blood cell mass, minimizing blood loss, and harnessing and optimizing the patient-specific tolerance of anemia and maximizing oxygen delivery, including restrictive transfusion strategies [
8]. The platelet management strategies for patients with GT include observing the clinical indications for prophylactic donor-specific and HLA-matched platelet transfusions, prior to the performance of surgical interventions or during the peripartum period. Unnecessary or chronic use of platelet transfusions may invariably lead to platelet alloimmunization and platelet transfusion refractoriness. PBM programs for both red cells and platelets have been implemented in Ampang Hospital since 2013, and a wide range of blood conservation strategies have been utilized. This study aimed to assess the clinical presentations and outcomes of PBM in our cohort of patients with GT.
Go to :

MATERIALS AND METHODS
A single-center retrospective observational study of 10 patients diagnosed with GT was performed in a tertiary care hospital. Ampang Hospital is a tertiary care hospital in Malaysia that is dedicated to treating patients with hematological and bleeding diseases. This hospital is located in the Selangor state. All 10 patients met all the inclusion criteria and were included in the study.
Inclusion criteria
Patients with a clear diagnosis of congenital GT (men and women of any age) presenting with a lifelong bleeding tendency
Patients demonstrating good observance of treatment and compliance to follow-up
Patients with a minimum follow-up of 9 years at the bleeding clinic
Exclusion criteria
Data of these patients within the period of 9 years (2009–2018) were collected. Our patients with GT were diagnosed using platelet flow cytometric analysis and platelet light transmission aggregometry testing to determine the presence of agonists such as collagen, adrenaline, arachidonic acid, adenosine diphosphate, and ristocetin. The platelets were identified based on their light scatter characteristics and the markedly decreased CD41 (GPIIb) and CD61 (GPIIIa) expression levels. The platelet function of all patients was measured under high shear using a platelet function analyzer (PFA)-100 (Siemens Healthineers, Erlangen, Germany). Genetic/molecular studies assessing the mutations in the ITGA2B and ITGB3 genes were unavailable at our center. Patients with acquired thrombasthenia were excluded due to the absence of antiplatelet autoantibodies (antibodies with specificity against GPIIb/IIIa) in the their serum, which was measured using the monoclonal antibody immobilization of platelet antigen method with a commercial enzyme-linked immunosorbent assay kit (apDia, Turnhout, Belgium). Clinical data, including age, sex, family history of bleeding tendencies, parental consanguinity, baseline hemoglobin (Hb) levels, bleeding symptoms, transfusion history (red cell and/or platelet units), iron stores, and PBM methods, including their complications, were obtained from the electronic hospital information system database. Anemia was defined as Hb levels <13 g/dL in men and 11.5 g/dL in women. Iron deficiency was defined as a serum ferritin level of <30 ng/mL. Other additional hematological parameters used to confirm iron deficiency were serum iron, transferrin saturation, mean corpuscular volume, mean corpuscular Hb, red cell distribution width, and absolute reticulocyte count. Non-transfusion PBM therapies employed for anemia optimization were oral iron, parenteral iron formulations such as iron sucrose (Venofer) or low molecular weight iron dextran (CosmoFer), erythropoiesis-stimulating agents such as subcutaneous recombinant erythropoietin-alpha (Eprex), combined hormonal therapy, recombinant human coagulation factor V11a (NovoSeven), and antifibrinolytic. Intravenous iron sucrose was administered at a dose of 200 mg three times a week. The blood usage data were verified by comparison with blood bank records. The primary clinical outcomes were Hb levels associated with non-transfusion PBM methods and the proportion of patients who received blood transfusion (red cell and/or platelet units). The secondary outcomes were morbidity and mortality due to non-transfusion PBM therapy.
Data were exported into Microsoft Excel files, which were then entered into SPSS using IBM SPSS version 21 (IBM Corp., Armonk, NY, USA). Continuous data were presented as mean, standard deviation (SD), minimum (min), and maximum (max) values. Categorical data were presented as numbers and percentages of individuals in each category. Student’s t-test was used to compare the continuous covariates of interest, while chi-square test was used to compare the categorical covariates of interest. All tests were two tailed with a probability of <0.05, which was considered significant. Data analyses were performed using IBM SPSS software version 21.
This study was registered with the National Medical Research Register (NMRR), Malaysia. NMRR ID number: NMRR-20-234-53359 as the reporting was anonymous, consent was waived.
Go to :

DISCUSSION
GT commonly presents with a variety of bleeding symptoms that may be observed during the first weeks of life or in early childhood. The mean age at diagnosis was 8.2 years, which is consistent with the mean age at diagnosis of 8 years reported by Toogeh
et al. [
9]. Our study demonstrated a higher incidence of this disease in women than in men, which may not be significant, although we were not able to demonstrate the presence of sporadic or spontaneous mutations as genetic studies were unavailable. GT is usually inherited in an autosomal recessive fashion, with both men and women equally affected.
GT is classified into three groups: type 1, type II, and type III (variant) [
10]. Type 1 is most common in patients with less than 5% GPIIb/IIIa, type II: 5–20% of the normal amount of platelet glycoprotein, and type III (variant): 50–100% of platelet glycoprotein with abnormal fibrinogen function. Most of our study patients presented with various bleeding phenotypes, ranging from simple gingival bleeding to significant menorrhagia. However, none of them developed life-threatening bleeding symptoms, such as intracranial, nasopharyngeal, or intracranial hemorrhage. There was little correlation between the severity of bleeding and platelet glycoprotein levels, which was consistent with our study findings [
11].
Gingival bleeding and menorrhagia were the most common symptoms observed in our study population. This finding is consistent with that of the study conducted by George
et al. [
12], who reported a menorrhagia incidence of 98% in patients with GT. All nine patients with menorrhagia in this study were assessed, and none of them were found to have any secondary organic causes by our gynecology colleagues. Menorrhagia was probably attributed to the platelet glycoprotein defects associated with GT. The high frequency of gingival bleeding may be due to other concomitant factors, such as poor gingival hygiene and lack of self-awareness. Hence, our patients underwent regular dental follow-ups within 3 to 6 months. They were advised to maintain good oral hygiene, brush their teeth at least twice a day using a medium texture bristle toothbrush or a sponge toothbrush with fluoride-containing toothpaste, floss their teeth daily, and apply topical tranexamic acid. All patients showed significant improvements in their dental hygiene.
Parental consanguinity is an important risk factor for GTs. It is more common in certain ethnic groups such as Iraqi Jews, Jordan, Iranians, and Southern Indians [
13]. Our study revealed parental consanguinity in 60% of patients as opposed to 94% consanguinity reported by Farsinejad
et al. [
14] in a study of 123 patients with GT in Iran.
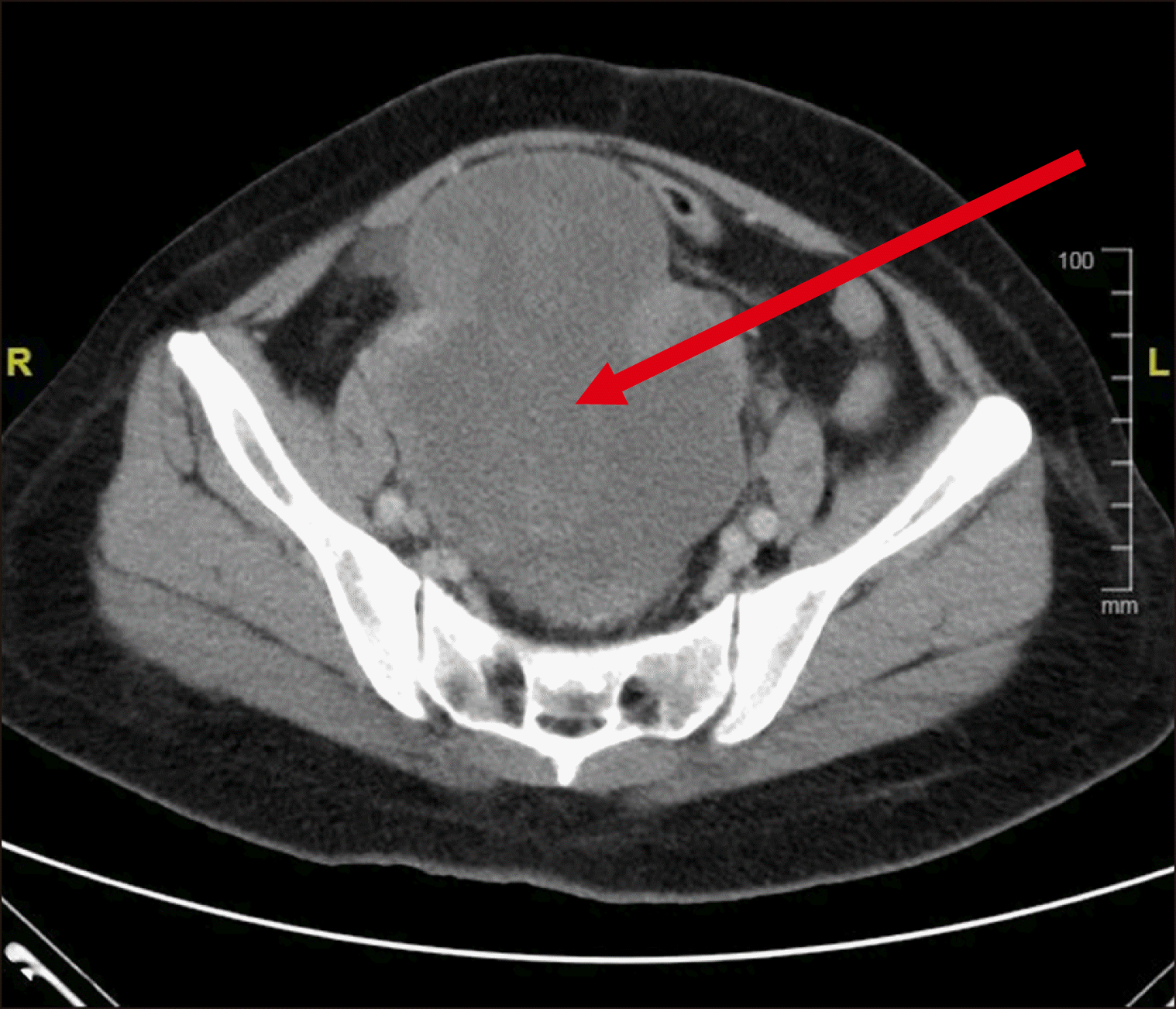
One patient in our study presented with acute lower abdominal pain and swelling and was found to have a tender large hematoma in the pelvis. The pelvic hematoma could have been caused by recurrent ovulatory bleeding, which is frequently associated with the rupture of ovarian cysts in women with bleeding disorders [
15]. Chronic ovulatory bleeding could result in hemoperitoneum, subfertility, ovarian cysts, bleeding into the corpus luteum, and endometriosis.
One patient experienced hemarthrosis of the left knee, which is rarely observed in those with GT and is more commonly associated with hemophilia. Although hemarthrosis rarely occurs in individuals with GT, it may be the only presenting symptom of patients with this condition.
Two of our patients conceived spontaneously and delivered the baby through spontaneous vaginal delivery. As prophylaxis during the puerperium period, the patients were administered with 1 g of intravenous tranexamic acid and recombinant factor VIIa when the cervical os was dilated at 8 cm. A bolus of oxytocin followed by infusion was administered soon after child birth. Cross-matched compatible platelets were requested as a precaution, but the platelets were not transfused. Vaginal delivery is usually the preferred mode of delivery in patients with GT. Platelet transfusion should be avoided due to the risk of platelet alloimmunization, which may result in neonatal alloimmune thrombocytopenia. Regional anesthesia, vacuum-assisted, and high-forceps deliveries are generally contraindicated in patients with GT.
Iron deficiency anemia was observed in all study participants. All patients demonstrated hypochromic microcytic anemia on complete blood count and previously received red cell or platelet transfusions. Those who received platelet transfusions frequently reported anaphylaxis and febrile reactions. Platelet transfusions should be avoided to prevent the production of antiplatelet glycoprotein antibodies (anti-GPIIb/IIIa), platelet transfusion refractoriness, and platelet alloimmunization [
16]. Platelet transfusion should be only be administered in patients with severe bleeding and should be donor specific, cross-matched compatible, or HLA matched [
16]. As most female patients with GT experience chronic prolonged menorrhagia, iron deficiency is usually the most common cause of anemia. Iron replacement therapy would be more appropriate than red cell transfusion because unnecessary transfusion in a hemodynamically stable patient may result in a variety of complications such as hemolytic transfusion reactions, red cell alloimmunization, bacterial contamination, transfusion-transmitted infections, anaphylaxis, and transfusion-related immuno-modulation.
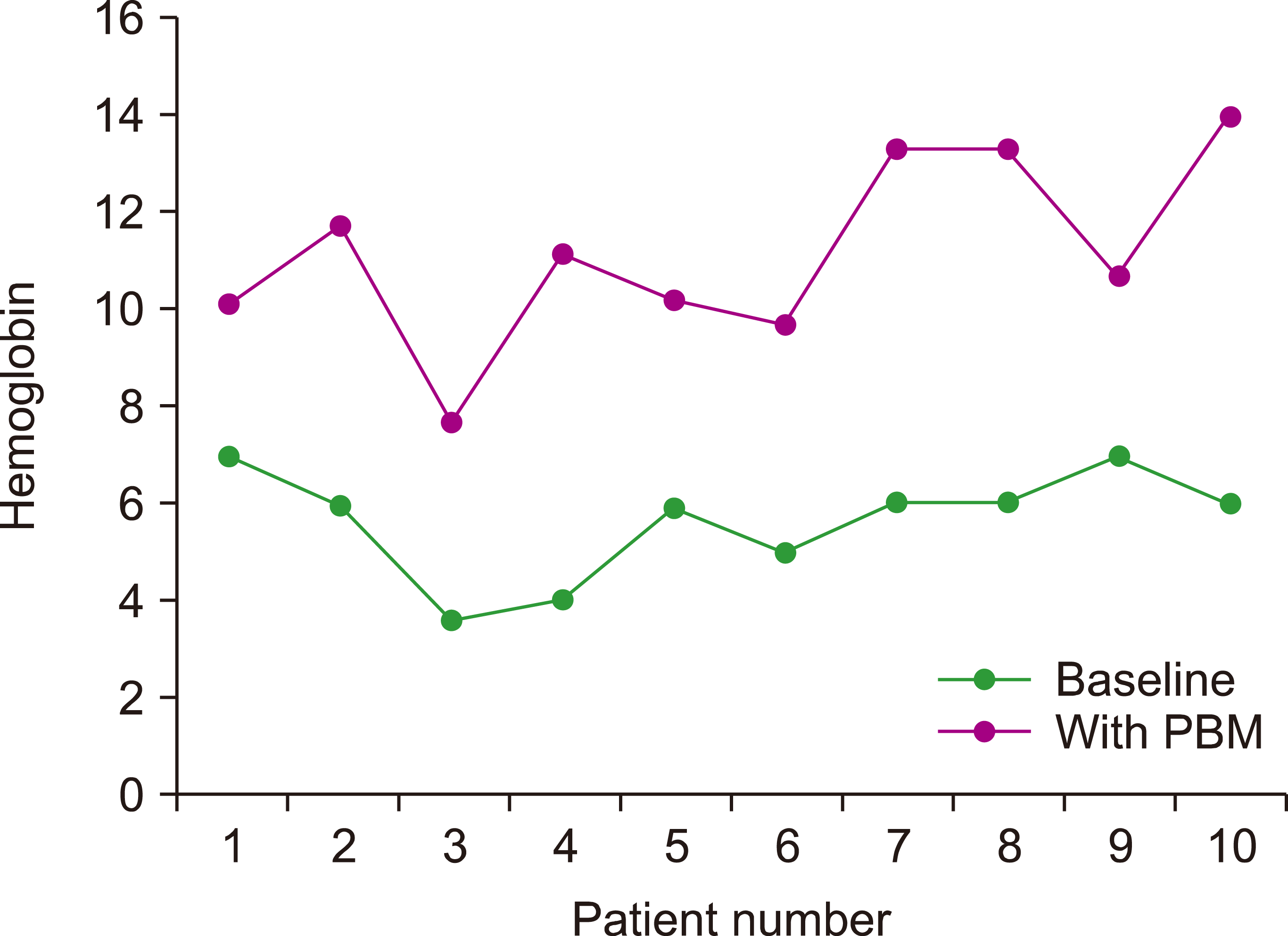
All patients in this study received intravenous iron, such as iron sucrose or iron dextran, administered thrice weekly in our day care. These were well tolerated, with no significant adverse effects reported. Intravenous iron significantly improved the patients’ Hb and ferritin concentrations, and these findings were also observed in other reports [
17].
Tranexamic acid plays a prominent role in reducing blood loss in patients with bleeding disorders. Tranexamic acid is an antifibrinolytic synthetic analog of the amino acid lysine and binds to the lysine receptor sites of plasminogen and plasmin, thus preventing fibrin degradation and preserving the matrix structure of fibrin [
18]. It has shown good efficacy in controlling various bleeding disorders, particularly gingival bleeding and menorrhagia. Intravenous and topical tranexamic acid is more effective than oral tranexamic acid alone in reducing blood loss without any noticeable increase in the incidence of thromboembolic complications, which was consistent with the findings of our study [
19]. Other antifibrinolytic agents unavailable at our center, such as epsilon-aminocaproic acid and aprotinin, are less potent and may affect the kidneys.
All female patients in this study developed menorrhagia, with one patient demonstrating severe pelvic pain due to a pelvic hematoma. Use of a combination of hormonal contraceptives containing estrogen and a progestogen suppressed ovulation, thereby preventing the development of hemorrhagic cysts and reducing heavy menstrual flow. Other options for inducing therapeutic amenorrhea include a progestogen-containing intrauterine system (Mirena coil) or non-hormonal copper coil, which requires replacement every 5–10 years; gonadotrophin-releasing hormone analogs such as subdermal implants, transdermal combination contraceptives, vaginal contraceptive devices; and depot medroxyprogesterone acetate [
20]. All nine female patients in our study were on hormonal therapy, which was effective in preventing the need for blood transfusion. However, these hormone-based regimens have limitations due to their inability to completely suppress menstruation, which could be attributed to various factors. These include the initiation of therapy at varying phases of the menstrual cycle and the status of the hypothalamic-pituitary-ovarian axis, which may enhance ovulatory or anovulatory cycles [
21].
Six patients used recombinant human coagulation factor VIIa to prevent menorrhagia as they planned to conceive. This bypassing agent promotes hemostasis by activating the extrinsic coagulation pathway cascade and is a vitamin K-dependent glycoprotein consisting of 406 amino acid residues [
22]. Use of this agent successfully prevented bleeding episodes in 94.4% of GT patients and prohibited the necessary performance of surgical interventional procedures in 99.4% of these patients [
23]. None of our patients developed side effects such as venous thromboembolism, pyrexia, or dizziness.
The implementation of PBM in patients with congenital bleeding disorders such as GT has effectively reduced the cost related to blood transfusion. PBM programs may assist bleeding clinics in reducing and, on several occasions, eliminating the need for blood transfusions, as demonstrated clearly in this study. The average costs of transfusing one unit of red blood cells and one unit of random platelets in a Malaysian public healthcare facility (based on the seventh fee schedule) are approximately RM 500 (USD 120) and RM 170 (USD 41), respectively. A higher cost incurred is if the patients stay longer in the ward due to the occurrence of blood transfusion-related reactions and transfusion-transmitted infections. Most of the patients in this study received an average of 5 units of red blood cell and 7 units of random platelet transfusions annually before PBM was implemented. This would translate to a significant cost saving of approximately RM 3,690 (USD 882) per patient annually. Intravenous iron (Venofer/Cosmofer) at a dose of 200 mg and subcutaneous recombinant erythropoietin-alpha at 4,000 IU per dose cost RM 28 (USD 6.70) and RM 35 (USD 8.40), respectively. A 2 mg dose of intravenous recombinant factor VIIa costs RM1, 500 (USD 358), whereas a month’s supply of combined oral contraceptive pills only costs RM 30 (USD 7.20).
Our study has three main limitations. First, the number of patients included was relatively small, although it was consistent with that in a single-center study, which included patients with an extremely rare bleeding disorder such as GT. Second, the majority of our study population included women with menorrhagia. Hence, we were unable to apply these blood management strategies to men, as their bleeding patterns may differ. Third, a prospective study analyzing blood management strategies in patients newly diagnosed with GT would be helpful.
Go to :





 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download