1. Urban P, Mehran R, Colleran R, Angiolillo DJ, Byrne RA, Capodanno D, et al. Defining high bleeding risk in patients undergoing percutaneous coronary intervention: a consensus document from the Academic Research Consortium for High Bleeding Risk. Eur Heart J. 2019; 40(31):2632–2653. PMID:
31116395.
2. Park KH, Jeong MH, Ahn Y, Ahn TH, Seung KB, Oh DJ, et al. Comparison of short-term clinical outcomes between ticagrelor versus clopidogrel in patients with acute myocardial infarction undergoing successful revascularization; from Korea Acute Myocardial Infarction Registry-National Institute of Health. Int J Cardiol. 2016; 215:193–200. PMID:
27128530.

3. Kang J, Han JK, Ahn Y, Chae SC, Kim YJ, Chae IH, et al. Third-generation P2Y12 inhibitors in East Asian acute myocardial infarction patients: a nationwide prospective multicentre study. Thromb Haemost. 2018; 118(3):591–600. PMID:
29534250.

4. Park DW, Kwon O, Jang JS, Yun SC, Park H, Kang DY, et al. Clinically significant bleeding with ticagrelor versus clopidogrel in Korean patients with acute coronary syndromes intended for invasive management: a randomized clinical trial. Circulation. 2019; 140(23):1865–1877. PMID:
31553203.
5. Yun JE, Kim YJ, Park JJ, Kim S, Park K, Cho MS, et al. Safety and effectiveness of contemporary P2Y12 inhibitors in an East Asian population with acute coronary syndrome: a nationwide population-based cohort study. J Am Heart Assoc. 2019; 8(14):e012078. PMID:
31310570.

6. You SC, Rho Y, Bikdeli B, Kim J, Siapos A, Weaver J, et al. Association of ticagrelor vs clopidogrel with net adverse clinical events in patients with acute coronary syndrome undergoing percutaneous coronary intervention. JAMA. 2020; 324(16):1640–1650. PMID:
33107944.

7. Kim JH, Chae SC, Oh DJ, Kim HS, Kim YJ, Ahn Y, et al. Multicenter cohort study of acute myocardial infarction in Korea interim analysis of the Korea Acute Myocardial Infarction Registry-National Institutes of Health Registry. Circ J. 2016; 80(6):1427–1436. PMID:
27118621.
8. Cao D, Mehran R, Dangas G, Baber U, Sartori S, Chandiramani R, et al. Validation of the academic research consortium high bleeding risk definition in contemporary PCI patients. J Am Coll Cardiol. 2020; 75(21):2711–2722. PMID:
32466887.

9. Natsuaki M, Morimoto T, Shiomi H, Yamaji K, Watanabe H, Shizuta S, et al. Application of the Academic Research Consortium High Bleeding Risk criteria in an all-comers registry of percutaneous coronary intervention. Circ Cardiovasc Interv. 2019; 12(11):e008307. PMID:
31707804.

10. Ueki Y, Bär S, Losdat S, Otsuka T, Zanchin C, Zanchin T, et al. Validation of the Academic Research Consortium for High Bleeding Risk (ARC-HBR) criteria in patients undergoing percutaneous coronary intervention and comparison with contemporary bleeding risk scores. EuroIntervention. 2020; 16(5):371–379. PMID:
32065586.

11. Kim HK, Tantry US, Smith SC Jr, Jeong MH, Park SJ, Kim MH, et al. The East Asian paradox: an updated position statement on the challenges to the current antithrombotic strategy in patients with cardiovascular disease. Thromb Haemost. 2021; 121(4):422–432. PMID:
33171520.

12. Navarese EP, Khan SU, Kołodziejczak M, Kubica J, Buccheri S, Cannon CP, et al. Comparative efficacy and safety of oral P2Y12 inhibitors in acute coronary syndrome: network meta-analysis of 52 816 patients from 12 randomized trials. Circulation. 2020; 142(2):150–160. PMID:
32468837.
13. Wallentin L, Becker RC, Budaj A, Cannon CP, Emanuelsson H, Held C, et al. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2009; 361(11):1045–1057. PMID:
19717846.

14. Völz S, Petursson P, Odenstedt J, Ioanes D, Haraldsson I, Angerås O, et al. Ticagrelor is not superior to clopidogrel in patients with acute coronary syndromes undergoing PCI: a report from Swedish Coronary Angiography and Angioplasty Registry. J Am Heart Assoc. 2020; 9(14):e015990. PMID:
32662350.

15. Park Y, Koh JS, Lee JH, Park JH, Shin ES, Oh JH, et al. Effect of ticagrelor on left ventricular remodeling in patients with ST-segment elevation myocardial infarction (HEALING-AMI). JACC Cardiovasc Interv. 2020; 13(19):2220–2234. PMID:
33032710.
16. Kim HK, Jeong MH, Lim KS, Kim JH, Lim HC, Kim MC, et al. Effects of ticagrelor on neointimal hyperplasia and endothelial function, compared with clopidogrel and prasugrel, in a porcine coronary stent restenosis model. Int J Cardiol. 2017; 240:326–331. PMID:
28487152.

17. Jeong HS, Hong SJ, Cho SA, Kim JH, Cho JY, Lee SH, et al. Comparison of ticagrelor versus prasugrel for inflammation, vascular function, and circulating endothelial progenitor cells in diabetic patients with non-ST-segment elevation acute coronary syndrome requiring coronary stenting: a prospective, randomized, crossover trial. JACC Cardiovasc Interv. 2017; 10(16):1646–1658. PMID:
28838475.
18. Saito S, Isshiki T, Kimura T, Ogawa H, Yokoi H, Nanto S, et al. Efficacy and safety of adjusted-dose prasugrel compared with clopidogrel in Japanese patients with acute coronary syndrome: the PRASFIT-ACS study. Circ J. 2014; 78(7):1684–1692. PMID:
24759796.
19. Kim HS, Kang J, Hwang D, Han JK, Yang HM, Kang HJ, et al. Prasugrel-based de-escalation of dual antiplatelet therapy after percutaneous coronary intervention in patients with acute coronary syndrome (HOST-REDUCE-POLYTECH-ACS): an open-label, multicentre, non-inferiority randomised trial. Lancet. 2020; 396(10257):1079–1089. PMID:
32882163.

20. Hahn JY, Song YB, Oh JH, Cho DK, Lee JB, Doh JH, et al. 6-month versus 12-month or longer dual antiplatelet therapy after percutaneous coronary intervention in patients with acute coronary syndrome (SMART-DATE): a randomised, open-label, non-inferiority trial. Lancet. 2018; 391(10127):1274–1284. PMID:
29544699.

21. Kedhi E, Fabris E, van der Ent M, Buszman P, von Birgelen C, Roolvink V, et al. Six months versus 12 months dual antiplatelet therapy after drug-eluting stent implantation in ST-elevation myocardial infarction (DAPT-STEMI): randomised, multicentre, non-inferiority trial. BMJ. 2018; 363:k3793. PMID:
30279197.

22. Vranckx P, Valgimigli M, Jüni P, Hamm C, Steg PG, Heg D, et al. Ticagrelor plus aspirin for 1 month, followed by ticagrelor monotherapy for 23 months vs aspirin plus clopidogrel or ticagrelor for 12 months, followed by aspirin monotherapy for 12 months after implantation of a drug-eluting stent: a multicentre, open-label, randomised superiority trial. Lancet. 2018; 392(10151):940–949. PMID:
30166073.
23. Mehran R, Baber U, Sharma SK, Cohen DJ, Angiolillo DJ, Briguori C, et al. Ticagrelor with or without aspirin in high-risk patients after PCI. N Engl J Med. 2019; 381(21):2032–2042. PMID:
31556978.

24. Kim BK, Hong SJ, Cho YH, Yun KH, Kim YH, Suh Y, et al. Effect of ticagrelor monotherapy vs ticagrelor with aspirin on major bleeding and cardiovascular events in patients with acute coronary syndrome: the TICO randomized clinical trial. JAMA. 2020; 323(23):2407–2416. PMID:
32543684.
25. Watanabe H, Domei T, Morimoto T, Natsuaki M, Shiomi H, Toyota T, et al. Effect of 1-month dual antiplatelet therapy followed by clopidogrel vs 12-month dual antiplatelet therapy on cardiovascular and bleeding events in patients receiving PCI: the STOPDAPT-2 randomized clinical trial. JAMA. 2019; 321(24):2414–2427. PMID:
31237644.
26. Cuisset T, Deharo P, Quilici J, Johnson TW, Deffarges S, Bassez C, et al. Benefit of switching dual antiplatelet therapy after acute coronary syndrome: the TOPIC (timing of platelet inhibition after acute coronary syndrome) randomized study. Eur Heart J. 2017; 38(41):3070–3078. PMID:
28510646.

27. Sibbing D, Aradi D, Jacobshagen C, Gross L, Trenk D, Geisler T, et al. Guided de-escalation of antiplatelet treatment in patients with acute coronary syndrome undergoing percutaneous coronary intervention (TROPICAL-ACS): a randomised, open-label, multicentre trial. Lancet. 2017; 390(10104):1747–1757. PMID:
28855078.
28. Miura K, Shimada T, Ohya M, Murai R, Amano H, Kubo S, et al. Prevalence of the academic research consortium for high bleeding risk criteria and prognostic value of a simplified definition. Circ J. 2020; 84(9):1560–1567. PMID:
32669530.

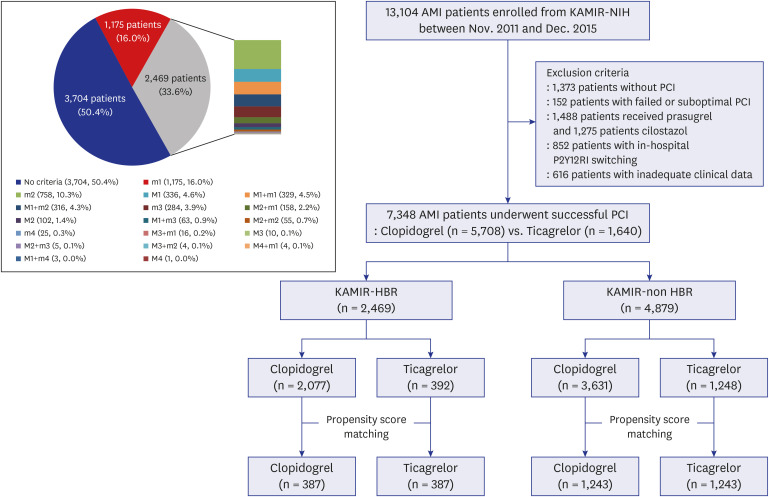




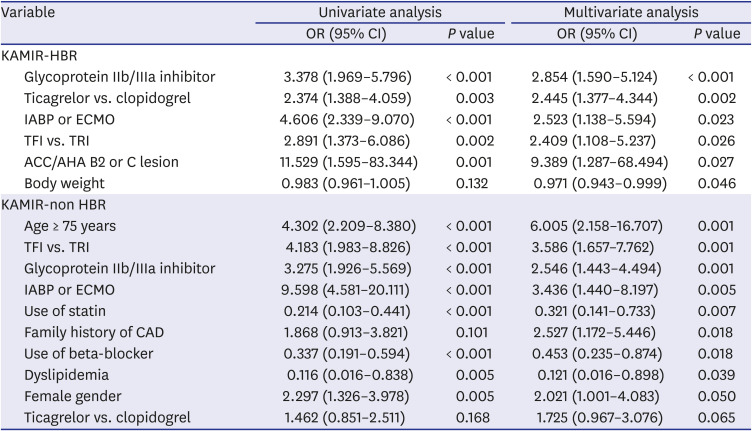
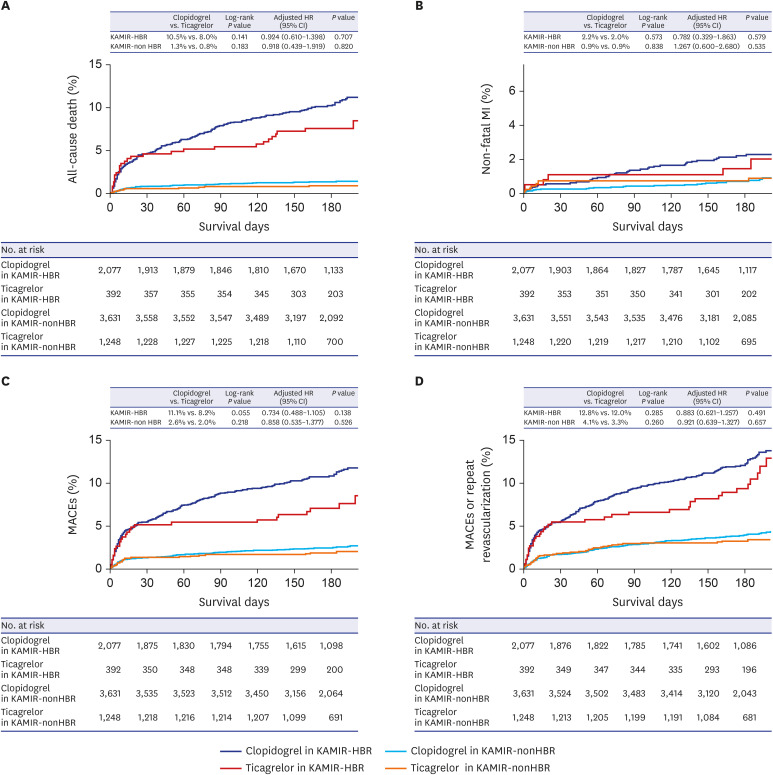





 PDF
PDF Citation
Citation Print
Print





 XML Download
XML Download