1. Lundy RF Jr. Paxinos G, editor. 2004. Gustatory system. The rat nervous system. 3rd ed. Elsevier Academic Press;Cambridge: p. 891–921. DOI:
10.1016/B978-012547638-6/50029-8.

3. Rolls ET. 2004; Smell, taste, texture, and temperature multimodal representations in the brain, and their relevance to the control of appetite. Nutr Rev. 62(11 Pt 2):S193–S204. discussion S224–S241. DOI:
10.1301/nr.2004.nov.S193-S204. PMID:
15630935.

5. Rolls ET. 1999. The brain and emotion. Oxford University Press;New York:
7. Giza BK, Deems RO, Vanderweele DA, Scott TR. 1993; Pancreatic glucagon suppresses gustatory responsiveness to glucose. Am J Physiol. 265(6 Pt 2):R1231–R1237. DOI:
10.1152/ajpregu.1993.265.6.R1231. PMID:
8285262.

8. Giza BK, Scott TR. 1983; Blood glucose selectively affects taste-evoked activity in rat nucleus tractus solitarius. Physiol Behav. 31:643–650. PMID:
6665054.
9. Giza BK, Scott TR. 1987; Intravenous insulin infusions in rats decrease gustatory-evoked responses to sugars. Am J Physiol. 252(5 Pt 2):R994–R1002. DOI:
10.1152/ajpregu.1987.252.5.R994. PMID:
3555122.

10. Whitehead MC. 1988; Neuronal architecture of the nucleus of the solitary tract in the hamster. J Comp Neurol. 276:547–572. DOI:
10.1002/cne.902760409. PMID:
2461969.

12. Giza BK, Ackroff K, McCaughey SA, Sclafani A, Scott TR. 1997; Preference conditioning alters taste responses in the nucleus of the solitary tract of the rat. Am J Physiol. 273:R1230–R1240. DOI:
10.1152/ajpregu.1997.273.4.R1230. PMID:
9362285.
14. Bachmanov AA, Kiefer SW, Molina JC, Tordoff MG, Duffy VB, Bartoshuk LM, Mennella JA. 2003; Chemosensory factors influencing alcohol perception, preferences, and consumption. Alcohol Clin Exp Res. 27:220–231. DOI:
10.1097/01.ALC.0000051021.99641.19. PMID:
12605071. PMCID:
PMC1940064.

16. Kiefer SW, Morrow NS, Metzler CW. 1988; Alcohol aversion generalization in rats: specific disruption of taste and odor cues with gustatory neocortex or olfactory bulb ablations. Behav Neurosci. 102:733–739. DOI:
10.1037/0735-7044.102.5.733. PMID:
2848538.

17. Kampov-Polevoy AB, Garbutt JC, Janowsky DS. 1999; Association between preference for sweets and excessive alcohol intake: a review of animal and human studies. Alcohol Alcohol. 34:386–395. DOI:
10.1093/alcalc/34.3.386. PMID:
10414615.

18. Hellekant G, Danilova V, Roberts T, Ninomiya Y. 1997; The taste of ethanol in a primate model: I. Chorda tympani nerve response in Macaca mulatta. Alcohol. 14:473–484. DOI:
10.1016/S0741-8329(96)00215-7. PMID:
9305463.

19. Sako N, Yamamoto T. 1999; Electrophysiological and behavioral studies on taste effectiveness of alcohols in rats. Am J Physiol. 276:R388–R396. DOI:
10.1152/ajpregu.1999.276.2.R388. PMID:
9950916.

20. Lemon CH, Brasser SM, Smith DV. 2004; Alcohol activates a sucrose-responsive gustatory neural pathway. J Neurophysiol. 92:536–544. DOI:
10.1152/jn.00097.2004. PMID:
14985409.

21. Lemon CH, Wilson DM, Brasser SM. 2011; Differential neural representation of oral ethanol by central taste-sensitive neurons in ethanol-preferring and genetically heterogeneous rats. J Neurophysiol. 106:3145–3156. DOI:
10.1152/jn.00580.2011. PMID:
21918002. PMCID:
PMC3234093.

22. Cho YK. 2007; Relationship between alcohol and sucrose/thermal stimulation in pontine taste neurons in the hamster. Exp Neurobiol. 16:79–87.
24. Bartoshuk LM, Rennert K, Rodin J, Stevens JC. 1982; Effects of temperature on the perceived sweetness of sucrose. Physiol Behav. 28:905–910. DOI:
10.1016/0031-9384(82)90212-8. PMID:
7100291.

25. Green BG, Frankmann SP. 1988; The effect of cooling on the perception of carbohydrate and intensive sweeteners. Physiol Behav. 43:515–519. DOI:
10.1016/0031-9384(88)90127-8. PMID:
3194473.

26. Cho YK, Li CS, Smith DV. 2002; Gustatory projections from the nucleus of the solitary tract to the parabrachial nuclei in the hamster. Chem Senses. 27:81–90. DOI:
10.1093/chemse/27.1.81. PMID:
11751472.

27. Cho YK, Li CS, Smith DV. 2002; Taste responses of neurons of the hamster solitary nucleus are enhanced by lateral hypothalamic stimulation. J Neurophysiol. 87:1981–1992. DOI:
10.1152/jn.00765.2001. PMID:
11929917.

28. Li CS, Lu DP, Cho YK. 2015; Descending projections from the nucleus accumbens shell excite activity of taste-responsive neurons in the nucleus of the solitary tract in the hamster. J Neurophysiol. 113:3778–3786. DOI:
10.1152/jn.00362.2014. PMID:
25744880. PMCID:
PMC4468968.

29. Duncan HJ, Smith DV. 1992; Concentration-response functions for thirty chemical stimuli in the hamster solitary nucleus. Chem Senses. 17:616.
30. Smith DV, Travers JB. 1979; A metric for the breadth of tuning of gustatory neurons. Chem Senses. 4:215–229. DOI:
10.1093/chemse/4.3.215.

31. McPheeters M, Hettinger TP, Nuding SC, Savoy LD, Whitehead MC, Frank ME. 1990; Taste-responsive neurons and their locations in the solitary nucleus of the hamster. Neuroscience. 34:745–758. DOI:
10.1016/0306-4522(90)90179-8. PMID:
2352650.

32. Di Lorenzo PM, Kiefer SW, Rice AG, Garcia J. 1986; Neural and behavioral responsivity to ethyl alcohol as a tastant. Alcohol. 3:55–61. DOI:
10.1016/0741-8329(86)90071-6. PMID:
3964438.

33. Danilova V, Hellekant G. 2000; The taste of ethanol in a primate model. II. Glossopharyngeal nerve response in Macaca mulatta. Alcohol. 21:259–269. DOI:
10.1016/S0741-8329(00)00094-X. PMID:
11091030.
34. Hellekant G. 1965; Electrophysiological investigation of the gustatory effect of ethyl alcohol. II. A single fibre analysis in the cat. Acta Physiol Scand. 64:398–406. DOI:
10.1111/j.1748-1716.1965.tb04197.x. PMID:
5853033.

35. Lemon CH, Imoto T, Smith DV. 2003; Differential gurmarin suppression of sweet taste responses in rat solitary nucleus neurons. J Neurophysiol. 90:911–923. DOI:
10.1152/jn.00215.2003. PMID:
12702710.
36. Kampov-Polevoy AB, Kasheffskaya OP, Sinclair JD. 1990; Initial acceptance of ethanol: gustatory factors and patterns of alcohol drinking. Alcohol. 7:83–85. DOI:
10.1016/0741-8329(90)90065-K. PMID:
2328091.

37. Kampov-Polevoy AB, Overstreet DH, Rezvani AH, Janowsky DS. 1995; Saccharin-induced increase in daily fluid intake as a predictor of voluntary alcohol intake in alcohol-preferring rats. Physiol Behav. 57:791–795. DOI:
10.1016/0031-9384(94)00389-0. PMID:
7777619.

39. Levine AS, Kotz CM, Gosnell BA. 2003; Sugars: hedonic aspects, neuroregulation, and energy balance. Am J Clin Nutr. 78:834S–842S. DOI:
10.1093/ajcn/78.4.834S. PMID:
14522747.

40. Hajnal A, Smith GP, Norgren R. 2004; Oral sucrose stimulation increases accumbens dopamine in the rat. Am J Physiol Regul Integr Comp Physiol. 286:R31–R37. DOI:
10.1152/ajpregu.00282.2003. PMID:
12933362.

42. Smith GP. 2004; Accumbens dopamine mediates the rewarding effect of orosensory stimulation by sucrose. Appetite. 43:11–13. DOI:
10.1016/j.appet.2004.02.006. PMID:
15262012.

43. Weiss F, Lorang MT, Bloom FE, Koob GF. 1993; Oral alcohol self-administration stimulates dopamine release in the rat nucleus accumbens: genetic and motivational determinants. J Pharmacol Exp Ther. 267:250–258. PMID:
8229752.
44. Zhang M, Kelley AE. 2002; Intake of saccharin, salt, and ethanol solutions is increased by infusion of a mu opioid agonist into the nucleus accumbens. Psychopharmacology (Berl). 159:415–423. DOI:
10.1007/s00213-001-0932-y. PMID:
11823894.

45. Loriaux AL, Roitman JD, Roitman MF. 2011; Nucleus accumbens shell, but not core, tracks motivational value of salt. J Neurophysiol. 106:1537–1544. DOI:
10.1152/jn.00153.2011. PMID:
21697439. PMCID:
PMC3174809.

46. Li CS, Chung S, Lu DP, Cho YK. 2012; Descending projections from the nucleus accumbens shell suppress activity of taste-responsive neurons in the hamster parabrachial nuclei. J Neurophysiol. 108:1288–1298. DOI:
10.1152/jn.00121.2012. PMID:
22696536.

47. Hayama T, Ito S, Ogawa H. 1985; Responses of solitary tract nucleus neurons to taste and mechanical stimulations of the oral cavity in decerebrate rats. Exp Brain Res. 60:235–242. DOI:
10.1007/BF00235918. PMID:
4054268.

48. Ogawa H, Hayama T, Yamashita Y. 1988; Thermal sensitivity of neurons in a rostral part of the rat solitary tract nucleus. Brain Res. 454:321–331. DOI:
10.1016/0006-8993(88)90833-5. PMID:
3409015.

49. Ogawa H, Imoto T, Hayama T. 1984; Responsiveness of solitario-parabrachial relay neurons to taste and mechanical stimulation applied to the oral cavity in rats. Exp Brain Res. 54:349–358. DOI:
10.1007/BF00236236. PMID:
6723854.

50. Travers SP, Norgren R. 1995; Organization of orosensory responses in the nucleus of the solitary tract of rat. J Neurophysiol. 73:2144–2162. DOI:
10.1152/jn.1995.73.6.2144. PMID:
7666129.

51. Green BG, George P. 2004; 'Thermal taste' predicts higher responsiveness to chemical taste and flavor. Chem Senses. 2:617–628. DOI:
10.1093/chemse/bjh065. PMID:
15337686.

52. Ogawa H, Sato M, Yamashita S. 1968; Multiple sensitivity of chordat typani fibres of the rat and hamster to gustatory and thermal stimuli. J Physiol. 199:223–240. DOI:
10.1113/jphysiol.1968.sp008650. PMID:
5684036. PMCID:
PMC1365355.
53. Lundy RF Jr, Contreras RJ. 1997; Temperature and amiloride alter taste nerve responses to Na
+, K
+, and NH
4+salts in rats. Brain Res. 744:309–317. DOI:
10.1016/S0006-8993(96)01118-3.
54. Li J, Lemon CH. 2015; Influence of stimulus and oral adaptation temperature on gustatory responses in central taste-sensitive neurons. J Neurophysiol. 113:2700–2712. DOI:
10.1152/jn.00736.2014. PMID:
25673737. PMCID:
PMC4416558.

56. Breza JM, Curtis KS, Contreras RJ. 2006; Temperature modulates taste responsiveness and stimulates gustatory neurons in the rat geniculate ganglion. J Neurophysiol. 95:674–685. DOI:
10.1152/jn.00793.2005. PMID:
16267112.

57. Cho YK, Smith ME, Norgren R. 2004; Low-dose furosemide modulates taste responses in the nucleus of the solitary tract of the rat. Am J Physiol Regul Integr Comp Physiol. 287:R706–R714. DOI:
10.1152/ajpregu.00090.2004. PMID:
15155275.

58. Cho YK, Mao L, Li CS. 2008; Modulation of solitary taste neurons by electrical stimulation of the ventroposteromedial nucleus of the thalamus in the hamster. Brain Res. 1221:67–79. DOI:
10.1016/j.brainres.2008.05.006. PMID:
18565498.

59. Li CS, Cho YK, Smith DV. 2002; Taste responses of neurons in the hamster solitary nucleus are modulated by the central nucleus of the amygdala. J Neurophysiol. 88:2979–2992. DOI:
10.1152/jn.00239.2002. PMID:
12466423.

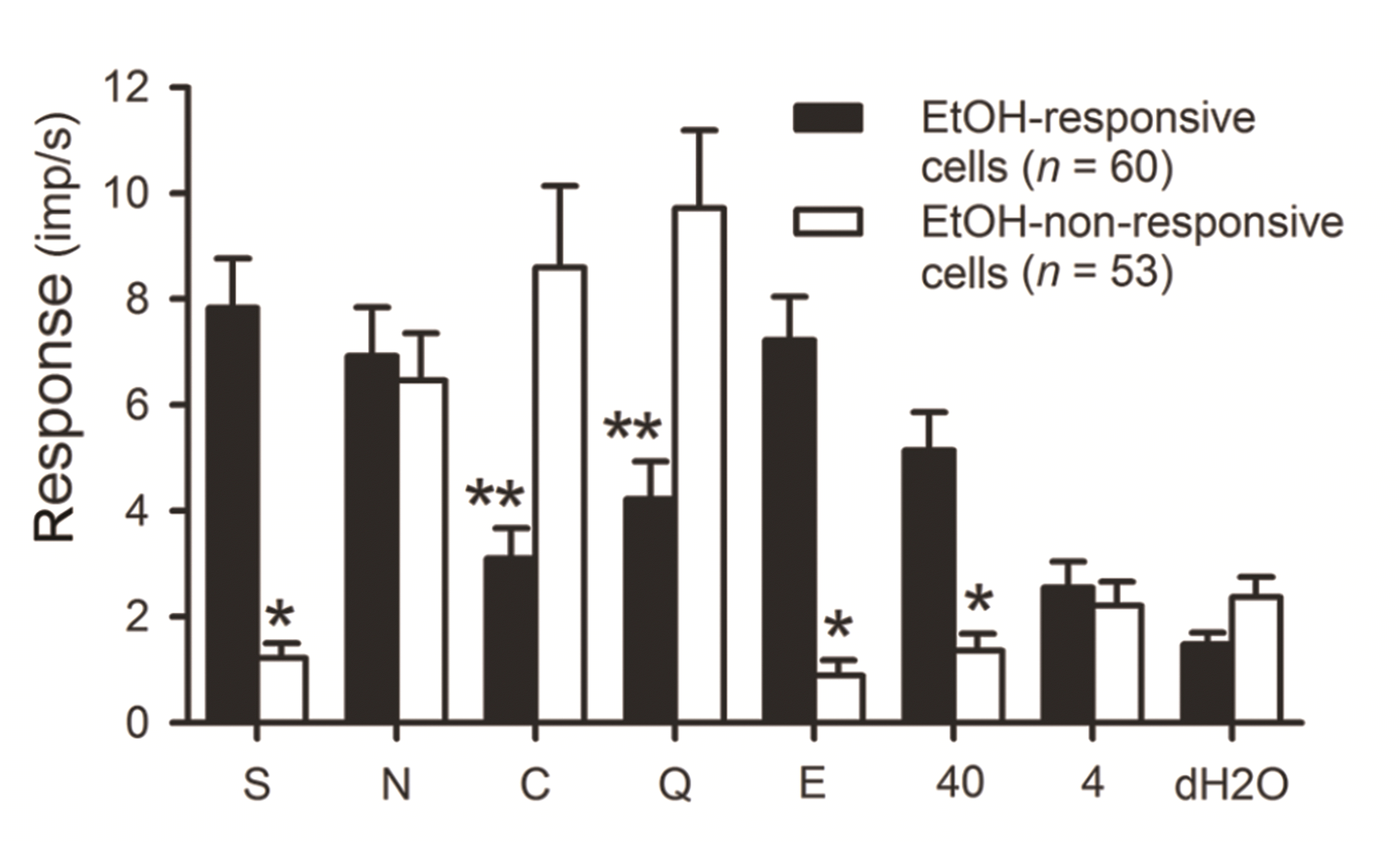
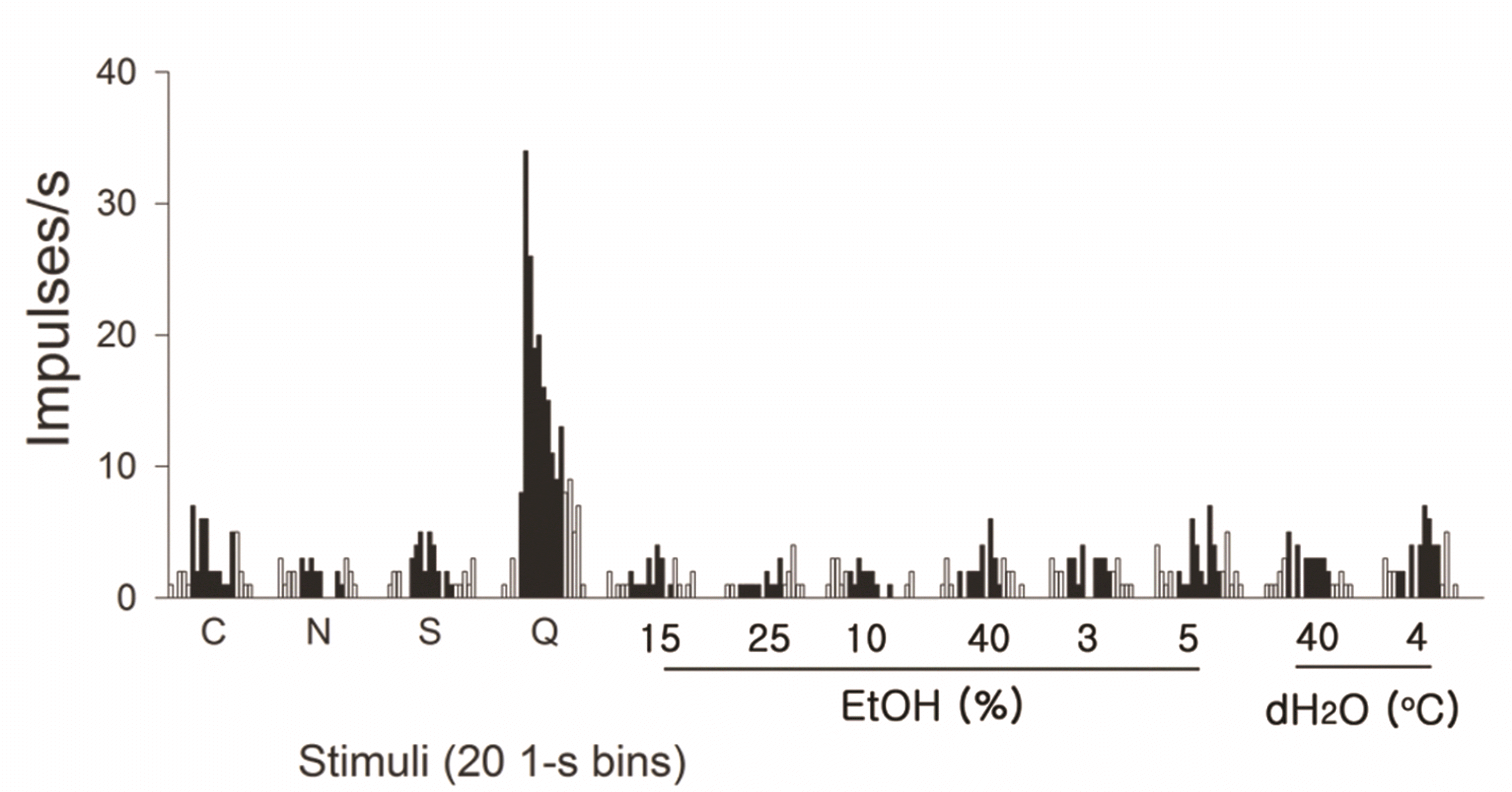
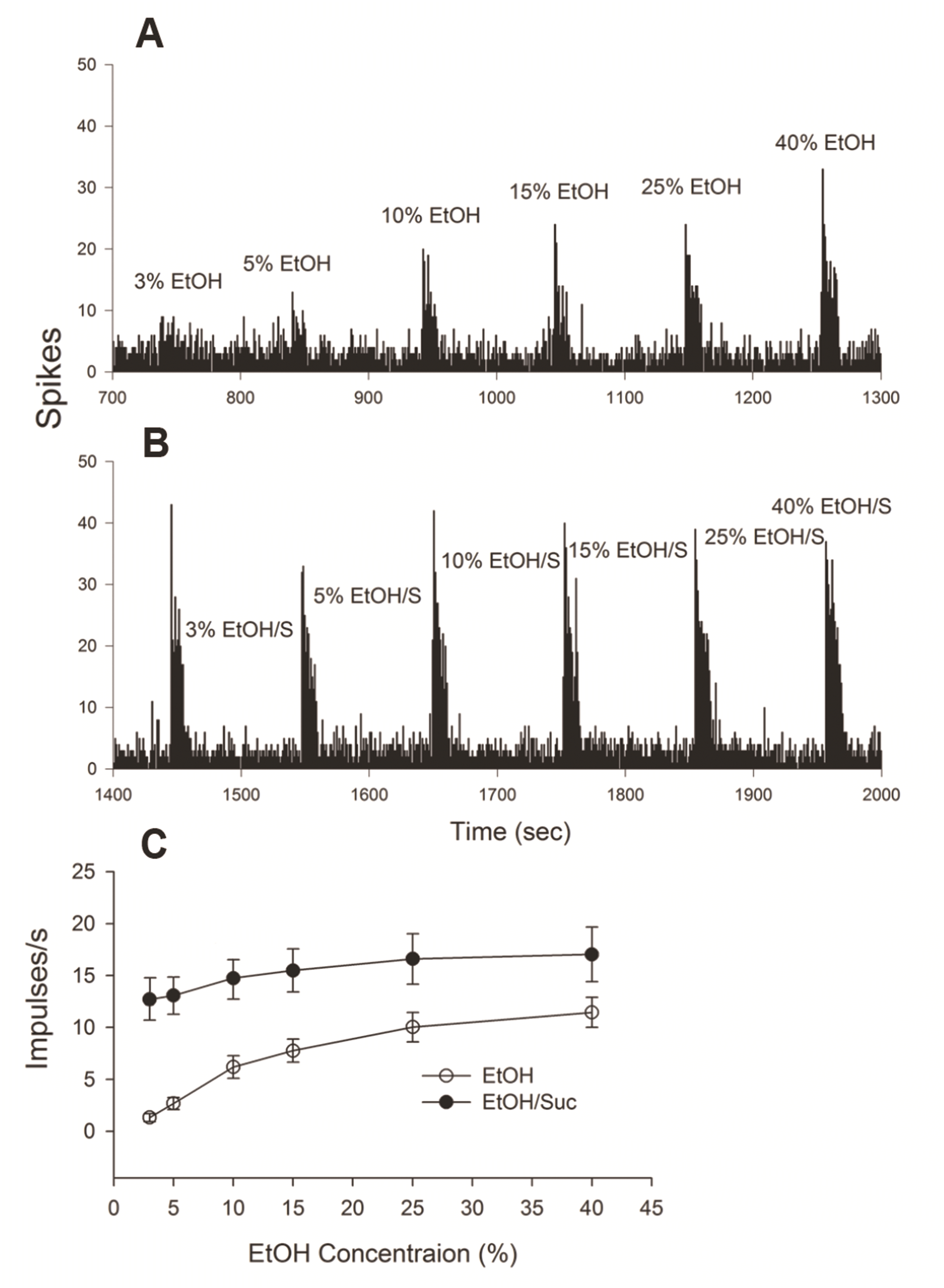




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download